TABLE OF CONTENTS
1 INTRODUCTION (Page No. - 52)
1.1 STUDY OBJECTIVES
1.2 MARKET DEFINITION
1.2.1 INCLUSIONS & EXCLUSIONS
1.3 MARKET SCOPE
1.3.1 MARKETS COVERED
1.3.2 REGIONAL SEGMENTATION
1.4 YEARS CONSIDERED
1.5 CURRENCY
1.6 LIMITATIONS
1.6.1 SCOPE-RELATED LIMITATIONS
1.6.2 METHODOLOGY-RELATED LIMITATIONS
1.7 STAKEHOLDERS
1.8 SUMMARY OF CHANGES
1.9 RECESSION IMPACT
2 RESEARCH METHODOLOGY (Page No. - 58)
2.1 RESEARCH APPROACH
2.2 RESEARCH METHODOLOGY DESIGN
FIGURE 1 MEDICAL DEVICE CONNECTIVITY MARKET: RESEARCH DESIGN
2.2.1 SECONDARY RESEARCH
2.2.1.1 Key data from secondary sources
2.2.2 PRIMARY DATA
2.2.2.1 Primary sources
2.2.2.2 Key data from primary sources
2.2.2.3 Insights from primary experts
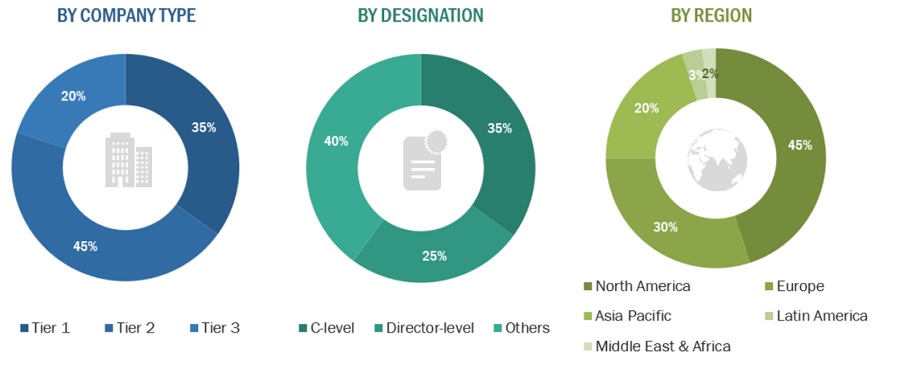
2.2.2.4 Breakdown of primary interviews: supply-side and demand-side participants
2.2.2.5 Breakdown of primary interviews: by company type, designation, and region
2.3 MARKET SIZE ESTIMATION
FIGURE 2 SUPPLY-SIDE MARKET SIZE ESTIMATION: REVENUE SHARE ANALYSIS
FIGURE 3 REVENUE SHARE ANALYSIS ILLUSTRATION
FIGURE 4 TOP-DOWN APPROACH
FIGURE 5 CAGR PROJECTIONS FROM ANALYSIS OF DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES IN MEDICAL DEVICE CONNECTIVITY MARKET
FIGURE 6 CAGR PROJECTIONS: SUPPLY-SIDE ANALYSIS
2.4 MARKET BREAKDOWN & DATA TRIANGULATION
FIGURE 7 DATA TRIANGULATION METHODOLOGY
2.5 MARKET SHARE ESTIMATION
2.6 STUDY ASSUMPTIONS
2.7 LIMITATIONS
2.7.1 METHODOLOGY-RELATED LIMITATIONS
2.7.2 SCOPE-RELATED LIMITATIONS
2.8 RISK ASSESSMENT
TABLE 1 RISK ASSESSMENT: MEDICAL DEVICE CONNECTIVITY MARKET
3 EXECUTIVE SUMMARY (Page No. - 72)
FIGURE 8 MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2023 VS. 2028 (USD MILLION)
FIGURE 9 MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2023 VS. 2028 (USD MILLION)
FIGURE 10 MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2023 VS. 2028 (USD MILLION)
FIGURE 11 MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2023 VS. 2028 (USD MILLION)
FIGURE 12 GEOGRAPHICAL SNAPSHOT OF MEDICAL DEVICE CONNECTIVITY MARKET
4 PREMIUM INSIGHTS (Page No. - 76)
4.1 MEDICAL DEVICE CONNECTIVITY MARKET OVERVIEW
FIGURE 13 GROWING NEED FOR CONTINUOUS PATIENT MONITORING TO DRIVE MARKET GROWTH
4.2 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE (2022)
FIGURE 1 MEDICAL DEVICE CONNECTIVITY SERVICES ACCOUNTED FOR LARGEST SHARE OF ASIA PACIFIC MARKET IN 2022
4.3 MEDICAL DEVICE CONNECTIVITY MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
FIGURE 2 MEXICO TO REGISTER HIGHEST REVENUE GROWTH DURING FORECAST PERIOD
4.4 MEDICAL DEVICE CONNECTIVITY MARKET, BY REGION (2021–2028)
FIGURE 3 NORTH AMERICA TO CONTINUE TO DOMINATE MEDICAL DEVICE CONNECTIVITY MARKET DURING FORECAST PERIOD
4.5 MEDICAL DEVICE CONNECTIVITY MARKET: DEVELOPED VS. EMERGING ECONOMIES
FIGURE 4 EMERGING ECONOMIES TO REGISTER HIGHER GROWTH RATES DURING FORECAST PERIOD
5 MARKET OVERVIEW (Page No. - 80)
5.1 INTRODUCTION
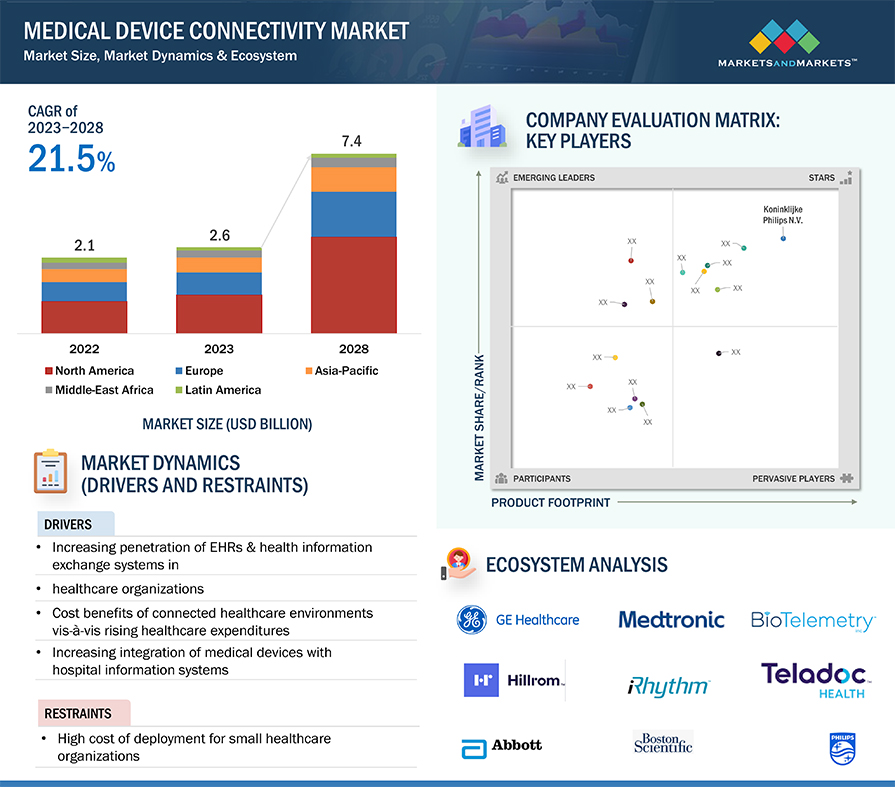
5.2 MARKET DYNAMICS
FIGURE 5 MEDICAL DEVICE CONNECTIVITY MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
5.2.1 DRIVERS
5.2.1.1 Increasing penetration of EHRs & health information exchange systems in healthcare organizations
5.2.1.2 Cost benefits of connected healthcare environments vis-à-vis rising healthcare expenditures
FIGURE 6 CURRENT EXPENDITURE ON HEALTH (2019)
5.2.1.3 Increasing integration of medical devices with hospital information systems
5.2.1.4 Favorable government support and initiatives
5.2.1.5 Growing focus on care quality and patient safety
5.2.1.6 Medical device connectivity aids in data analytics
5.2.1.7 Growing investments to improve healthcare infrastructure
5.2.2 RESTRAINTS
5.2.2.1 High cost of deployment for small healthcare organizations
5.2.2.2 Reluctance of medical professionals to adopt advanced healthcare IT solutions
5.2.3 OPPORTUNITIES
5.2.3.1 Increasing consolidation in healthcare industry
FIGURE 7 US: HOSPITAL MERGERS & ACQUISITIONS, 2015–2019
5.2.3.2 Growing inclination towards home healthcare
5.2.3.3 Increasing investments in telehealth & RPM
5.2.4 CHALLENGES
5.2.4.1 Integration between various hospital information systems and medical devices
5.2.4.2 Data security concerns
5.2.4.3 Lack of skilled healthcare IT professionals
5.3 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES
FIGURE 8 REVENUE SHIFTS AND NEW REVENUE POCKETS FOR MEDICAL DEVICE CONNECTIVITY MARKET
5.4 INDUSTRY TRENDS
5.4.1 SHIFT TOWARDS CLOUD-BASED SOLUTIONS
5.4.2 INTEGRATION OF MEDICAL DEVICE DATA INTO CARE DELIVERY PROCESSES
5.4.3 GROWING NEED FOR SEMANTIC INTEROPERABILITY THROUGH MEDICAL DEVICE CONNECTIVITY SOLUTIONS
5.4.4 TRANSITION OF POC DIAGNOSTICS FROM HOSPITALS TO HOME SETTINGS
5.4.5 GROWING ADOPTION OF TELEHEALTH SOLUTIONS
5.4.6 SHIFT FROM PAYMENT-FOR-PERFORMANCE TO PAYMENT-FOR-OUTCOMES MODELS
5.4.7 M2M COMMUNICATIONS & ARTIFICIAL INTELLIGENCE BOOM IN HEALTHCARE
5.4.8 LOW-POWER CONNECTIVITY SOLUTIONS & ADVANCEMENTS IN SOFTWARE ARCHITECTURE
5.4.9 SUBSTITUTABLE MEDICAL APPLICATIONS, REUSABLE TECHNOLOGIES (SMART) ON FAST HEALTHCARE INTEROPERABILITY RESOURCES (FHIR)
5.5 VALUE CHAIN ANALYSIS
FIGURE 9 MEDICAL DEVICE CONNECTIVITY MARKET: VALUE CHAIN ANALYSIS
5.6 ECOSYSTEM ANALYSIS
FIGURE 10 MEDICAL DEVICE CONNECTIVITY MARKET: ECOSYSTEM ANALYSIS
5.7 TECHNOLOGY ANALYSIS
5.7.1 IOMT (INTERNET OF MEDICAL THINGS)
5.7.2 EDGE COMPUTING
5.8 TARIFF & REGULATORY ANALYSIS
5.8.1 TARIFF FOR MEDICAL DEVICE CONNECTIVITY SOLUTIONS
TABLE 2 LIST OF REGULATORY STANDARDS FOR MEDICAL DEVICE CONNECTIVITY SOLUTIONS
5.8.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
5.8.2.1 North America
5.8.2.1.1 US
TABLE 3 TESTS AND CERTIFICATION REQUIREMENTS FOR WIRELESSLY CONNECTED MEDICAL DEVICES
5.8.2.1.2 Canada
5.8.2.2 Europe
5.8.2.3 Asia Pacific
5.8.2.3.1 Japan
TABLE 4 CLASSIFICATION OF MEDICAL DEVICES IN JAPAN
5.8.2.3.2 China
5.8.2.3.3 India
5.9 PORTER’S FIVE FORCES ANALYSIS
TABLE 5 MEDICAL DEVICE CONNECTIVITY MARKET: PORTER’S FIVE FORCES ANALYSIS
5.9.1 INTENSITY OF COMPETITIVE RIVALRY
5.9.2 BARGAINING POWER OF SUPPLIERS
5.9.3 BARGAINING POWER OF BUYERS
5.9.4 THREAT OF NEW ENTRANTS
5.9.5 THREAT OF SUBSTITUTES
5.10 PATENT ANALYSIS
