TABLE OF CONTENTS
1 INTRODUCTION (Page No. - 40)
1.1 STUDY OBJECTIVES
1.2 MARKET DEFINITION
1.2.1 INCLUSIONS & EXCLUSIONS
1.3 MARKET SCOPE
1.3.1 MARKETS COVERED
1.3.2 YEARS CONSIDERED
1.4 CURRENCY CONSIDERED
1.5 RESEARCH LIMITATIONS
1.6 STAKEHOLDERS
1.7 SUMMARY OF CHANGES
1.8 RECESSION IMPACT
2 RESEARCH METHODOLOGY (Page No. - 45)
2.1 RESEARCH APPROACH
FIGURE 1 RESEARCH DESIGN
2.1.1 PRIMARY RESEARCH
FIGURE 2 CLINICAL TRIAL SUPPLIES MARKET: PRIMARY RESPONDENTS
2.1.2 SECONDARY DATA
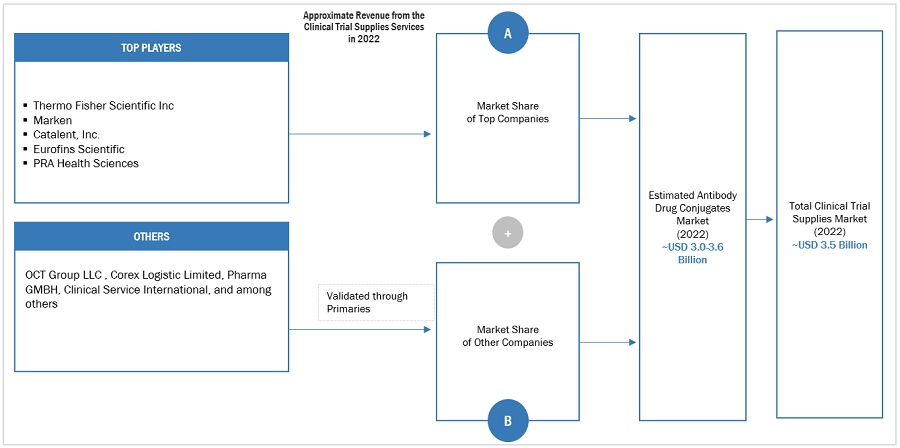
2.2 MARKET SIZE ESTIMATION
FIGURE 3 CLINICAL TRIAL SUPPLIES INDUSTRY SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2022
FIGURE 4 MARKET SIZE ESTIMATION: APPROACH 1 (COMPANY REVENUE ANALYSIS-BASED ESTIMATION), 2022
2.2.1 INSIGHTS FROM PRIMARY
FIGURE 5 MARKET SIZE VALIDATION FROM PRIMARY EXPERTS
2.2.2 SEGMENTAL ASSESSMENT (BY SERVICE, PHASE, TYPE, THERAPEUTIC AREA, END USER, AND REGION)
FIGURE 6 MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
2.3 GROWTH RATE ASSUMPTIONS
FIGURE 7 MARKET (DEMAND SIDE): GROWTH ANALYSIS OF DEMAND-SIDE FACTORS
FIGURE 8 MARKET: GROWTH ANALYSIS OF DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
FIGURE 9 DATA TRIANGULATION METHODOLOGY
2.4 STUDY ASSUMPTIONS
2.5 RISK ANALYSIS
2.6 RECESSION IMPACT ANALYSIS
TABLE 1 GLOBAL INFLATION RATE PROJECTIONS, 2021–2028 (% GROWTH)
TABLE 2 US HEALTH EXPENDITURE, 2019–2022 (USD MILLION)
TABLE 3 US HEALTH EXPENDITURE, 2023–2027 (USD MILLION)
3 EXECUTIVE SUMMARY (Page No. - 57)
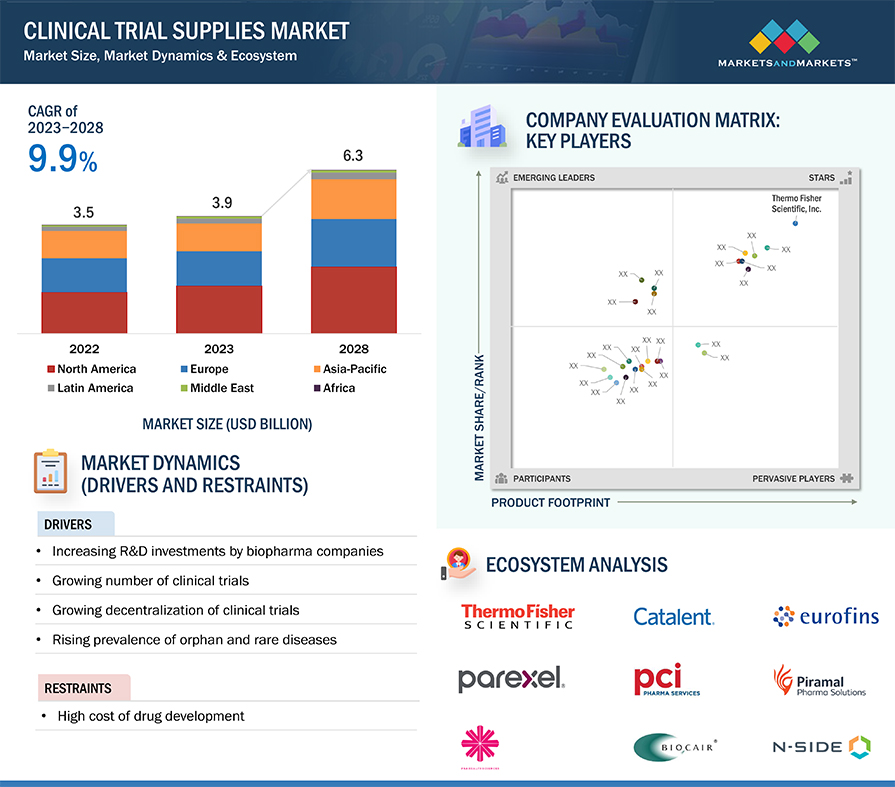
FIGURE 10 CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2023 VS. 2028 (USD MILLION)
FIGURE 11 CLINICAL TRIAL SUPPLIES INDUSTRY, BY TYPE, 2023 VS. 2028 (USD MILLION)
FIGURE 12 MARKET, BY THERAPEUTIC AREA, 2023 VS. 2028 (USD MILLION)
FIGURE 13 MARKET, BY END USER, 2023 VS. 2028 (USD MILLION)
FIGURE 14 MARKET, BY PHASE, 2023 VS. 2028 (USD MILLION)
FIGURE 15 GEOGRAPHICAL SNAPSHOT OF MARKET
4 PREMIUM INSIGHTS (Page No. - 61)
