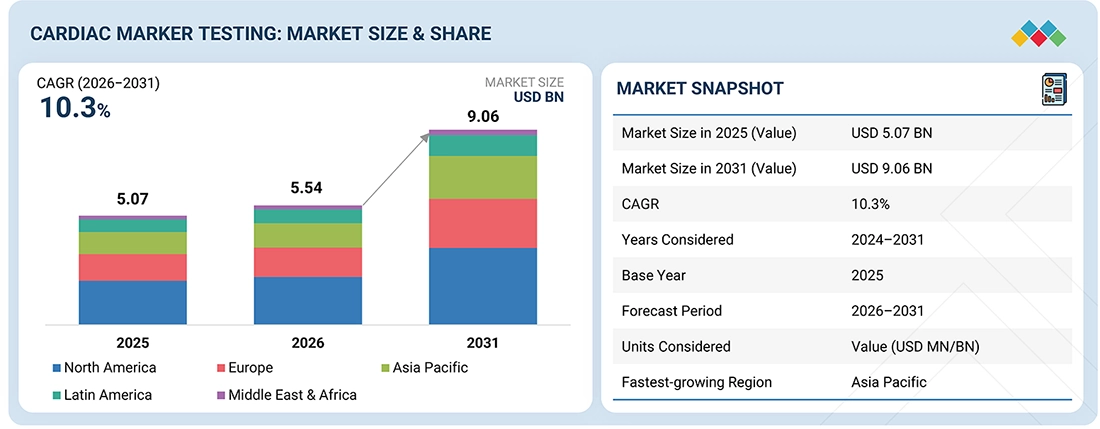
4
MARKET OVERVIEW
Cardiac biomarker innovations drive growth amid aging populations and regulatory challenges.
50
4.2.1.1
RISING PREVALENCE OF CARDIOVASCULAR DISEASES
4.2.1.2
INCREASING GERIATRIC POPULATION AND SUBSEQUENT RISE IN HEART CONDITIONS WORLDWIDE
4.2.1.3
RISING CLINICAL STUDIES FOR IDENTIFICATION OF NOVEL CARDIAC BIOMARKERS
4.2.1.4
GROWING DEMAND FOR POC TESTING
4.2.2.1
TECHNICAL ISSUES RELATED TO SAMPLE COLLECTION AND STORAGE
4.2.2.2
UNFAVORABLE REGULATORY PROCESSES AND LIMITED REIMBURSEMENT STRUCTURE
4.2.3.1
ONGOING RESEARCH ON NOVEL CARDIAC BIOMARKERS FOR CARDIOVASCULAR DISEASES
4.2.4.1
CHALLENGES ASSOCIATED WITH BIOMARKER ASSAY VALIDATION
4.3
UNMET NEEDS & WHITE SPACES
4.3.1
UNMET NEEDS IN CARDIAC MARKER TESTING MARKET
4.3.2
WHITE SPACE OPPORTUNITIES
4.4
INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
4.4.1
INTERCONNECTED MARKETS
4.4.2
CROSS-SECTOR OPPORTUNITIES
4.5
STRATEGIC MOVES BY TIER 1/2/3 PLAYERS
4.5.1
KEY MOVES & STRATEGIC FOCUS
5
INDUSTRY TRENDS
Discover how economic forces and pricing dynamics shape the cardiac marker testing market landscape.
59
5.1
PORTER’S FIVE FORCES ANALYSIS
5.1.1
THREAT OF NEW ENTRANTS
5.1.2
THREAT OF SUBSTITUTES
5.1.3
BARGAINING POWER OF BUYERS
5.1.4
BARGAINING POWER OF SUPPLIERS
5.1.5
INTENSITY OF COMPETITIVE RIVALRY
5.2
MACROECONOMIC INDICATORS
5.2.2
GDP TRENDS & FORECAST
5.3
SUPPLY CHAIN ANALYSIS
5.6.1
AVERAGE SELLING PRICE OF CARDIAC MARKER TESTING PRODUCTS, BY TYPE, 2024–2026
5.6.2
AVERAGE SELLING PRICE OF CARDIAC MARKER TESTING PRODUCTS, BY KEY PLAYER, 2024–2026
5.6.3
AVERAGE SELLING PRICE OF CARDIAC MARKER TESTING PRODUCTS, BY REGION, 2024–2026
5.7.1
IMPORT SCENARIO (HS CODE 3822)
5.7.2
EXPORT SCENARIO (HS CODE 3822)
5.8
KEY CONFERENCES & EVENTS, 2026–2027
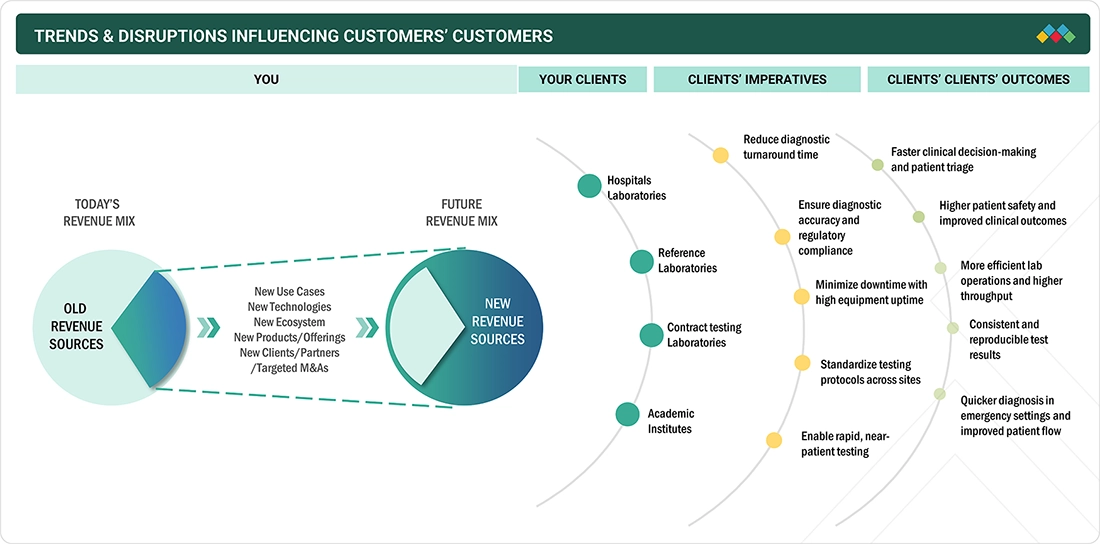
5.9
TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES
5.10
INVESTMENT & FUNDING SCENARIO
5.11.1
CASE STUDY 1: IMPLEMENTATION OF HIGH-SENSITIVITY TROPONIN (HS-CTN) ASSAYS FOR RAPID MYOCARDIAL INFARCTION DIAGNOSIS
5.11.2
CASE STUDY 2: USE OF BNP/NT-PROBNP TESTING FOR HEART FAILURE DIAGNOSIS AND MANAGEMENT
5.11.3
CASE STUDY 3: USE OF HIGH-SENSITIVITY TROPONIN FOR LONG-TERM CARDIOVASCULAR RISK PREDICTION IN GENERAL POPULATION
5.12
IMPACT OF 2025 US TARIFFS ON CARDIAC MARKER TESTING MARKET
5.12.3
PRICE IMPACT ANALYSIS
5.12.5
IMPACT ON END-USE INDUSTRIES
5.12.5.1
HOSPITAL LABORATORIES
5.12.5.2
REFERENCE LABORATORIES
5.12.5.3
CONTRACT TESTING LABORATORIES
5.12.5.4
ACADEMIC INSTITUTES
6
TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