5.10.1 PATENT PUBLICATION TRENDS
FIGURE 11 PATENT PUBLICATION TRENDS: JANUARY 2011−NOVEMBER 2023
5.10.2 JURISDICTION ANALYSIS: TOP APPLICANTS (COUNTRIES) FOR MEDICAL DEVICE CONNECTIVITY PATENTS (2011−2023)
TABLE 6 JURISDICTION ANALYSIS: TOP APPLICANT COUNTRIES FOR MEDICAL DEVICE CONNECTIVITY PATENTS
FIGURE 12 NUMBER OF PATENT APPLICATION, BY JURISDICTION
5.10.3 MEDICAL DEVICE CONNECTIVITY MARKET: TOP APPLICANTS
FIGURE 13 TOP APPLICANTS FOR MEDICAL DEVICE CONNECTIVITY PATENTS (2011–2023)
5.10.4 MEDICAL DEVICE CONNECTIVITY MARKET: LIST OF PATENTS/PATENT APPLICATIONS
5.11 TRADE ANALYSIS
5.11.1 TRADE ANALYSIS FOR MEDICAL DEVICE CONNECTIVITY SOLUTIONS
FIGURE 14 IMPORTS OF PRODUCTS UNDER HSN CODE: 85176290, BY COUNTRY, 2022 (USD THOUSAND)
FIGURE 15 EXPORTS OF PRODUCTS UNDER HSN CODE: 85176290, BY COUNTRY, 2022 (USD THOUSAND)
5.12 CASE STUDY ANALYSIS
TABLE 7 CASE STUDY 1: PROVIDED A SOLUTION TO HOSPITALS TO OVERCOME NETWORK INFRASTRUCTURE CONNECTION CHALLENGES AND IMPROVE PATIENT CARE AND TRANSMISSION OF CRITICAL DEVICE OPERATIONAL DATA
TABLE 8 CASE STUDY 2: MEDICAL DEVICE INTEGRATION
5.13 KEY CONFERENCES & EVENTS
TABLE 9 LIST OF KEY CONFERENCES & EVENTS (2023–2024)
5.14 KEY STAKEHOLDERS & BUYING CRITERIA
5.14.1 KEY STAKEHOLDERS IN BUYING PROCESS
TABLE 10 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR MEDICAL DEVICE CONNECTIVITY
TABLE 11 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE
5.14.2 BUYING CRITERIA
FIGURE 16 KEY BUYING CRITERIA FOR MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE
TABLE 12 KEY BUYING CRITERIA FOR MEDICAL DEVICE CONNECTIVITY MARKET
5.15 PRICING ANALYSIS
TABLE 13 AVERAGE SELLING PRICE FOR MEDICAL DEVICE CONNECTIVITY SOLUTIONS, BY KEY PLAYER (2022)
TABLE 14 REGIONAL PRICING ANALYSIS OF REMOTE PATIENT MONITORING DEVICES, BY TYPE, 2021 (USD)
5.16 UNMET NEEDS ANALYSIS
TABLE 15 UNMET NEEDS IN MEDICAL DEVICE CONNECTIVITY MARKET
5.16.1 MEDICAL DEVICE CONNECTIVITY MARKET: END-USER EXPECTATIONS
TABLE 16 END-USER EXPECTATIONS IN MEDICAL DEVICE CONNECTIVITY MARKET
5.17 REIMBURSEMENT SCENARIO
TABLE 17 CPT CODES AND REIMBURSEMENT ANALYSIS OF US MEDICAL DEVICE CONNECTIVITY MARKET
5.18 INTEROPERABILITY STANDARDS
TABLE 18 KEY STANDARDS FOR INTEROPERABILITY IN MEDICAL DEVICE CONNECTIVITY MARKET
TABLE 19 QUALITATIVE ANALYSIS OF WIDELY USED DATA TRANSMISSION STANDARDS IN DIGITAL HEALTH SPECTRUM
5.19 MEDICAL DEVICE CONNECTIVITY BUSINESS MODELS
5.19.1 DEVICE MANUFACTURERS LICENSING MODELS
5.19.2 PAY-PER-USE OR PAY-PER-DEVICE MODELS
5.19.3 PLATFORM AS A SERVICE (PAAS) MODELS
5.20 MEDICAL DEVICE CONNECTIVITY INVESTMENT LANDSCAPE
FIGURE 17 INVESTOR DEALS AND FUNDING IN MEDICAL DEVICE CONNECTIVITY SOARED IN 2022
FIGURE 18 MOST VALUED MEDICAL DEVICE CONNECTIVITY FIRMS IN 2022 (USD BILLION)
6 MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE (Page No. - 129)
6.1 INTRODUCTION
TABLE 20 MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
6.2 MEDICAL DEVICE CONNECTIVITY SOLUTIONS
TABLE 21 MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 22 MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.2.1 MEDICAL DEVICE INTEGRATION SOLUTIONS
6.2.1.1 Increasing demand for EHR implementation and need for accurate patient information to drive adoption
TABLE 23 MEDICAL DEVICE INTEGRATION SOLUTIONS OFFERED BY KEY MARKET PLAYERS
TABLE 24 MEDICAL DEVICE INTEGRATION SOLUTIONS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.2.2 TELEMETRY SYSTEMS
6.2.2.1 Rising demand for remote patient monitoring to drive adoption of telemetry systems
TABLE 25 TELEMETRY SYSTEMS OFFERED BY KEY MARKET PLAYERS
TABLE 26 TELEMETRY SYSTEMS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.2.3 CONNECTIVITY HUBS
6.2.3.1 Growing demand for remote patient monitoring and need to simplify workflows in healthcare settings to support growth
TABLE 27 CONNECTIVITY HUBS OFFERED BY KEY MARKET PLAYERS
TABLE 28 CONNECTIVITY HUBS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.2.4 INTERFACE DEVICES
6.2.4.1 Rising importance of health information exchange and EHR implementation to fuel market growth
TABLE 29 INTERFACE DEVICES OFFERED BY KEY MARKET PLAYERS
TABLE 30 INTERFACE DEVICES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.3 PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES
TABLE 31 PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 32 PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.3.1 PATIENT MONITORING DEVICES
6.3.1.1 Increasing need for remote patient monitoring to drive growth
TABLE 33 PATIENT MONITORING DEVICES OFFERED BY KEY MARKET PLAYERS
TABLE 34 PATIENT MONITORING DEVICES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.3.2 CRITICAL CARE INSTRUMENTS
6.3.2.1 Rising demand for constant monitoring to drive adoption of critical care instruments
TABLE 35 CRITICAL CARE INSTRUMENTS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.3.3 IMAGING SYSTEMS
6.3.3.1 Growing demand for connected imaging systems to favor growth
TABLE 36 IMAGING SYSTEMS MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.3.4 OTHER PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES
TABLE 37 OTHER PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.4 MEDICAL DEVICE CONNECTIVITY SERVICES
TABLE 38 MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 39 MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
6.4.1 SUPPORT & MAINTENANCE SERVICES
6.4.1.1 Growing trend of outsourcing of healthcare IT support and services to drive market
TABLE 40 SUPPORT & MAINTENANCE SERVICES OFFERED BY KEY MARKET PLAYERS
TABLE 41 MEDICAL DEVICE CONNECTIVITY MARKET FOR SUPPORT & MAINTENANCE SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.4.2 IMPLEMENTATION & INTEGRATION SERVICES
6.4.2.1 Requirement of software interoperability and need to comply with regulatory standards to boost market growth
TABLE 42 IMPLEMENTATION & INTEGRATION SERVICES OFFERED BY KEY MARKET PLAYERS
TABLE 43 MEDICAL DEVICE CONNECTIVITY MARKET FOR IMPLEMENTATION & INTEGRATION SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.4.3 TRAINING SERVICES
6.4.3.1 Need to increase adoption of healthcare IT solutions to support market growth