4.1 CLINICAL TRIAL SUPPLIES MARKET OVERVIEW
FIGURE 16 RISING PREVALENCE OF INFECTIOUS DISEASES TO DRIVE MARKET GROWTH
4.2 NORTH AMERICA: CLINICAL TRIAL SUPPLIES INDUSTRY, BY TYPE AND COUNTRY (2022)
FIGURE 17 SMALL-MOLECULE DRUGS ACCOUNTED FOR LARGEST SHARE OF NORTH AMERICAN MARKET IN 2022
4.3 MARKET SHARE, BY SERVICE, 2023 VS. 2028
FIGURE 18 LOGISTICS & DISTRIBUTION SERVICES TO CONTINUE TO DOMINATE MARKET IN 2028
4.4 MARKET SHARE, BY END USER, 2022
FIGURE 19 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES ACCOUNTED FOR LARGEST MARKET SHARE IN 2022
4.5 MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
FIGURE 20 ASIA PACIFIC COUNTRIES TO REGISTER HIGHEST GROWTH RATES FROM 2023 TO 2028
5 MARKET OVERVIEW (Page No. - 65)
5.1 INTRODUCTION
5.2 MARKET DYNAMICS
FIGURE 21 CLINICAL TRIAL SUPPLIES MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
5.3 CLINICAL TRIAL SUPPLIES INDUSTRY: IMPACT ANALYSIS
5.3.1 DRIVERS
5.3.1.1 Increasing R&D investments by biopharma companies
TABLE 4 R&D INVESTMENTS BY MAJOR PLAYERS (2022)
5.3.1.2 Growing number of clinical trials
FIGURE 22 NUMBER OF REGISTERED CLINICAL TRIALS (2011–2022)
FIGURE 23 ONGOING CLINICAL TRIALS, BY PHASE (% SHARE), 2022
5.3.1.3 Growing decentralization of clinical trials
5.3.1.4 Rising prevalence of orphan and rare diseases
FIGURE 24 OVERVIEW OF TOP 20 COMPANIES FOCUSING ON RARE DISEASES (AS OF 2023)
FIGURE 25 PERCENTAGE OF NEW ACTIVE SUBSTANCES LAUNCHED WITH ORPHAN DRUG DESIGNATION, 2012–2022
5.3.1.5 Increasing outsourcing activities in emerging Asian economies
5.3.2 RESTRAINTS
5.3.2.1 High cost of drug development
5.3.3 OPPORTUNITIES
5.3.3.1 Growth opportunities in emerging markets
5.3.3.2 Increased investments in development of biologics and biosimilars
FIGURE 26 TOTAL NUMBER OF NEW DRUGS (NEW CHEMICAL ENTITIES AND BIOLOGICS) APPROVED BY US FDA (2015–2022)
TABLE 5 LIST OF BIOLOGICS APPROVED BY US FDA, 2022
FIGURE 27 BIOSIMILAR APPROVALS AND LAUNCHES (2015–2022)