AI and emerging tech revolutionize cardiac marker testing, unlocking new market potentials and innovations.
79
6.1
KEY EMERGING TECHNOLOGIES
6.1.1
CHEMILUMINESCENCE IMMUNOASSAYS (CLIA)
6.1.2
IMMUNOFLUORESCENCE ASSAYS (IFA)
6.1.3
ENZYME-LINKED IMMUNOSORBENT ASSAYS (ELISA)
6.1.4
LATERAL FLOW ASSAYS
6.2
COMPLEMENTARY TECHNOLOGIES
6.2.1
POC IMMUNOANALYZERS
6.2.2
DIGITAL IMMUNOASSAY PLATFORMS
6.3
TECHNOLOGY/PRODUCT ROADMAP
6.4.1
LIST OF MAJOR PATENTS
6.4.2
LIST OF KEY PATENTS
6.6
IMPACT OF AI/GENERATIVE AI ON CARDIAC MARKER TESTING MARKET
6.6.2
TOP USE CASES & MARKET POTENTIAL
6.6.3
KEY COMPANIES IMPLEMENTING AI
7
SUSTAINABILITY & REGULATORY LANDSCAPE
Navigate complex regulations with eco-friendly cardiac marker testing strategies for global compliance.
87
7.1
REGIONAL REGULATIONS & COMPLIANCE
7.1.1
REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
7.2
SUSTAINABILITY INITIATIVES
7.2.1
ENVIRONMENTAL IMPACT AND ECO-FRIENDLY INITIATIVES IN CARDIAC MARKER TESTING PRODUCTS
7.2.1.1
ECO-FRIENDLY INITIATIVES
7.3
SUSTAINABILITY IMPACT & REGULATORY POLICY INITIATIVES
8
CUSTOMER LANDSCAPE & BUYER BEHAVIOR
Unlock growth by navigating buyer dynamics and addressing unmet industry needs for enhanced profitability.
91
8.1
DECISION-MAKING PROCESS
8.2
BUYER STAKEHOLDERS & BUYING EVALUATION CRITERIA
8.2.1
KEY STAKEHOLDERS IN BUYING PROCESS
8.3
ADOPTION BARRIERS & INTERNAL CHALLENGES
8.4
UNMET NEEDS IN VARIOUS END-USE INDUSTRIES
8.5.3
MARGIN OPPORTUNITIES IN KEY APPLICATIONS
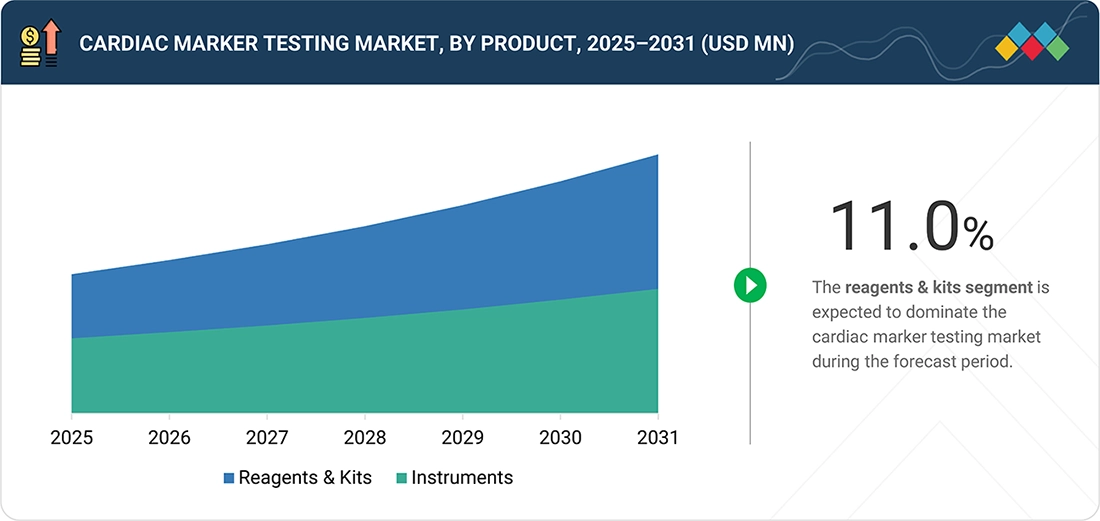
9
CARDIAC MARKER TESTING MARKET, BY PRODUCT
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 13 Data Tables
97
9.2.1
RISING VOLUME OF DIAGNOSTIC TESTS TO DRIVE DEMAND FOR REAGENTS & KITS
9.3.1
EXPANDING DIAGNOSTIC INFRASTRUCTURE TO DRIVE MARKET GROWTH
10
CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 32 Data Tables
105
10.2.1.1
HIGH SENSITIVITY, WIDE LINEAR RANGE, AND GOOD SPECIFICITY TO AID MARKET GROWTH
10.2.2
IMMUNOFLUORESCENCE
10.2.2.1
ADVANCEMENTS IN FLUORESCENCE TECHNOLOGIES TO DRIVE IMMUNOFLUORESCENCE ADOPTION
10.2.3
LATERAL FLOW IMMUNOASSAYS
10.2.3.1
INCREASING USE IN POINT-OF-CARE SETTINGS TO PROPEL ADOPTION
10.2.4.1
LONG SHELF LIFE AND EASE OF USE TO PROPEL GROWTH
10.3.1
COST-EFFECTIVENESS AND HIGH-THROUGHPUT TESTING TO SUSTAIN GROWTH IN CLINICAL CHEMISTRY MARKET
11
CARDIAC MARKER TESTING MARKET, BY INDICATION
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 26 Data Tables
123
11.2
MYOCARDIAL INFARCTION
11.2.1
RISING MYOCARDIAL INFARCTION BURDEN AND NEED FOR EARLY DIAGNOSIS TO DRIVE CARDIAC MARKER TESTING DEMAND
11.3