TABLE 44 TRAINING SERVICES OFFERED BY KEY MARKET PLAYERS
TABLE 45 MEDICAL DEVICE CONNECTIVITY MARKET FOR TRAINING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
6.4.4 CONSULTING SERVICES
6.4.4.1 Need to guide healthcare providers in implementation of medical device connectivity solutions to boost market
TABLE 46 CONSULTING SERVICES OFFERED BY KEY MARKET PLAYERS
TABLE 47 MEDICAL DEVICE CONNECTIVITY MARKET FOR CONSULTING SERVICES, BY COUNTRY, 2021–2028 (USD MILLION)
7 MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY (Page No. - 154)
7.1 INTRODUCTION
TABLE 48 MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
7.2 WIRELESS TECHNOLOGIES
TABLE 49 MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 50 MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.2.1 CLOUD-BASED TECHNOLOGIES
7.2.1.1 Seamless real-time communication and data exchange between medical devices to propel market growth
TABLE 51 MEDICAL DEVICE CONNECTIVITY MARKET FOR CLOUD-BASED TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.2.2 BLUETOOTH TECHNOLOGIES
7.2.2.1 Seamless connection between wireless devices to foster adoption
TABLE 52 MEDICAL DEVICE CONNECTIVITY MARKET FOR BLUETOOTH TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.2.3 OTHER WIRELESS TECHNOLOGIES
TABLE 53 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER WIRELESS TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.3 WIRED TECHNOLOGIES
TABLE 54 MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 55 MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.3.1 LAN TECHNOLOGIES
7.3.1.1 5G networks to propel adoption of LAN connectivity
TABLE 56 MEDICAL DEVICE CONNECTIVITY MARKET FOR LAN TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.3.2 WAN TECHNOLOGIES
7.3.2.1 Ability to provide greater flexibility to healthcare providers to support market growth
TABLE 57 MEDICAL DEVICE CONNECTIVITY MARKET FOR WAN TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
7.4 HYBRID TECHNOLOGIES
7.4.1 INCREASING NEED FOR MOBILITY AND SAFETY ALONG WITH DATA SECURITY TO DRIVE DEMAND FOR HYBRID TECHNOLOGIES
TABLE 58 MEDICAL DEVICE CONNECTIVITY MARKET FOR HYBRID TECHNOLOGIES, BY COUNTRY, 2021–2028 (USD MILLION)
8 MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION (Page No. - 168)
8.1 INTRODUCTION
TABLE 59 MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
8.2 VITAL SIGNS & PATIENT MONITORS
TABLE 60 MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 61 MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.1 BLOOD GLUCOSE MONITORS
8.2.1.1 Growing demand for continuous glucose monitoring to drive market
TABLE 62 BLOOD GLUCOSE MONITORS OFFERED BY KEY MARKET PLAYERS
TABLE 63 MEDICAL DEVICE CONNECTIVITY MARKET FOR BLOOD GLUCOSE MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.2 ECG/HEART RATE MONITORS
8.2.2.1 Rising incidence of CVDs to drive market growth
TABLE 64 ECG/HEART RATE MONITORS OFFERED BY KEY MARKET PLAYERS
TABLE 65 MEDICAL DEVICE CONNECTIVITY MARKET FOR ECG/HEART RATE MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.3 BLOOD PRESSURE MONITORS
8.2.3.1 Growing prevalence of hypertension and obesity to fuel market
TABLE 66 BLOOD PRESSURE MONITORS OFFERED BY KEY MARKET PLAYERS
TABLE 67 MEDICAL DEVICE CONNECTIVITY MARKET FOR BLOOD PRESSURE MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.4 MULTIPARAMETER MONITORS
8.2.4.1 Rising preference for home care to contribute to demand
TABLE 68 MULTIPARAMETER MONITORS OFFERED BY KEY MARKET PLAYERS
TABLE 69 MEDICAL DEVICE CONNECTIVITY MARKET FOR MULTIPARAMETER MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.5 OXIMETERS
8.2.5.1 Increasing focus on self-monitoring to drive market
TABLE 70 OXIMETERS OFFERED BY KEY MARKET PLAYERS
TABLE 71 MEDICAL DEVICE CONNECTIVITY MARKET FOR OXIMETERS, BY COUNTRY, 2021–2028 (USD MILLION)
8.2.6 PATIENT MONITORS
8.2.6.1 Growing adoption of patient monitors to boost market
TABLE 72 PATIENT MONITORS OFFERED BY KEY MARKET PLAYERS
TABLE 73 MEDICAL DEVICE CONNECTIVITY MARKET FOR PATIENT MONITORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.3 ANESTHESIA MACHINES
8.3.1 OPERATIONAL ADVANTAGES OF ANESTHESIA MACHINES TO PROPEL GROWTH
TABLE 74 ANESTHESIA MACHINES OFFERED BY KEY MARKET PLAYERS
TABLE 75 MEDICAL DEVICE CONNECTIVITY MARKET FOR ANESTHESIA MACHINES, BY COUNTRY, 2021–2028 (USD MILLION)
8.4 VENTILATORS
8.4.1 HIGH DEMAND FOR INTENSIVE CARE TO DRIVE MARKET GROWTH
TABLE 76 VENTILATORS OFFERED BY KEY MARKET PLAYERS
TABLE 77 MEDICAL DEVICE CONNECTIVITY MARKET FOR VENTILATORS, BY COUNTRY, 2021–2028 (USD MILLION)
8.5 INFUSION PUMPS
8.5.1 INCREASED ADOPTION OF PUMP-EMR INTEROPERABILITY DURING IV THERAPY TO DRIVE GROWTH
TABLE 78 DEVELOPMENTS IN SMART PUMP-EMR INTEROPERABILITY
TABLE 79 MEDICAL DEVICE CONNECTIVITY MARKET FOR INFUSION PUMPS, BY COUNTRY, 2021–2028 (USD MILLION)
8.6 IMAGING SYSTEMS
TABLE 80 IMAGING SYSTEMS OFFERED BY KEY MARKET PLAYERS
TABLE 81 MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 82 MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY COUNTRY, 2021–2028 (USD MILLION)
8.6.1 MRI & CT MACHINES
8.6.1.1 Reduced examination time and improved image quality to drive market
TABLE 83 MEDICAL DEVICE CONNECTIVITY MARKET FOR MRI & CT MACHINES, BY COUNTRY, 2021–2028 (USD MILLION)
8.6.2 ULTRASOUND SYSTEMS
8.6.2.1 Growing trend of point-of-care ultrasound to drive demand for connectivity solutions
TABLE 84 MEDICAL DEVICE CONNECTIVITY MARKET FOR ULTRASOUND SYSTEMS, BY COUNTRY, 2021–2028 (USD MILLION)
8.6.3 X-RAY MACHINES
8.6.3.1 High-throughput capabilities of X-ray machines to propel market
TABLE 85 MEDICAL DEVICE CONNECTIVITY MARKET FOR X-RAY MACHINES, BY COUNTRY, 2021–2028 (USD MILLION)
8.6.4 OTHER IMAGING SYSTEMS
TABLE 86 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER IMAGING SYSTEMS, BY COUNTRY, 2021–2028 (USD MILLION)
8.7 RESPIRATORY DEVICES
8.7.1 RISING PREVALENCE OF RESPIRATORY DISORDERS TO DRIVE DEMAND
TABLE 87 RESPIRATORY DEVICES OFFERED BY KEY MARKET PLAYERS
TABLE 88 MEDICAL DEVICE CONNECTIVITY MARKET FOR RESPIRATORY DEVICES, BY COUNTRY, 2021–2028 (USD MILLION)
8.8 OTHER APPLICATIONS