5.3.3.3 Rising demand for specialized testing services
5.3.4 CHALLENGES
5.3.4.1 High cost of clinical trials
5.4 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES
FIGURE 28 REVENUE SHIFT AND NEW REVENUE POCKETS FOR CLINICAL TRIAL SERVICE PROVIDERS
5.5 PRICING ANALYSIS
5.5.1 INDICATIVE PRICING ANALYSIS OF OVERALL CLINICAL TRIALS, BY PHASE.
TABLE 6 PHASE III ONCOLOGY CLINICAL TRIAL COST (US AND EUROPEAN COUNTRIES)
TABLE 7 PHASE I ONCOLOGY CLINICAL TRIAL COST (SPAIN)
5.6 TECHNOLOGY ANALYSIS
5.6.1 INTERACTIVE RESPONSE TECHNOLOGY (IRT)
5.7 VALUE CHAIN ANALYSIS
FIGURE 29 VALUE CHAIN ANALYSIS: RAW MATERIAL AND MANUFACTURING PHASES CONTRIBUTE MAXIMUM VALUE
5.8 ECOSYSTEM ANALYSIS
5.9 SUPPLY CHAIN ANALYSIS
TABLE 8 MARKET: SUPPLY CHAIN ANALYSIS
5.10 KEY CONFERENCES & EVENTS IN 2023–2024
TABLE 9 MARKET: DETAILED LIST OF CONFERENCES & EVENTS
5.11 REGULATORY ANALYSIS
5.11.1 REGULATORY LANDSCAPE FOR CLINICAL TRIAL SUPPLIES
5.11.1.1 North America
5.11.1.2 Europe
5.11.1.3 Asia Pacific
5.11.1.4 Rest of the World
5.11.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 10 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 11 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 12 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 13 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 14 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
5.12 PORTER’S FIVE FORCES ANALYSIS
TABLE 15 MARKET: PORTER’S FIVE FORCES ANALYSIS
5.12.1 THREAT OF NEW ENTRANTS
5.12.2 THREAT OF SUBSTITUTES
5.12.3 BARGAINING POWER OF SUPPLIERS
5.12.4 BARGAINING POWER OF BUYERS
5.12.5 DEGREE OF COMPETITION
5.13 KEY STAKEHOLDERS & BUYING CRITERIA
5.13.1 KEY STAKEHOLDERS IN BUYING PROCESS
FIGURE 30 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS OF CLINICAL TRIAL SUPPLY SERVICES
5.13.2 BUYING CRITERIA FOR CLINICAL TRIAL SUPPLY SERVICES
FIGURE 31 KEY BUYING CRITERIA FOR END USERS
6 CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE (Page No. - 94)
6.1 INTRODUCTION
TABLE 16 CLINICAL TRIAL SUPPLIES INDUSTRY, BY SERVICE, 2021–2028 (USD MILLION)
6.2 LOGISTICS & DISTRIBUTION
6.2.1 INCREASING COMPLEXITY AND EVOLVING TREATMENT PROTOCOLS TO DRIVE DEMAND FOR LOGISTICS & DISTRIBUTION SERVICES
TABLE 17 MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 18 NORTH AMERICA: MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 19 EUROPE: MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 20 ASIA PACIFIC: MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 21 LATIN AMERICA: MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.3 STORAGE & RETENTION
6.3.1 GROWING DEMAND FOR DIRECT-TO-PATIENT AND DIRECT-TO-SITE SERVICES TO DRIVE GROWTH
TABLE 22 MARKET FOR STORAGE & RETENTION SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 23 NORTH AMERICA: MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 24 EUROPE: MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 25 ASIA PACIFIC: MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 26 LATIN AMERICA: MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.4 PACKAGING, LABELING, AND BLINDING
6.4.1 RISING DEMAND FOR ADVANCED PACKAGING TO DRIVE SEGMENTAL GROWTH
TABLE 27 MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 28 NORTH AMERICA: MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 29 EUROPE: MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 30 ASIA PACIFIC: MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 31 LATIN AMERICA: MARKET FOR LABELING, AND BLINDING SERVICES, COUNTRY, 2021–2028 (USD MILLION)
6.5 MANUFACTURING
6.5.1 GROWING FOCUS OF COMPANIES ON INCREASING MANUFACTURING CAPACITIES TO DRIVE MARKET GROWTH
TABLE 32 MARKET FOR MANUFACTURING SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 33 NORTH AMERICA: MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 34 EUROPE: MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 35 ASIA PACIFIC: MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 36 LATIN AMERICA: MARKET FOR MANUFACTURING SERVICES, COUNTRY, 2021–2028 (USD MILLION)
6.6 COMPARATOR SOURCING
6.6.1 INTENSE MARKET COMPETITION AND RISING NUMBER OF CLINICAL TRIALS IN EMERGING COUNTRIES TO DRIVE GROWTH
TABLE 37 MARKET FOR COMPARATOR SOURCING SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 38 NORTH AMERICA: MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 39 EUROPE: MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 40 ASIA PACIFIC: MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 41 LATIN AMERICA: MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.7 OTHER SERVICES
TABLE 42 MARKET FOR OTHER SERVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 43 NORTH AMERICA: MARKET FOR OTHER SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 44 EUROPE: MARKET FOR OTHER SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 45 ASIA PACIFIC: MARKET FOR OTHER SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 46 LATIN AMERICA: MARKET FOR OTHER SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
7 CLINICAL TRIAL SUPPLIES MARKET, BY PHASE (Page No. - 114)
7.1 INTRODUCTION
TABLE 47 CLINICAL TRIAL SUPPLIES INDUSTRY, BY PHASE, 2021–2028 (USD MILLION)
7.2 PHASE III
7.2.1 LARGE PATIENT POPULATION ASSOCIATED WITH PHASE III TRIALS TO DRIVE DEMAND
TABLE 48 MARKET FOR PHASE III CLINICAL TRIALS, BY REGION, 2021–2028 (USD MILLION)
TABLE 49 NORTH AMERICA: MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 50 EUROPE: MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 51 ASIA PACIFIC: MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 52 LATIN AMERICA: MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