CONGESTIVE HEART FAILURE
11.3.1
INCREASING USE OF MULTIMARKER TESTING TO SUPPORT MARKET GROWTH
11.4
ACUTE CORONARY SYNDROME
11.4.1
INCREASING INCIDENCE OF RISK FACTORS ASSOCIATED WITH ACS TO DRIVE ADOPTION OF CARDIAC MARKERS
11.5.1
HIGH PREVALENCE OF ATHEROSCLEROSIS TO DRIVE MARKET
11.6.1
INCREASE IN CARDIAC ISCHEMIA TO BOOST DEMAND FOR CARDIAC MARKER TESTS
12
CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 36 Data Tables
138
12.2.1
HIGH SENSITIVITY AND SPECIFICITY IN DETECTION OF CVD TO DRIVE MARKET
12.3
CREATININE KINASE-MB
12.3.1
POOR SPECIFICITY AND BRIEF ELEVATION DURATION TO HAMPER MARKET GROWTH
12.4
BRAIN NATRIURETIC PEPTIDE
12.4.1
USE OF BNP WITH OTHER CARDIAC MARKERS TO ENHANCE UTILIZATION
12.5.1
GROWING USE TO ASSESS PATIENTS WITH CHEST PAIN IN EMERGENCY DEPARTMENT SETTINGS TO BOOST GROWTH
12.6.1
RISK STRATIFICATION AND LONG-TERM CARDIOVASCULAR RISK ASSESSMENT TO DRIVE MARKET GROWTH
12.7
HIGH-SENSITIVITY C-REACTIVE PROTEIN
12.7.1
INCREASING INCIDENCE OF CARDIOVASCULAR DISEASES TO PROPEL ADOPTION
12.8
OTHER CARDIAC BIOMARKERS
13
CARDIAC MARKER TESTING MARKET VOLUME ANALYSIS, BY BIOMARKER TYPE (NUMBER OF TESTS)
Market Size & Growth Rate Forecast Analysis to 2031 in Units | 12 Data Tables
159
14
CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 11 Data Tables
165
14.2.1
HIGH-SENSITIVITY, AUTOMATION, AND FLEXIBLE PROCUREMENT MODELS TO DRIVE LAB-BASED TESTING DOMINANCE
14.3.1
RISING DEMAND FOR DECENTRALIZED HEALTHCARE SERVICES TO DRIVE MARKET
15
CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 11 Data Tables
172
15.2.1
HIGH ACCURACY AND OPERATIONAL SIMPLICITY TO SUSTAIN SINGLEPLEX ASSAY DOMINANCE
15.3.1
COMPREHENSIVE MULTI-BIOMARKER DETECTION AND RAPID DIAGNOSIS TO DRIVE MULTIPLEX ASSAY GROWTH
16
CARDIAC MARKER TESTING MARKET, BY END USER
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 32 Data Tables
179
16.2
LABORATORY TESTING FACILITIES
16.2.1
HOSPITAL LABORATORIES
16.2.1.1
GROWING FOCUS ON HEALTHCARE SYSTEM TO STIMULATE GROWTH
16.2.2
REFERENCE LABORATORIES
16.2.2.1
RISING OUTSOURCING AND ADVANCED TESTING CAPABILITIES TO POSITION REFERENCE LABORATORIES AS KEY GROWTH DRIVERS
16.2.3
CONTRACT TESTING LABORATORIES
16.2.3.1
OUTSOURCED EXPERTISE AND SCALABLE TESTING CAPABILITIES TO BOOST ROLE OF CONTRACT LABORATORIES
16.3.1
CONTINUOUS RESEARCH & DEVELOPMENT OF NOVEL BIOMARKERS TO DRIVE MARKET
16.4
POINT-OF-CARE TESTING
16.4.1
EARLY DETECTION AND LOWER TURNAROUND TIME TO BOOST MARKET
17
CARDIAC MARKER TESTING MARKET, BY REGION
Comprehensive coverage of 7 Regions with country-level deep-dive of 14 Countries | 203 Data Tables.