TABLE 89 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 90 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY COUNTRY, 2021–2028 (USD MILLION)
8.8.1 IMPLANTS
8.8.1.1 Ease of remote monitoring to drive demand
TABLE 91 IMPLANTABLE CARDIAC DEVICES OFFERED BY KEY MARKET PLAYERS
TABLE 92 MEDICAL DEVICE CONNECTIVITY MARKET FOR IMPLANTS, BY COUNTRY, 2021–2028 (USD MILLION)
8.8.2 PACEMAKERS
8.8.2.1 Rising number of cardiac patients to drive demand
TABLE 93 PACEMAKERS OFFERED BY KEY MARKET PLAYERS
TABLE 94 MEDICAL DEVICE CONNECTIVITY MARKET FOR PACEMAKERS, BY COUNTRY, 2021–2028 (USD MILLION)
8.8.3 OTHER MEDICAL DEVICE CONNECTIVITY SOLUTIONS
TABLE 95 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER MEDICAL DEVICE CONNECTIVITY SOLUTIONS, BY COUNTRY, 2021–2028 (USD MILLION)
9 MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER (Page No. - 206)
9.1 INTRODUCTION
TABLE 96 MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
9.2 HOSPITALS & SURGICAL CENTERS
9.2.1 RISING PURCHASING POWER OF HOSPITALS TO DRIVE ADOPTION OF MEDICAL DEVICE CONNECTIVITY SOLUTIONS
TABLE 97 MEDICAL DEVICE CONNECTIVITY MARKET FOR HOSPITALS & SURGICAL CENTERS, BY COUNTRY, 2021–2028 (USD MILLION)
9.3 MATERNITY & FERTILITY CARE CENTERS
9.3.1 ADOPTION OF ELECTRONIC FETAL MONITORING DEVICES AND TELEMETRY SOLUTIONS TO DRIVE GROWTH
TABLE 98 MEDICAL DEVICE CONNECTIVITY MARKET FOR MATERNITY & FERTILITY CARE CENTERS, BY COUNTRY, 2021–2028 (USD MILLION)
9.4 TRAUMA & EMERGENCY CARE UNITS
9.4.1 ADOPTION OF TELEPRESENCE AND TELEMEDICINE IN TRAUMA AND EMERGENCY CARE MANAGEMENT TO SUPPORT MARKET GROWTH
TABLE 99 MEDICAL DEVICE CONNECTIVITY MARKET FOR TRAUMA & EMERGENCY CARE UNITS, BY COUNTRY, 2021–2028 (USD MILLION)
9.5 TERTIARY CARE CENTERS
9.5.1 ADVANCEMENTS IN TECHNOLOGY AND GROWING SHIFT TOWARDS HOME CARE FOR ELDERLY TO FUEL GROWTH
TABLE 100 MEDICAL DEVICE CONNECTIVITY MARKET FOR TERTIARY CARE CENTERS, BY COUNTRY, 2021–2028 (USD MILLION)
9.6 HOME CARE SETTINGS
9.6.1 GROWING BENEFITS AND ADOPTION OF TELEHEALTH SOLUTIONS TO BOOST MARKET GROWTH
TABLE 101 MEDICAL DEVICE CONNECTIVITY MARKET FOR HOME CARE SETTINGS, BY COUNTRY, 2021–2028 (USD MILLION)
9.7 AMBULATORY & OPD SETTINGS
9.7.1 RISING PATIENT VOLUME AT AMBULATORY CARE CENTERS TO FAVOR MARKET GROWTH
TABLE 102 MEDICAL DEVICE CONNECTIVITY MARKET FOR AMBULATORY & OPD SETTINGS, BY COUNTRY, 2021–2028 (USD MILLION)
9.8 DIAGNOSTIC & IMAGING CENTERS
9.8.1 GROWING VOLUME OF MEDICAL DATA TO BOOST MARKET
TABLE 103 MEDICAL DEVICE CONNECTIVITY MARKET FOR DIAGNOSTIC & IMAGING CENTERS, BY COUNTRY, 2021–2028 (USD MILLION)
9.9 OTHER END USERS
TABLE 104 MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER END USERS, BY COUNTRY, 2021–2028 (USD MILLION)
10 MEDICAL DEVICE CONNECTIVITY MARKET, BY REGION (Page No. - 220)
10.1 INTRODUCTION
FIGURE 19 MEDICAL DEVICE CONNECTIVITY MARKET: GEOGRAPHIC SNAPSHOT (2022)
TABLE 105 MEDICAL DEVICE CONNECTIVITY MARKET, BY REGION, 2021–2028 (USD MILLION)
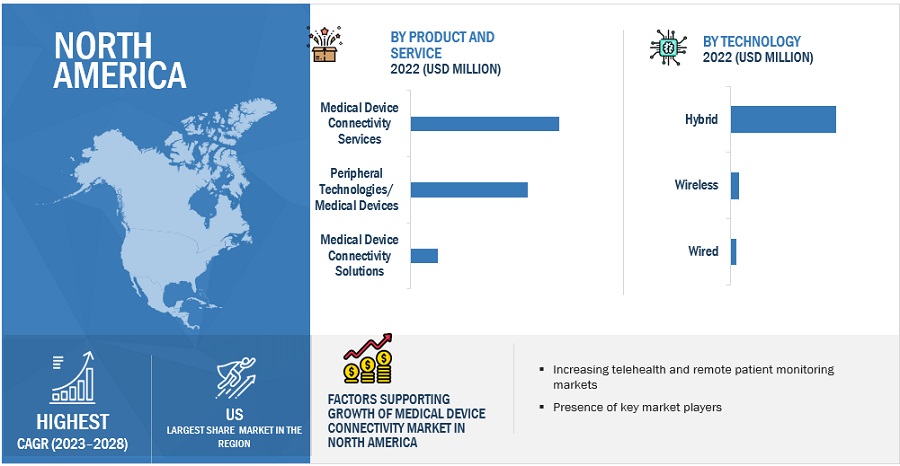
10.2 NORTH AMERICA
FIGURE 20 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET SNAPSHOT
TABLE 106 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 107 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 108 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 109 NORTH AMERICA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 110 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 111 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 112 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 113 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 114 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 115 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 116 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 117 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 118 NORTH AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.2.1 US
10.2.1.1 Advanced medical infrastructure and increasing healthcare expenditure to drive market growth
TABLE 119 US: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 120 US: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 121 US: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 122 US: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 123 US: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 124 US: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 125 US: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 126 US: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 127 US: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 128 US: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 129 US: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 130 US: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.2.2 CANADA
10.2.2.1 Growing adoption of technologically advanced HCIT solutions to propel market growth in Canada
TABLE 131 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 132 CANADA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 133 CANADA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 134 CANADA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 135 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 136 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 137 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 138 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 139 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 140 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 141 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 142 CANADA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.2.3 NORTH AMERICA: RECESSION IMPACT ANALYSIS
10.3 EUROPE
TABLE 143 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 144 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 145 EUROPE: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 146 EUROPE: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 147 EUROPE: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 148 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 149 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 150 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 151 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 152 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 153 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 154 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 155 EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.1 GERMANY