7.3 PHASE II
7.3.1 ABILITY OF PHASE II CLINICAL TRIALS TO PROVIDE CLARITY ON EFFECTIVENESS AND SAFETY OF DRUGS TO DRIVE MARKET GROWTH
TABLE 53 MARKET FOR PHASE II CLINICAL TRIALS, BY REGION, 2021–2028 (USD MILLION)
TABLE 54 NORTH AMERICA: MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 55 EUROPE: MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 56 ASIA PACIFIC: MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 57 LATIN AMERICA: MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
7.4 PHASE I
7.4.1 IMPORTANCE OF QUALITY CLINICAL TRIAL SUPPLIES IN PHASE I CLINICAL TRIALS TO BOOST MARKET
TABLE 58 MARKET FOR PHASE I CLINICAL TRIALS, BY REGION, 2021–2028 (USD MILLION)
TABLE 59 NORTH AMERICA: MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 60 EUROPE: MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 61 ASIA PACIFIC: MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 62 LATIN AMERICA: MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
7.5 PHASE IV
7.5.1 INCREASING NUMBER OF STUDIES UNDER PHASE IV TRIAL TO SUPPORT GROWTH
TABLE 63 MARKET FOR PHASE IV CLINICAL TRIALS, BY REGION, 2021–2028 (USD MILLION)
TABLE 64 NORTH AMERICA: MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 65 EUROPE: MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 66 ASIA PACIFIC: MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 67 LATIN AMERICA: MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021–2028 (USD MILLION)
7.6 BA/BE STUDIES
7.6.1 INCREASING PATENT EXPIRATION TO INCREASE DEMAND FOR BA/BE STUDIES
TABLE 68 MARKET FOR BA/BE STUDIES, BY REGION, 2021–2028 (USD MILLION)
TABLE 69 NORTH AMERICA: MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 70 EUROPE: MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 71 ASIA PACIFIC: MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 72 LATIN AMERICA: MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021–2028 (USD MILLION)
8 CLINICAL TRIAL SUPPLIES MARKET, BY TYPE (Page No. - 130)
8.1 INTRODUCTION
TABLE 73 CLINICAL TRIAL SUPPLIES INDUSTRY, BY TYPE, 2021–2028 (USD MILLION)
8.2 SMALL-MOLECULE DRUGS
8.2.1 LOW COST OF DEVELOPING SMALL-MOLECULE DRUGS TO DRIVE MARKET GROWTH
TABLE 74 MARKET FOR SMALL-MOLECULE DRUGS, BY REGION, 2021–2028 (USD MILLION)
TABLE 75 NORTH AMERICA: MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 76 EUROPE: MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 77 ASIA PACIFIC: MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 78 LATIN AMERICA: MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
8.3 BIOLOGIC DRUGS
8.3.1 RELATIVELY LOW RATE OF ATTRITION OF BIOLOGIC DRUGS COMPARED TO SMALL-MOLECULE DRUGS TO BOOST GROWTH
TABLE 79 MARKET FOR BIOLOGIC DRUGS, BY REGION, 2021–2028 (USD MILLION)
TABLE 80 NORTH AMERICA: MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 81 EUROPE: MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 82 ASIA PACIFIC: MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 83 LATIN AMERICA: MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021–2028 (USD MILLION)
8.4 MEDICAL DEVICES
8.4.1 INCREASING NUMBER OF TRIALS FOR MEDICAL DEVICES TO SUPPORT GROWTH
TABLE 84 MARKET FOR MEDICAL DEVICES, BY REGION, 2021–2028 (USD MILLION)
TABLE 85 NORTH AMERICA: MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 86 EUROPE: MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 87 ASIA PACIFIC: MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 88 LATIN AMERICA: MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021–2028 (USD MILLION)
9 CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA (Page No. - 141)
9.1 INTRODUCTION
TABLE 89 CLINICAL TRIAL SUPPLIES INDUSTRY, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
9.2 ONCOLOGY
9.2.1 GROWING STUDIES ON CANCER THERAPEUTICS TO DRIVE MARKET GROWTH
FIGURE 32 US CANCER INCIDENCE IN MALES, BY TYPE (2022)
FIGURE 33 US CANCER INCIDENCE IN FEMALES, BY TYPE (2022)
FIGURE 34 GLOBAL ONCOLOGY-RELATED CLINICAL TRIALS, 2010–2021 (THOUSAND)
TABLE 90 NEW FDA-APPROVED ONCOLOGY DRUGS (2023)
TABLE 91 MARKET FOR ONCOLOGY, BY REGION, 2021–2028 (USD MILLION)
TABLE 92 NORTH AMERICA: MARKET FOR ONCOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 93 EUROPE: MARKET FOR ONCOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 94 ASIA PACIFIC: MARKET FOR ONCOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 95 LATIN AMERICA: MARKET FOR ONCOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
9.3 INFECTIOUS DISEASES
9.3.1 INCREASING CASES OF INFECTIOUS DISEASES TO ACCELERATE EFFORTS TO DEVELOP NEW DRUGS
TABLE 96 MARKET FOR INFECTIOUS DISEASES, BY REGION, 2021–2028 (USD MILLION)
TABLE 97 NORTH AMERICA: MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 98 EUROPE: MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 99 ASIA PACIFIC: MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 100 LATIN AMERICA: MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
9.4 NEUROLOGY
9.4.1 GROWING INVESTMENTS BY PHARMACEUTICAL COMPANIES FOR NEUROLOGY RESEARCH TO DRIVE GROWTH
TABLE 101 LIST OF PIPELINE DRUGS FOR NEUROLOGICAL DISORDERS (2020)
TABLE 102 MARKET FOR NEUROLOGY, BY REGION, 2021–2028 (USD MILLION)
TABLE 103 NORTH AMERICA: MARKET FOR NEUROLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 104 EUROPE: MARKET FOR NEUROLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 105 ASIA PACIFIC: MARKET FOR NEUROLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 106 LATIN AMERICA: MARKET FOR NEUROLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