197
17.2.1.1
GROWING SUPPORT FROM NIH FOR DEVELOPMENT OF NOVEL CARDIAC BIOMARKERS TO DRIVE MARKET
17.2.2.1
GOVERNMENT INITIATIVES TO SUPPORT MARKET GROWTH
17.3.1.1
STRINGENT REGULATIONS FOR FOOD, PHARMACEUTICAL, AND MEDICAL DEVICE INDUSTRIES TO FUEL GROWTH
17.3.2.1
INCREASING CARDIOVASCULAR PATIENT VOLUME TO SPUR GROWTH
17.3.3.1
HIGHLY ADVANCED AND WELL-ESTABLISHED HEALTHCARE SYSTEM TO DRIVE DEMAND FOR MARKET
17.3.4.1
GROWING GERIATRIC POPULATION AND IMPROVED LIFE EXPECTANCY TO SUPPORT MARKET GROWTH
17.3.5.1
GOVERNMENT EFFORTS TO IMPROVE HEALTH INFRASTRUCTURE TO FUEL DEMAND
17.4.1.1
GROWING GERIATRIC POPULATION AND INCREASING INCIDENCE OF CHRONIC DISEASES TO DRIVE MARKET
17.4.2.1
RISING NUMBER OF HOSPITALS AND INCREASING HEALTHCARE EXPENDITURE TO BOOST ADOPTION
17.4.3.1
INFRASTRUCTURAL IMPROVEMENTS TO SUPPORT GROWTH
17.4.4.1
HIGHER GOVERNMENT HEALTHCARE EXPENDITURE TO SUPPORT MARKET GROWTH
17.4.5.1
COUNTRY’S FAST-AGING DEMOGRAPHIC TO DRIVE MARKET
17.4.6
REST OF ASIA PACIFIC
17.5.1.1
RISING GOVERNMENT SUPPORT TO BOOST ADOPTION OF MARKET
17.5.2.1
RISING GERIATRIC POPULATION TO SUPPORT MARKET GROWTH
17.5.3
REST OF LATIN AMERICA
17.6
MIDDLE EAST & AFRICA
18
COMPETITIVE LANDSCAPE
Uncover strategic market positions and financial metrics of key players in cardiac marker testing.
285
18.2
KEY PLAYER STRATEGIES/RIGHT TO WIN
18.2.1
OVERVIEW OF STRATEGIES DEPLOYED BY PLAYERS IN CARDIAC MARKER TESTING MARKET
18.3
REVENUE ANALYSIS, 2023–2025
18.4
MARKET SHARE ANALYSIS, 2025
18.4.1
GLOBAL MARKET SHARE ANALYSIS, 2025
18.4.2
US MARKET SHARE ANALYSIS, 2025
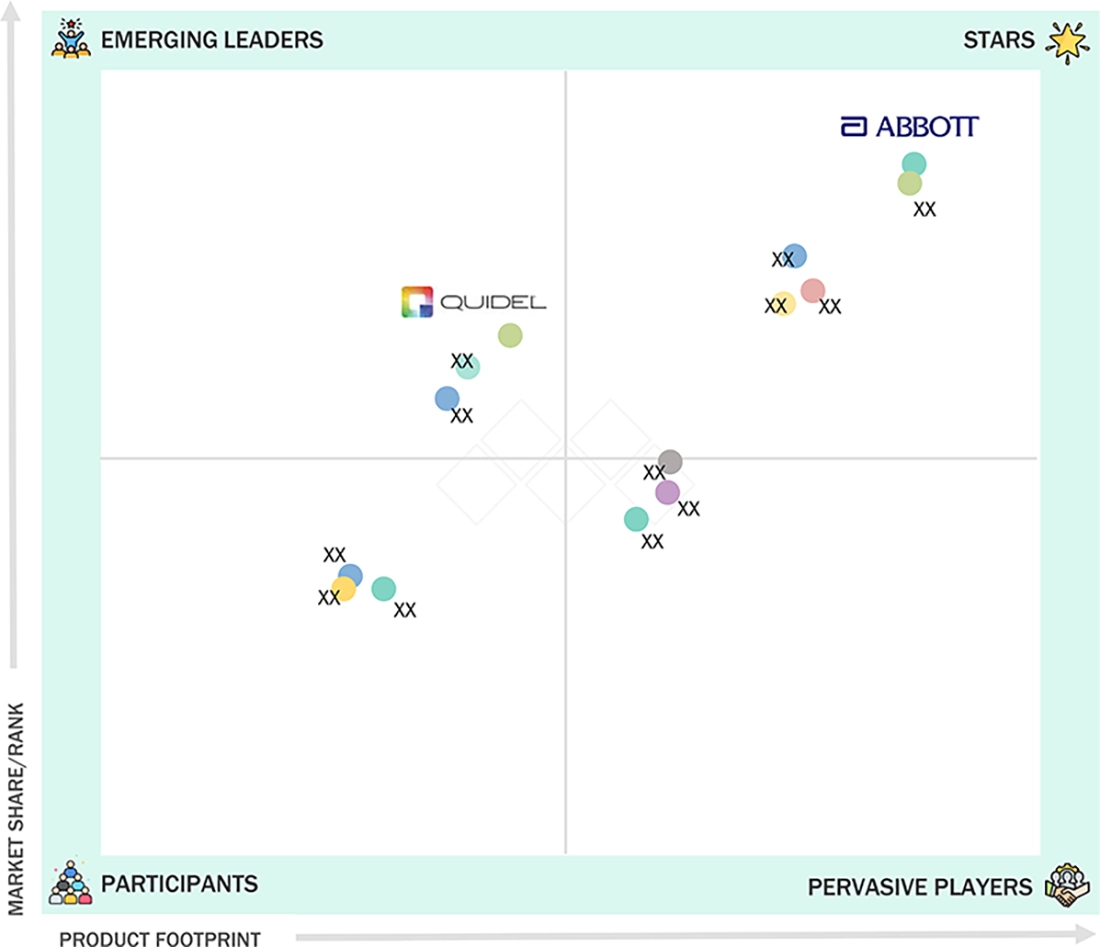
18.5
COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
18.5.5
COMPANY FOOTPRINT: KEY PLAYERS, 2025
18.5.5.1
COMPANY FOOTPRINT
18.5.5.2
REGION FOOTPRINT
18.5.5.3
PRODUCT FOOTPRINT
18.5.5.4
TECHNOLOGY FOOTPRINT
18.5.5.5
POINT OF TESTING FOOTPRINT
18.6
COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
18.6.1
PROGRESSIVE COMPANIES
18.6.2
RESPONSIVE COMPANIES
18.6.5
COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
18.6.5.1
DETAILED LIST OF KEY STARTUPS/SMES
18.6.5.2
COMPETITIVE BENCHMARKING OF STARTUPS/SMES (1/2)
18.6.5.3
COMPETITIVE BENCHMARKING OF STARTUPS/SMES (2/2)
18.7
COMPANY VALUATION & FINANCIAL METRICS
18.8
BRAND/PRODUCT COMPARISON
18.9
COMPETITIVE SCENARIO
19
COMPANY PROFILES
In-depth Company Profiles of Leading Market Players with detailed Business Overview, Product and Service Portfolio, Recent Developments, and Unique Analyst Perspective (MnM View)
302
19.1.1
F. HOFFMANN-LA ROCHE AG
19.1.1.1
BUSINESS OVERVIEW
19.1.1.2
PRODUCTS OFFERED
19.1.1.3
RECENT DEVELOPMENTS
19.1.1.3.1
PRODUCT APPROVALS
19.1.1.4.2
STRATEGIC CHOICES
19.1.1.4.3
WEAKNESSES & COMPETITIVE THREATS
19.1.2
ABBOTT LABORATORIES
19.1.3
DANAHER CORPORATION
19.1.4
SIEMENS HEALTHINEERS AG
19.1.5
THERMO FISHER SCIENTIFIC
19.1.6
BIO-RAD LABORATORIES
19.1.8
BECTON, DICKINSON AND COMPANY
19.1.11
TOSOH CORPORATION
19.1.12
QUIDEL CORPORATION
19.2.1
LSI MEDIENCE CORPORATION (PHC HOLDINGS CORPORATION)
19.2.2
RANDOX LABORATORIES
19.2.3
GUANGZHOU WONDFO BIOTECH CO., LTD.
19.2.6
RESPONSE BIOMEDICAL
19.2.7
ALFA SCIENTIFIC DESIGNS, INC.
19.2.9
CREATIVE DIAGNOSTICS
19.2.11
DIASYS DIAGNOSTIC SYSTEMS GMBH
19.2.12
ATLAS MEDICAL GMBH
19.2.13
GETEIN BIOTECH INC.
20
RESEARCH METHODOLOGY
351
20.1.1.1
LIST OF SECONDARY SOURCES
20.1.1.2
KEY DATA FROM SECONDARY SOURCES
20.1.2.1
LIST OF PRIMARY SOURCES
20.1.2.2
KEY DATA FROM PRIMARY SOURCES
20.1.2.3
KEY INDUSTRY INSIGHTS
20.1.2.4
BREAKDOWN OF PRIMARY INTERVIEWS
20.2
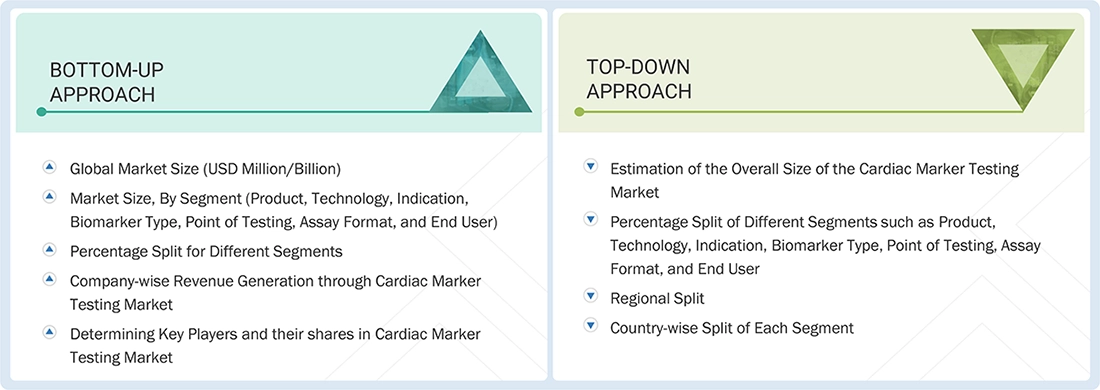
MARKET SIZE ESTIMATION
20.2.1
TOTAL MARKET SIZE: CARDIAC MARKER TESTING MARKET
20.2.2
BOTTOM-UP APPROACH
20.2.2.1
APPROACH 1: REVENUE ESTIMATION OF KEY PLAYERS
20.2.2.2
APPROACH 2: STUDY OF ANNUAL REPORTS AND INVESTOR PRESENTATIONS
20.2.2.3
APPROACH 3: PRIMARY INTERVIEWS
20.2.2.5
CAGR PROJECTIONS
20.4
RESEARCH ASSUMPTIONS
20.4.1
STUDY-RELATED ASSUMPTIONS
20.4.2
GROWTH RATE ASSUMPTIONS
20.5
RESEARCH LIMITATIONS
21.2
KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL
21.3
CUSTOMIZATION OPTIONS
TABLE 1
CARDIAC MARKER TESTING MARKET: IMPACT OF PORTER’S FORCES
TABLE 2
GDP PERCENTAGE CHANGE (%), BY KEY COUNTRY, 2023–2030
TABLE 3
NORTH AMERICA: MACROECONOMIC INDICATORS
TABLE 4
EUROPE: MACROECONOMIC INDICATORS
TABLE 5
ASIA PACIFIC: MACROECONOMIC INDICATORS
TABLE 6
LATIN AMERICA: MACROECONOMIC INDICATORS
TABLE 7
MIDDLE EAST & AFRICA: MACROECONOMIC INDICATORS
TABLE 8
CARDIAC MARKER TESTING MARKET: ROLE OF COMPANIES IN ECOSYSTEM
TABLE 9
AVERAGE SELLING PRICING OF CARDIAC MARKER TESTING PRODUCTS, BY TYPE, 2024–2026 (USD)
TABLE 10
AVERAGE SELLING PRICE OF CARDIAC MARKER TESTING PRODUCTS, BY KEY PLAYER, 2024–2026 (USD)
TABLE 11
AVERAGE SELLING PRICE OF CARDIAC MARKER TESTING PRODUCTS, BY REGION, 2024–2026 (USD)
TABLE 12
IMPORT DATA FOR HS CODE 3822 DIAGNOSTIC KITS & REAGENTS, BY COUNTRY, 2021–2025 (USD MILLION)
TABLE 13
EXPORT DATA FOR HS CODE 3822 DIAGNOSTIC KITS & REAGENTS, BY COUNTRY, 2021–2025 (USD MILLION)
TABLE 14
CARDIAC MARKER TESTING MARKET: KEY CONFERENCES & EVENTS, 2026–2027
TABLE 15
US-ADJUSTED RECIPROCAL TARIFF RATES
TABLE 16
KEY PATENTS IN CARDIAC MARKER TESTING MARKET, 2022–2026
TABLE 17
KEY COMPANIES IMPLEMENTING AI IN CARDIAC MARKER TESTING MARKET
TABLE 18
NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 19
EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 20
ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 21
LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 22
REST OF THE WORLD: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 23
INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS, BY END USER (%)
TABLE 24
KEY BUYING CRITERIA, BY END USER
TABLE 25
ADOPTION BARRIERS & INTERNAL CHALLENGES
TABLE 26
CARDIAC MARKER TESTING MARKET: UNMET NEEDS IN VARIOUS END-USE INDUSTRIES
TABLE 27
CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 28
SOME KEY COMPANIES PROVIDING CARDIAC MARKER TESTING REAGENTS & KITS
TABLE 29
CARDIAC MARKER TESTING REAGENTS & KITS MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 30
NORTH AMERICA: CARDIAC MARKER TESTING REAGENTS & KITS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 31
EUROPE: CARDIAC MARKER TESTING REAGENTS & KITS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 32
ASIA PACIFIC: CARDIAC MARKER TESTING REAGENTS & KITS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 33
LATIN AMERICA: CARDIAC MARKER TESTING REAGENTS & KITS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 34
SOME KEY COMPANIES PROVIDING CARDIAC MARKER TESTING INSTRUMENTS
TABLE 35
CARDIAC MARKER TESTING INSTRUMENTS MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 36
NORTH AMERICA: CARDIAC MARKER TESTING INSTRUMENTS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 37
EUROPE: CARDIAC MARKER TESTING INSTRUMENTS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 38
ASIA PACIFIC: CARDIAC MARKER TESTING INSTRUMENTS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 39
LATIN AMERICA: CARDIAC MARKER TESTING INSTRUMENTS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 40
CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 41
CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 42
CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY REGION, 2024–2031 (USD MILLION)
TABLE 43