10.3.1.1 Increasing health expenditure and growing network of hospitals to drive adoption of connectivity solutions
TABLE 156 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 157 GERMANY: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 158 GERMANY: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 159 GERMANY: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 160 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 161 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 162 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 163 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 164 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 165 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 166 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 167 GERMANY: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.2 FRANCE
10.3.2.1 High healthcare spending and favorable government initiatives to drive demand for medical device connectivity solutions
TABLE 168 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 169 FRANCE: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 170 FRANCE: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 171 FRANCE: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 172 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 173 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 174 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 175 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 176 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 177 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 178 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 179 FRANCE: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.3 UK
10.3.3.1 Rising geriatric population & government initiatives to drive adoption of medical device connectivity solutions in the UK
TABLE 180 UK: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 181 UK: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 182 UK: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 183 UK: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 184 UK: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 185 UK: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 186 UK: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 187 UK: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 188 UK: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 189 UK: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 190 UK: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 191 UK: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.4 ITALY
10.3.4.1 Increasing adoption of eHealth technologies to propel market growth
TABLE 192 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 193 ITALY: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 194 ITALY: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 195 ITALY: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 196 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 197 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 198 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 199 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 200 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 201 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 202 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 203 ITALY: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.5 SPAIN
10.3.5.1 Growing focus on improving healthcare infrastructure to support market growth
TABLE 204 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 205 SPAIN: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 206 SPAIN: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 207 SPAIN: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 208 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 209 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 210 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 211 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 212 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 213 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 214 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 215 SPAIN: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.6 REST OF EUROPE
TABLE 216 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 217 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 218 REST OF EUROPE: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 219 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 220 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 221 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 222 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 223 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 224 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 225 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 226 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 227 REST OF EUROPE: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.3.7 EUROPE: RECESSION IMPACT ANALYSIS
10.4 ASIA PACIFIC
FIGURE 21 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET SNAPSHOT
TABLE 228 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 229 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 230 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 231 ASIA PACIFIC: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 232 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 233 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 234 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 235 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 236 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 237 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 238 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 239 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 240 ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.4.1 JAPAN