9.5 METABOLIC DISORDERS
9.5.1 INCREASING PREVALENCE OF DIABETES AND OTHER METABOLIC DISORDERS TO DRIVE MARKET GROWTH
FIGURE 35 ESTIMATED NUMBER OF INDIVIDUALS WITH DIABETES, 2000–2045 (MILLION)
TABLE 107 MARKET FOR METABOLIC DISORDERS, BY REGION, 2021–2028 (USD MILLION)
TABLE 108 NORTH AMERICA: MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 109 EUROPE: MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 110 ASIA PACIFIC: MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 111 LATIN AMERICA: MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021–2028 (USD MILLION)
9.6 IMMUNOLOGY
9.6.1 CONTINUOUS GROWTH IN CLINICAL STUDIES FOR IMMUNE SYSTEM DISEASES TO FUEL DEMAND FOR CLINICAL TRIAL SUPPLIES
TABLE 112 LIST OF PIPELINE DRUGS FOR IMMUNOLOGICAL DISORDERS (2022)
TABLE 113 MARKET FOR IMMUNOLOGY, BY REGION, 2021–2028 (USD MILLION)
TABLE 114 NORTH AMERICA: MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 115 EUROPE: MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 116 ASIA PACIFIC: MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 117 LATIN AMERICA: MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
9.7 CARDIOLOGY
9.7.1 GROWING INVESTMENTS BY PHARMACEUTICAL COMPANIES FOR CVD RESEARCH TO DRIVE GROWTH
TABLE 118 LIST OF PIPELINE DRUGS FOR CARDIOVASCULAR DISEASES (2023)
TABLE 119 MARKET FOR CARDIOLOGY, BY REGION, 2021–2028 (USD MILLION)
TABLE 120 NORTH AMERICA: MARKET FOR CARDIOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 121 EUROPE: MARKET FOR CARDIOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 122 ASIA PACIFIC: MARKET FOR CARDIOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 123 LATIN AMERICA: MARKET FOR CARDIOLOGY, BY COUNTRY, 2021–2028 (USD MILLION)
9.8 GENETIC & RARE DISEASES
9.8.1 INCREASING R&D FOR DEVELOPMENT OF EFFECTIVE DRUGS TO DRIVE DEMAND
TABLE 124 MARKET FOR GENETIC & RARE DISEASES, BY REGION, 2021–2028 (USD MILLION)
TABLE 125 NORTH AMERICA: MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 126 EUROPE: MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 127 ASIA PACIFIC: MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 128 LATIN AMERICA: MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021–2028 (USD MILLION)
9.9 OTHER THERAPEUTIC AREAS
TABLE 129 LIST OF PIPELINE DRUGS FOR SKIN DISEASES (2022)
TABLE 130 MARKET FOR OTHER THERAPEUTIC AREAS, BY REGION, 2021–2028 (USD MILLION)
TABLE 131 NORTH AMERICA: MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 132 EUROPE: MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 133 ASIA PACIFIC: MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 134 LATIN AMERICA: MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021–2028 (USD MILLION)
10 CLINICAL TRIAL SUPPLIES MARKET, BY END USER (Page No. - 173)
10.1 INTRODUCTION
TABLE 135 CLINICAL TRIAL SUPPLIES INDUSTRY, BY END USER, 2021–2028 (USD MILLION)
10.2 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
10.2.1 INCREASING OUTSOURCING OF CLINICAL TRIAL SUPPLIES BY PHARMA & BIOTECH COMPANIES TO BOOST MARKET
TABLE 136 MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY REGION, 2021–2028 (USD MILLION)
TABLE 137 NORTH AMERICA: MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 138 EUROPE: MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 139 ASIA PACIFIC: MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 140 LATIN AMERICA: MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
10.3 CONTRACT RESEARCH ORGANIZATIONS
10.3.1 INCREASING OUTSOURCING OF CLINICAL TRIALS TO DRIVE ADOPTION OF CLINICAL TRIAL SUPPLIES AMONG CROS
TABLE 141 MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY REGION, 2021–2028 (USD MILLION)
TABLE 142 NORTH AMERICA: MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 143 EUROPE: MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 144 ASIA PACIFIC: MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 145 LATIN AMERICA: MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021–2028 (USD MILLION)
10.4 MEDICAL DEVICE COMPANIES
10.4.1 INCREASING NUMBER OF TRIALS OF MEDICAL DEVICES TO DRIVE MARKET
TABLE 146 MARKET FOR MEDICAL DEVICE COMPANIES, BY REGION, 2021–2028 (USD MILLION)
TABLE 147 NORTH AMERICA: MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 148 EUROPE: MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 149 ASIA PACIFIC: MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 150 LATIN AMERICA: MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021–2028 (USD MILLION)
11 CLINICAL TRIAL SUPPLIES MARKET, BY REGION (Page No. - 183)
11.1 INTRODUCTION
TABLE 151 CLINICAL TRIAL SUPPLIES INDUSTRY, BY REGION, 2021–2028 (USD MILLION)
11.2 NORTH AMERICA
FIGURE 36 NORTH AMERICA: MARKET SNAPSHOT
TABLE 152 NORTH AMERICA: MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 153 NORTH AMERICA: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 154 NORTH AMERICA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 155 NORTH AMERICA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 156 NORTH AMERICA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 157 NORTH AMERICA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.2.1 US
11.2.1.1 Increasing R&D investments to drive market growth in US