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 44
EUROPE: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 45
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 46
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 47
CARDIAC MARKER TESTING MARKET FOR CHEMILUMINESCENCE, BY REGION, 2024–2031 (USD MILLION)
TABLE 48
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR CHEMILUMINESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 49
EUROPE: CARDIAC MARKER TESTING MARKET FOR CHEMILUMINESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 50
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR CHEMILUMINESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 51
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR CHEMILUMINESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 52
CARDIAC MARKER TESTING MARKET FOR IMMUNOFLUORESCENCE, BY REGION, 2024–2031 (USD MILLION)
TABLE 53
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOFLUORESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 54
EUROPE: CARDIAC MARKER TESTING MARKET FOR IMMUNOFLUORESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 55
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR IMMUNOFLUORESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 56
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOFLUORESCENCE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 57
CARDIAC MARKER TESTING MARKET FOR LATERAL FLOW IMMUNOASSAYS, BY REGION, 2024–2031 (USD MILLION)
TABLE 58
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR LATERAL FLOW IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 59
EUROPE: CARDIAC MARKER TESTING MARKET FOR LATERAL FLOW IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 60
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR LATERAL FLOW IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 61
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR LATERAL FLOW IMMUNOASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 62
CARDIAC MARKER TESTING MARKET FOR ELISA, BY REGION, 2024–2031 (USD MILLION)
TABLE 63
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR ELISA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 64
EUROPE: CARDIAC MARKER TESTING MARKET FOR ELISA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 65
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR ELISA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 66
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR ELISA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 67
CARDIAC MARKER TESTING MARKET FOR CLINICAL CHEMISTRY, BY REGION, 2024–2031 (USD MILLION)
TABLE 68
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR CLINICAL CHEMISTRY, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 69
EUROPE: CARDIAC MARKER TESTING MARKET FOR CLINICAL CHEMISTRY, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 70
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR CLINICAL CHEMISTRY, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 71
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR CLINICAL CHEMISTRY, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 72
CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 73
CARDIAC MARKER TESTING MARKET FOR MYOCARDIAL INFARCTION, BY REGION, 2024–2031 (USD MILLION)
TABLE 74
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR MYOCARDIAL INFARCTION, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 75
EUROPE: CARDIAC MARKER TESTING MARKET FOR MYOCARDIAL INFARCTION, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 76
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR MYOCARDIAL INFARCTION, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 77
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR MYOCARDIAL INFARCTION, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 78
CARDIAC MARKER TESTING MARKET FOR CONGESTIVE HEART FAILURE, BY REGION, 2024–2031 (USD MILLION)
TABLE 79
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR CONGESTIVE HEART FAILURE , BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 80
EUROPE: CARDIAC MARKER TESTING MARKET FOR CONGESTIVE HEART FAILURE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 81
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR CONGESTIVE HEART FAILURE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 82
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR CONGESTIVE HEART FAILURE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 83
CARDIAC MARKER TESTING MARKET FOR ACUTE CORONARY SYNDROME, BY REGION, 2024–2031 (USD MILLION)
TABLE 84
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR ACUTE CORONARY SYNDROME, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 85
EUROPE: CARDIAC MARKER TESTING MARKET FOR ACUTE CORONARY SYNDROME, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 86
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR ACUTE CORONARY SYNDROME, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 87
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR ACUTE CORONARY SYNDROME, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 88
CARDIAC MARKER TESTING MARKET FOR ATHEROSCLEROSIS, BY REGION, 2024–2031 (USD MILLION)
TABLE 89
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR ATHEROSCLEROSIS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 90
EUROPE: CARDIAC MARKER TESTING MARKET FOR ATHEROSCLEROSIS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 91
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR ATHEROSCLEROSIS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 92
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR ATHEROSCLEROSIS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 93
CARDIAC MARKER TESTING MARKET FOR ISCHEMIA, BY REGION, 2024–2031 (USD MILLION)
TABLE 94
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR ISCHEMIA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 95
EUROPE: CARDIAC MARKER TESTING MARKET FOR ISCHEMIA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 96
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR ISCHEMIA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 97
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR ISCHEMIA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 98
CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 99
CARDIAC MARKER TESTING MARKET FOR TROPONIN I & T, BY REGION, 2024–2031 (USD MILLION)
TABLE 100
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR TROPONIN I & T, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 101
EUROPE: CARDIAC MARKER TESTING MARKET FOR TROPONIN I & T, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 102
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR TROPONIN I & T, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 103
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR TROPONIN I & T, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 104
CARDIAC MARKER TESTING MARKET FOR CREATININE KINASE-MB, BY REGION, 2024–2031 (USD MILLION)
TABLE 105
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR CREATININE KINASE-MB, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 106
EUROPE: CARDIAC MARKER TESTING MARKET FOR CREATININE KINASE-MB, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 107