10.4.1.1 Growing aging population in country to drive market growth
TABLE 241 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 242 JAPAN: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 243 JAPAN: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 244 JAPAN: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 245 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 246 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 247 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 248 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 249 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 250 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 251 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 252 JAPAN: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.4.2 CHINA
10.4.2.1 Large patient pool to drive adoption of medical device connectivity solutions
TABLE 253 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 254 CHINA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 255 CHINA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 256 CHINA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 257 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 258 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 259 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 260 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 261 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 262 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 263 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 264 CHINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.4.3 INDIA
10.4.3.1 Growing investments in country’s healthcare infrastructure to support market growth
TABLE 265 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 266 INDIA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 267 INDIA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 268 INDIA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 269 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 270 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 271 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 272 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 273 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 274 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 275 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 276 INDIA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.4.4 REST OF ASIA PACIFIC
TABLE 277 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 278 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 279 REST OF ASIA PACIFIC: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 280 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 281 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 282 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 283 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 284 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 285 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 286 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 287 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 288 REST OF ASIA PACIFIC: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.4.5 ASIA PACIFIC: RECESSION IMPACT ANALYSIS
10.5 LATIN AMERICA
TABLE 289 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 290 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 291 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 292 LATIN AMERICA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 293 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 294 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 295 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 296 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 297 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 298 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 299 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 300 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 301 LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.5.1 BRAZIL
10.5.1.1 Growing emphasis on reducing healthcare costs to favor market growth
TABLE 302 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 303 BRAZIL: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 304 BRAZIL: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 305 BRAZIL: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 306 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 307 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 308 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 309 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 310 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 311 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 312 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 313 BRAZIL: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.5.2 MEXICO
10.5.2.1 Increasing demand and adoption of healthcare IT solutions to drive growth
TABLE 314 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 315 MEXICO: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 316 MEXICO: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 317 MEXICO: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 318 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 319 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 320 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 321 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 322 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 323 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 324 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 325 MEXICO: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.5.3 ARGENTINA
10.5.3.1 Rising demand for home care to drive growth of market in Argentina