TABLE 158 US: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 159 US: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 160 US: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 161 US: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 162 US: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.2.2 CANADA
11.2.2.1 Growing preference of pharmaceutical companies to conduct trials in Canada to favor market growth
TABLE 163 CANADA: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 164 CANADA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 165 CANADA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 166 CANADA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 167 CANADA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.2.3 NORTH AMERICA: RECESSION IMPACT
11.3 EUROPE
TABLE 168 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 169 EUROPE: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 170 EUROPE: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 171 EUROPE: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 172 EUROPE: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 173 EUROPE: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.1 GERMANY
11.3.1.1 Growing number of sponsored trials to boost demand for clinical trial supplies in Germany
TABLE 174 GERMANY: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 175 GERMANY: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 176 GERMANY: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 177 GERMANY: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 178 GERMANY: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.2 UK
11.3.2.1 Government policies leading to costlier R&D and supply chain restrictions post Brexit to limit market growth
TABLE 179 UK: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 180 UK: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 181 UK: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 182 UK: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 183 UK: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.3 FRANCE
11.3.3.1 Presence of leading pharmaceutical companies to support market growth in France
TABLE 184 FRANCE: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 185 FRANCE: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 186 FRANCE: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 187 FRANCE: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 188 FRANCE: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.4 ITALY
11.3.4.1 Increasing number of drug approvals to drive market for clinical trial supplies in Italy
TABLE 189 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 190 ITALY: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 191 ITALY: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 192 ITALY: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 193 ITALY: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.5 SPAIN
11.3.5.1 Increasing biologics production to support market growth in Spain
TABLE 194 SPAIN: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 195 SPAIN: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 196 SPAIN: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 197 SPAIN: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 198 SPAIN: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.6 SWITZERLAND
11.3.6.1 Growing R&D efforts to provide growth opportunities for clinical trial supplies market in Switzerland
TABLE 199 SWITZERLAND: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 200 SWITZERLAND: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 201 SWITZERLAND: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 202 SWITZERLAND: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 203 SWITZERLAND: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.7 NETHERLANDS
11.3.7.1 Increasing pharmaceutical production to boost market in Netherlands
TABLE 204 NETHERLANDS: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 205 NETHERLANDS: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 206 NETHERLANDS: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 207 NETHERLANDS: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 208 NETHERLANDS: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.8 REST OF EUROPE
TABLE 209 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 210 REST OF EUROPE: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 211 REST OF EUROPE: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 212 REST OF EUROPE: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 213 REST OF EUROPE: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.3.9 EUROPE: RECESSION IMPACT
11.4 ASIA PACIFIC
FIGURE 37 ASIA PACIFIC: MARKET SNAPSHOT
TABLE 214 ASIA PACIFIC: MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 215 ASIA PACIFIC: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 216 ASIA PACIFIC: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 217 ASIA PACIFIC: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 218 ASIA PACIFIC: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 219 ASIA PACIFIC: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.1 CHINA
11.4.1.1 Favorable government regulations to boost market growth in China
TABLE 220 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 221 CHINA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 222 CHINA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 223 CHINA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 224 CHINA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.2 JAPAN
11.4.2.1 Government initiatives for drug innovation to support market growth