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR CREATININE KINASE-MB, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 108
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR CREATININE KINASE-MB, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 109
CARDIAC MARKER TESTING MARKET FOR BRAIN NATRIURETIC PEPTIDE, BY REGION, 2024–2031 (USD MILLION)
TABLE 110
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR BRAIN NATRIURETIC PEPTIDE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 111
EUROPE: CARDIAC MARKER TESTING MARKET FOR BRAIN NATRIURETIC PEPTIDE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 112
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR BRAIN NATRIURETIC PEPTIDE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 113
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR BRAIN NATRIURETIC PEPTIDE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 114
CARDIAC MARKER TESTING MARKET FOR MYOGLOBIN, BY REGION, 2024–2031 (USD MILLION)
TABLE 115
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR MYOGLOBIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 116
EUROPE: CARDIAC MARKER TESTING MARKET FOR MYOGLOBIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 117
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR MYOGLOBIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 118
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR MYOGLOBIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 119
CARDIAC MARKER TESTING MARKET FOR HOMOCYSTEINE, BY REGION, 2024–2031 (USD MILLION)
TABLE 120
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR HOMOCYSTEINE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 121
EUROPE: CARDIAC MARKER TESTING MARKET FOR HOMOCYSTEINE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 122
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR HOMOCYSTEINE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 123
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR HOMOCYSTEINE, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 124
CARDIAC MARKER TESTING MARKET FOR HIGH-SENSITIVITY C-REACTIVE PROTEIN, BY REGION, 2024–2031 (USD MILLION)
TABLE 125
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR HIGH-SENSITIVITY C-REACTIVE PROTEIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 126
EUROPE: CARDIAC MARKER TESTING MARKET FOR HIGH-SENSITIVITY C-REACTIVE PROTEIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 127
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR HIGH-SENSITIVITY C-REACTIVE PROTEIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 128
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR HIGH-SENSITIVITY C-REACTIVE PROTEIN, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 129
CARDIAC MARKER TESTING MARKET FOR OTHER CARDIAC BIOMARKERS, BY REGION, 2024–2031 (USD MILLION)
TABLE 130
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR OTHER CARDIAC BIOMARKERS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 131
EUROPE: CARDIAC MARKER TESTING MARKET FOR OTHER CARDIAC BIOMARKERS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 132
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR OTHER CARDIAC BIOMARKERS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 133
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR OTHER CARDIAC BIOMARKERS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 134
US: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 135
CANADA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 136
GERMANY: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 137
UK: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 138
FRANCE: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 139
ITALY: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 140
SPAIN: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 141
CHINA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 142
JAPAN: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 143
INDIA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 144
BRAZIL: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 145
MEXICO: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (MILLION UNITS)
TABLE 146
CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 147
CARDIAC MARKER TESTING MARKET FOR LAB TESTS, BY REGION, 2024–2031 (USD MILLION)
TABLE 148
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR LAB TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 149
EUROPE: CARDIAC MARKER TESTING MARKET FOR LAB TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 150
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR LAB TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 151
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR LAB TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 152
CARDIAC MARKER TESTING MARKET FOR POC TESTS, BY REGION, 2024–2031 (USD MILLION)
TABLE 153
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR POC TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 154
EUROPE: CARDIAC MARKER TESTING MARKET FOR POC TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 155
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR POC TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 156
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR POC TESTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 157
CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 158
CARDIAC MARKER TESTING MARKET FOR SINGLEPLEX ASSAYS, BY REGION, 2024–2031 (USD MILLION)
TABLE 159
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR SINGLEPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 160
EUROPE: CARDIAC MARKER TESTING MARKET FOR SINGLEPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 161
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR SINGLEPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 162
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR SINGLEPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 163
CARDIAC MARKER TESTING MARKET FOR MULTIPLEX ASSAYS, BY REGION, 2024–2031 (USD MILLION)
TABLE 164
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR MULTIPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 165
EUROPE: CARDIAC MARKER TESTING MARKET FOR MULTIPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 166
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR MULTIPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 167
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR MULTIPLEX ASSAYS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 168
CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 169
CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 170
CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY REGION, 2024–2031 (USD MILLION)
TABLE 171
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 172
EUROPE: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 173
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 174
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 175
CARDIAC MARKER TESTING MARKET FOR HOSPITAL LABORATORIES, BY REGION, 2024–2031 (USD MILLION)
TABLE 176
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR HOSPITAL LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 177
EUROPE: CARDIAC MARKER TESTING MARKET FOR HOSPITAL LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 178