TABLE 326 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 327 ARGENTINA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 328 ARGENTINA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 329 ARGENTINA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 330 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 331 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 332 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 333 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 334 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 335 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 336 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 337 ARGENTINA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.5.4 REST OF LATIN AMERICA (ROLA)
TABLE 338 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 339 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 340 REST OF LATIN AMERICA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 341 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 342 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 343 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 344 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 345 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 346 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 347 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 348 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 349 REST OF LATIN AMERICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.5.5 LATIN AMERICA: RECESSION IMPACT ANALYSIS
10.6 MIDDLE EAST & AFRICA
TABLE 350 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY COUNTRY, 2021–2028 (USD MILLION)
TABLE 351 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 352 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 353 MIDDLE EAST & AFRICA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 354 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 355 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 356 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 357 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 358 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 359 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 360 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 361 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 362 MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.6.1 GCC COUNTRIES
10.6.1.1 Rising focus on expanding and improving present health infrastructure to boost market growth
TABLE 363 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 364 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 365 GCC COUNTRIES: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 366 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 367 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 368 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 369 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 370 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 371 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 372 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 373 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 374 GCC COUNTRIES: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.6.2 REST OF MIDDLE EAST & AFRICA
TABLE 375 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY PRODUCT & SERVICE, 2021–2028 (USD MILLION)
TABLE 376 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY SOLUTIONS MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 377 REST OF MIDDLE EAST & AFRICA: PERIPHERAL TECHNOLOGIES/MEDICAL DEVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 378 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY SERVICES MARKET, BY TYPE, 2021–2028 (USD MILLION)
TABLE 379 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY TECHNOLOGY, 2021–2028 (USD MILLION)
TABLE 380 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRELESS TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 381 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR WIRED TECHNOLOGIES, BY TYPE, 2021–2028 (USD MILLION)
TABLE 382 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY APPLICATION, 2021–2028 (USD MILLION)
TABLE 383 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR VITAL SIGNS & PATIENT MONITORS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 384 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR IMAGING SYSTEMS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 385 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET FOR OTHER APPLICATIONS, BY TYPE, 2021–2028 (USD MILLION)
TABLE 386 REST OF MIDDLE EAST & AFRICA: MEDICAL DEVICE CONNECTIVITY MARKET, BY END USER, 2021–2028 (USD MILLION)
10.6.3 MIDDLE EAST & AFRICA: RECESSION IMPACT ANALYSIS
11 COMPETITIVE LANDSCAPE (Page No. - 378)
11.1 OVERVIEW
11.2 STRATEGIES ADOPTED BY KEY PLAYERS/RIGHT TO WIN
11.2.1 OVERVIEW OF STRATEGIES ADOPTED BY PLAYERS IN MEDICAL DEVICE CONNECTIVITY MARKET
11.3 REVENUE ANALYSIS
FIGURE 22 REVENUE ANALYSIS OF KEY PLAYERS, 2018–2022
11.4 MARKET SHARE ANALYSIS
FIGURE 23 MEDICAL DEVICE CONNECTIVITY MARKET: MARKET SHARE ANALYSIS (2022)
TABLE 387 MEDICAL DEVICE CONNECTIVITY MARKET: DEGREE OF COMPETITION
FIGURE 24 MEDICAL DEVICE CONNECTIVITY MARKET: RANKING OF KEY COMPANIES (2022)
11.5 COMPANY EVALUATION MATRIX
11.5.1 STARS
11.5.2 EMERGING LEADERS
11.5.3 PERVASIVE PLAYERS
11.5.4 PARTICIPANTS
FIGURE 25 MEDICAL DEVICE CONNECTIVITY MARKET: COMPANY EVALUATION MATRIX, 2022
11.5.5 COMPANY FOOTPRINT ANALYSIS
TABLE 388 OVERALL COMPANY FOOTPRINT
TABLE 389 PRODUCT & SERVICE FOOTPRINT
TABLE 390 TECHNOLOGY FOOTPRINT
TABLE 391 APPLICATION FOOTPRINT
TABLE 392 END-USER FOOTPRINT
TABLE 393 REGIONAL FOOTPRINT
11.6 START-UP/SME EVALUATION MATRIX
11.6.1 PROGRESSIVE COMPANIES
11.6.2 DYNAMIC COMPANIES
11.6.3 RESPONSIVE COMPANIES
11.6.4 STARTING BLOCKS
FIGURE 26 MEDICAL DEVICE CONNECTIVITY MARKET: STARTUP/SME EVALUATION MATRIX (2022)
11.6.5 COMPETITIVE BENCHMARKING
TABLE 394 MEDICAL DEVICE CONNECTIVITY MARKET: DETAILED LIST OF KEY START-UPS/SMES
11.7 COMPETITIVE SCENARIO
11.7.1 PRODUCT/SERVICE LAUNCHES & APPROVALS
TABLE 395 PRODUCT/SERVICE LAUNCHES & APPROVALS, 2020–2023
11.7.2 DEALS
TABLE 396 DEALS, 2020–2023
11.7.3 OTHER DEVELOPMENTS
TABLE 397 OTHER DEVELOPMENTS, 2020–2023
12 COMPANY PROFILES (Page No. - 399)
12.1 KEY PLAYERS
(Business Overview, Products/Services/Solutions Offered, MnM View, Key Strengths and Right to Win, Strategic Choices Made, Weaknesses and Competitive Threats, Recent Developments)*