TABLE 225 JAPAN: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 226 JAPAN: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 227 JAPAN: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 228 JAPAN: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 229 JAPAN: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.3 INDIA
11.4.3.1 Low manufacturing costs and skilled workforce to attract outsourcing and investments to India
TABLE 230 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 231 INDIA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 232 INDIA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 233 INDIA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 234 INDIA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.4 AUSTRALIA
11.4.4.1 Presence of many research institutes conducting clinical trials to boost market growth
TABLE 235 AUSTRALIA: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 236 AUSTRALIA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 237 AUSTRALIA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 238 AUSTRALIA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 239 AUSTRALIA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.5 SOUTH KOREA
11.4.5.1 Government investments to contribute to market growth in South Korea
TABLE 240 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 241 SOUTH KOREA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 242 SOUTH KOREA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 243 SOUTH KOREA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 244 SOUTH KOREA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.6 SINGAPORE
11.4.6.1 Presence of world-class infrastructure to drive market growth in Singapore
TABLE 245 SINGAPORE: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 246 SINGAPORE: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 247 SINGAPORE: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 248 SINGAPORE: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 249 SINGAPORE: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.7 REST OF ASIA PACIFIC
TABLE 250 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 251 REST OF ASIA PACIFIC: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 252 REST OF ASIA PACIFIC: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 253 REST OF ASIA PACIFIC: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 254 REST OF ASIA PACIFIC: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.4.8 ASIA PACIFIC: RECESSION IMPACT
11.5 LATIN AMERICA
TABLE 255 LATIN AMERICA: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 256 LATIN AMERICA: MARKET, BY REGION, 2021–2028 (USD MILLION)
TABLE 257 LATIN AMERICA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 258 LATIN AMERICA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 259 LATIN AMERICA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 260 LATIN AMERICA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.5.1 BRAZIL
11.5.1.1 Growth in pharmaceutical R&D to support market growth
TABLE 261 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 262 BRAZIL: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 263 BRAZIL: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 264 BRAZIL: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 265 BRAZIL: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.5.2 MEXICO
11.5.2.1 Growing number of Phase III clinical trials to drive market growth
TABLE 266 MEXICO: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 267 MEXICO: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 268 MEXICO: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 269 MEXICO: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 270 MEXICO: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.5.3 REST OF LATIN AMERICA
TABLE 271 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 272 REST OF LATIN AMERICA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 273 REST OF LATIN AMERICA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 274 REST OF LATIN AMERICA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 275 REST OF LATIN AMERICA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.5.4 LATIN AMERICA: RECESSION IMPACT
11.6 MIDDLE EAST
11.6.1 GROWING PRESENCE OF PHARMACEUTICAL COMPANIES TO DRIVE MARKET GROWTH
TABLE 276 MIDDLE EAST: MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 277 MIDDLE EAST: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 278 MIDDLE EAST: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 279 MIDDLE EAST: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 280 MIDDLE EAST: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.6.2 MIDDLE EAST: RECESSION IMPACT
11.7 AFRICA
11.7.1 GROWING DEMAND FOR OUTSOURCING SERVICES TO DRIVE MARKET GROWTH
TABLE 281 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021–2028 (USD MILLION)
TABLE 282 AFRICA: MARKET, BY PHASE, 2021–2028 (USD MILLION)
TABLE 283 AFRICA: MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 284 AFRICA: MARKET, BY THERAPEUTIC AREA, 2021–2028 (USD MILLION)
TABLE 285 AFRICA: MARKET, BY END USER, 2021–2028 (USD MILLION)
11.7.2 AFRICA: RECESSION IMPACT
12 COMPETITIVE LANDSCAPE (Page No. - 268)
12.1 INTRODUCTION
12.2 STRATEGIES ADOPTED BY KEY PLAYERS
FIGURE 38 CLINICAL TRIAL SUPPLIES MARKET: STRATEGIES ADOPTED BY KEY PLAYERS
12.3 REVENUE SHARE ANALYSIS
FIGURE 39 REVENUE ANALYSIS OF KEY PLAYERS (2020–2022)
12.4 MARKET SHARE ANALYSIS
FIGURE 40 MARKET SHARE ANALYSIS OF KEY PLAYERS (2022)