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR HOSPITAL LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 179
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR HOSPITAL LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 180
CARDIAC MARKER TESTING MARKET FOR REFERENCE LABORATORIES, BY REGION, 2024–2031 (USD MILLION)
TABLE 181
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR REFERENCE LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 182
EUROPE: CARDIAC MARKER TESTING MARKET FOR REFERENCE LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 183
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR REFERENCE LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 184
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR REFERENCE LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 185
CARDIAC MARKER TESTING MARKET FOR CONTRACT TESTING LABORATORIES, BY REGION, 2024–2031 (USD MILLION)
TABLE 186
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR CONTRACT TESTING LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 187
EUROPE: CARDIAC MARKER TESTING MARKET FOR CONTRACT TESTING LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 188
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR CONTRACT TESTING LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 189
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR CONTRACT TESTING LABORATORIES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 190
CARDIAC MARKER TESTING MARKET FOR ACADEMIC INSTITUTES, BY REGION, 2024–2031 (USD MILLION)
TABLE 191
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR ACADEMIC INSTITUTES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 192
EUROPE: CARDIAC MARKER TESTING MARKET FOR ACADEMIC INSTITUTES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 193
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR ACADEMIC INSTITUTES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 194
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR ACADEMIC INSTITUTES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 195
CARDIAC MARKER TESTING MARKET FOR POINT-OF-CARE TESTING, BY REGION, 2024–2031 (USD MILLION)
TABLE 196
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR POINT-OF-CARE TESTING, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 197
EUROPE: CARDIAC MARKER TESTING MARKET FOR POINT-OF-CARE TESTING, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 198
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR POINT-OF-CARE TESTING, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 199
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR POINT-OF-CARE TESTING, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 200
CARDIAC MARKER TESTING MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 201
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 202
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 203
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 204
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 205
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 206
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 207
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 208
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 209
NORTH AMERICA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 210
NORTH AMERICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 211
US: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 212
US: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 213
US: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 214
US: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 215
US: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 216
US: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 217
US: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 218
US: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 219
US: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 220
CANADA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 221
CANADA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 222
CANADA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 223
CANADA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 224
CANADA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 225
CANADA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 226
CANADA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 227
CANADA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 228
CANADA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 229
EUROPE: CARDIAC MARKER TESTING MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 230
EUROPE: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 231
EUROPE: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 232
EUROPE: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 233
EUROPE: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 234
EUROPE: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 235
EUROPE: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 236
EUROPE: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 237
EUROPE: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 238
EUROPE: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 239
GERMANY: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 240
GERMANY: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 241
GERMANY: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 242
GERMANY: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 243
GERMANY: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 244
GERMANY: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 245
GERMANY: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 246
GERMANY: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 247
GERMANY: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 248
UK: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 249
UK: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 250
UK: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 251
UK: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 252
UK: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 253
UK: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 254
UK: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 255
UK: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 256
UK: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 257
FRANCE: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 258
FRANCE: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 259
FRANCE: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 260
FRANCE: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 261
FRANCE: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 262
FRANCE: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 263
FRANCE: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 264
FRANCE: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 265
FRANCE: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 266
ITALY: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 267
ITALY: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 268
ITALY: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 269
ITALY: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 270
ITALY: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 271