12.1.1 KONINKLIJKE PHILIPS N.V.
TABLE 398 KONINKLIJKE PHILIPS N.V.: BUSINESS OVERVIEW
FIGURE 27 KONINKLIJKE PHILIPS N.V.: COMPANY SNAPSHOT (2022)
12.1.2 ORACLE CORPORATION
TABLE 399 ORACLE CORPORATION: BUSINESS OVERVIEW
FIGURE 28 ORACLE CORPORATION: COMPANY SNAPSHOT (2023)
12.1.3 GE HEALTHCARE
TABLE 400 GE HEALTHCARE: BUSINESS OVERVIEW
FIGURE 29 GE HEALTHCARE: COMPANY SNAPSHOT (2022)
12.1.4 MASIMO CORPORATION
TABLE 401 MASIMO CORPORATION: BUSINESS OVERVIEW
FIGURE 30 MASIMO CORPORATION: COMPANY SNAPSHOT (2021)
12.1.5 MEDTRONIC PLC
TABLE 402 MEDTRONIC PLC: BUSINESS OVERVIEW
FIGURE 31 MEDTRONIC PLC: COMPANY SNAPSHOT (2022)
12.1.6 CISCO SYSTEMS, INC.
TABLE 403 CISCO SYSTEMS, INC.: BUSINESS OVERVIEW
FIGURE 32 CISCO SYSTEMS, INC.: COMPANY SNAPSHOT (2023)
12.1.7 IHEALTH LABS INC.
TABLE 404 IHEALTH LABS: BUSINESS OVERVIEW
12.1.8 LANTRONIX, INC.
TABLE 405 LANTRONIX, INC.: BUSINESS OVERVIEW
FIGURE 33 LANTRONIX, INC.: COMPANY SNAPSHOT (2023)
12.1.9 INFOSYS LIMITED
TABLE 406 INFOSYS LIMITED: BUSINESS OVERVIEW
FIGURE 34 INFOSYS LIMITED: COMPANY SNAPSHOT (2022)
12.1.10 S3 CONNECTED HEALTH
TABLE 407 S3 CONNECTED HEALTH: BUSINESS OVERVIEW
12.1.11 SPECTRUM MEDICAL LTD.
TABLE 408 SPECTRUM MEDICAL LTD.: BUSINESS OVERVIEW
12.1.12 IATRIC SYSTEMS, INC.
TABLE 409 IATRIC SYSTEMS INC.: BUSINESS OVERVIEW
12.1.13 BAXTER INTERNATIONAL INC.
TABLE 410 BAXTER INTERNATIONAL INC.: BUSINESS OVERVIEW
FIGURE 35 BAXTER INTERNATIONAL INC.: COMPANY SNAPSHOT (2022)
12.1.14 SILEX TECHNOLOGY, INC.
TABLE 411 SILEX TECHNOLOGY, INC.: BUSINESS OVERVIEW
12.1.15 BRIDGE-TECH MEDICAL, INC.
TABLE 412 BRIDGE-TECH MEDICAL, INC.: BUSINESS OVERVIEW
12.1.16 MEDICOLLECTOR LLC
TABLE 413 MEDICOLLECTOR LLC: BUSINESS OVERVIEW
12.1.17 TE CONNECTIVITY LTD.
TABLE 414 TE CONNECTIVITY LTD.: BUSINESS OVERVIEW
FIGURE 36 TE CONNECTIVITY LTD.: COMPANY SNAPSHOT (2022)
12.1.18 TRUE PROCESS
TABLE 415 TRUE PROCESS: BUSINESS OVERVIEW
12.1.19 DIGI INTERNATIONAL INC.
TABLE 416 DIGI INTERNATIONAL INC.: BUSINESS OVERVIEW
FIGURE 37 DIGI INTERNATIONAL INC.: COMPANY SNAPSHOT (2022)
12.1.20 STRYKER CORPORATION
TABLE 417 STRYKER CORPORATION: BUSINESS OVERVIEW
FIGURE 38 STRYKER CORPORATION: COMPANY SNAPSHOT (2022)
*Business Overview, Products/Services/Solutions Offered, MnM View, Key Strengths and Right to Win, Strategic Choices Made, Weaknesses and Competitive Threats, Recent Developments might not be captured in case of unlisted companies.
12.2 OTHER PLAYERS
12.2.1 DRÄGERWERK AG & CO. KGAA
12.2.2 HONEYWELL INTERNATIONAL INC.
12.2.3 ASCOM HOLDINGS AG
12.2.4 WIPRO LIMITED
12.2.5 OSI SYSTEMS, INC.
13 APPENDIX (Page No. - 461)
13.1 DISCUSSION GUIDE
13.2 KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL
13.3 CUSTOMIZATION OPTIONS
13.4 RELATED REPORTS
13.5 AUTHOR DETAILS






 Generating Response ...
Generating Response ...







Growth opportunities and latent adjacency in Medical Device Connectivity Market