TABLE 286 CLINICAL TRIAL SUPPLIES INDUSTRY: DEGREE OF COMPETITION
12.5 COMPANY EVALUATION MATRIX
FIGURE 41 CLINICAL TRIAL SUPPLIES INDUSTRY: COMPANY EVALUATION MATRIX FOR KEY PLAYERS, 2022
12.5.1 STARS
12.5.2 EMERGING LEADERS
12.5.3 PERVASIVE PLAYERS
12.5.4 PARTICIPANTS
12.6 COMPANY FOOTPRINT
12.6.1 SERVICE FOOTPRINT OF TOP 25 COMPANIES
TABLE 287 CLINICAL TRIAL SUPPLIES INDUSTRY: COMPANY SERVICE FOOTPRINT ANALYSIS OF KEY PLAYERS
12.6.2 REGIONAL FOOTPRINT OF TOP 25 COMPANIES
TABLE 288 CLINICAL TRIAL SUPPLIES INDUSTRY: REGIONAL FOOTPRINT ANALYSIS OF KEY PLAYERS
12.7 COMPANY EVALUATION MATRIX: START-UPS/SMES
FIGURE 42 CLINICAL TRIAL SUPPLIES INDUSTRY: COMPANY EVALUATION MATRIX FOR START-UPS/SMES, 2022
12.7.1 PROGRESSIVE COMPANIES
12.7.2 STARTING BLOCKS
12.7.3 RESPONSIVE COMPANIES
12.7.4 DYNAMIC COMPANIES
12.8 COMPETITIVE BENCHMARKING OF START-UP/SME PLAYERS
TABLE 289 CLINICAL TIAL SUPPLIES MARKET: DETAILED LIST OF KEY START-UP/SME PLAYERS
TABLE 290 CLINICAL TRIAL SUPPLIES INDUSTRY: COMPETITIVE BENCHMARKING OF START-UPS/SME PLAYERS
12.9 COMPETITIVE SCENARIO AND TRENDS
12.9.1 SERVICES LAUNCHES & APPROVALS
TABLE 291 CLINICAL TRIAL SUPPLIES INDUSTRY: SERVICE LAUNCHES, JANUARY 2021–AUGUST 2023
12.9.2 DEALS
TABLE 292 CLINICAL TRIAL SUPPLIES INDUSTRY: DEALS, JANUARY 2021–JULY 2023
12.9.3 OTHER DEVELOPMENTS
TABLE 293 CLINICAL TRIAL SUPPLIES MARKET: OTHER DEVELOPMENTS, JANUARY 2021–JULY 2023
13 COMPANY PROFILES (Page No. - 283)
(Business overview, Services offered, Recent Developments, MNM view)*
13.1 KEY MARKET PLAYERS
13.1.1 THERMO FISHER SCIENTIFIC, INC.
TABLE 294 THERMO FISHER SCIENTIFIC: BUSINESS OVERVIEW
FIGURE 43 THERMO FISHER SCIENTIFIC, INC.: COMPANY SNAPSHOT (2022)
13.1.2 CATALENT, INC.
TABLE 295 CATALENT, INC.: BUSINESS OVERVIEW
FIGURE 44 CATALENT, INC.: COMPANY SNAPSHOT (2022)
13.1.3 EUROFINS SCIENTIFIC
TABLE 296 EUROFINS SCIENTIFIC: BUSINESS OVERVIEW
FIGURE 45 EUROFINS SCIENTIFIC: COMPANY SNAPSHOT (2022)
13.1.4 PIRAMAL PHARMA SOLUTIONS
TABLE 297 PIRAMAL PHARMA SOLUTIONS: BUSINESS OVERVIEW
FIGURE 46 PIRAMAL PHARMA SOLUTIONS: COMPANY SNAPSHOT (2022)
13.1.5 PRA HEALTH SCIENCES (A SUBSIDIARY OF ICON PLC)
TABLE 298 PRA HEALTH SCIENCES: BUSINESS OVERVIEW
FIGURE 47 ICON PLC: COMPANY SNAPSHOT (2022)
13.1.6 MARKEN (A SUBSIDIARY OF UPS)
TABLE 299 UPS: BUSINESS OVERVIEW
FIGURE 48 UPS: COMPANY SNAPSHOT (2022)
13.1.7 PAREXEL INTERNATIONAL (MA) CORPORATION
TABLE 300 PAREXEL INTERNATIONAL (MA) CORPORATION: BUSINESS OVERVIEW
13.1.8 BIOCAIR
TABLE 301 BIOCAIR: BUSINESS OVERVIEW
13.1.9 ALMAC GROUP
TABLE 302 ALMAC GROUP: BUSINESS OVERVIEW
13.1.10 SHARP SERVICES, LLC
TABLE 303 SHARP SERVICES, LLC: BUSINESS OVERVIEW
13.1.11 PCI PHARMA SERVICES
TABLE 304 PCI PHARMA SERVICES: BUSINESS OVERVIEW
13.1.12 NUVISAN
TABLE 305 NUVISAN: BUSINESS OVERVIEW
13.1.13 LONZA GROUP
TABLE 306 LONZA GROUP: BUSINESS OVERVIEW
FIGURE 49 LONZA GROUP: COMPANY SNAPSHOT (2022)
13.2 OTHER PLAYERS
13.2.1 OCT GROUP LLC
13.2.2 COREX LOGISTICS LIMITED
13.2.3 ACNOS PHARMA GMBH
13.2.4 CLINICAL SERVICES INTERNATIONAL
13.2.5 KLIFO
13.2.6 CLINIGEN LIMITED
13.2.7 ANCILLARE LP
13.2.8 N-SIDE
13.2.9 ADALLEN PHARMA
13.2.10 DURBIN
13.2.11 RECIPHARM AB
13.2.12 IPS PHARMA
*Details on Business overview, Services offered, Recent Developments, MNM view might not be captured in case of unlisted companies.
14 APPENDIX (Page No. - 333)
14.1 DISCUSSION GUIDE
14.2 KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL
14.3 CUSTOMIZATION OPTIONS
14.4 RELATED REPORTS
14.5 AUTHOR DETAILS







 Generating Response ...
Generating Response ...







Growth opportunities and latent adjacency in Clinical Trial Supplies Market
What are the major growth strategies prevalent in the Clinical Trial Supplies Market?
Which factors are influencing the global growth of Clinical Trial Supplies Market?
Which are the different geographies covered in the global study of Clinical Trial Supplies Market?