ITALY: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 272
ITALY: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 273
ITALY: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 274
ITALY: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 275
SPAIN: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 276
SPAIN: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 277
SPAIN: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 278
SPAIN: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 279
SPAIN: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 280
SPAIN: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 281
SPAIN: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 282
SPAIN: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 283
SPAIN: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 284
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 285
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 286
REST OF EUROPE: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 287
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 288
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 289
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 290
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 291
REST OF EUROPE: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 292
REST OF EUROPE: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 293
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 294
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 295
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 296
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 297
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 298
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 299
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 300
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 301
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 302
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 303
JAPAN: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 304
JAPAN: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 305
JAPAN: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 306
JAPAN: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 307
JAPAN: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 308
JAPAN: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 309
JAPAN: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 310
JAPAN: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 311
JAPAN: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 312
CHINA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 313
CHINA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 314
CHINA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 315
CHINA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 316
CHINA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 317
CHINA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 318
CHINA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 319
CHINA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 320
CHINA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 321
INDIA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 322
INDIA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 323
INDIA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 324
INDIA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 325
INDIA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 326
INDIA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 327
INDIA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 328
INDIA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 329
INDIA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 330
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 331
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 332
AUSTRALIA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 333
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 334
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 335
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 336
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 337
AUSTRALIA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 338
AUSTRALIA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 339
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 340
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 341
SOUTH KOREA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAY, BY TYPE, 2024–2031 (USD MILLION)
TABLE 342
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 343
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 344
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 345
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 346
SOUTH KOREA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 347
SOUTH KOREA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 348
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 349
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 350
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 351
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 352
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 353
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 354
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 355
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 356
REST OF ASIA PACIFIC: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 357
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 358
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 359
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 360
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 361
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 362
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 363
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 364
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 365
LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 366
LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 367
BRAZIL: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 368
BRAZIL: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 369
BRAZIL: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 370
BRAZIL: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 371
BRAZIL: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 372
BRAZIL: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 373
BRAZIL: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 374
BRAZIL: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 375
BRAZIL: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 376
MEXICO: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 377
MEXICO: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 378
MEXICO: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 379
MEXICO: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 380
MEXICO: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 381
MEXICO: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 382
MEXICO: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 383
MEXICO: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 384
MEXICO: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 385
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 386
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 387
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAY, BY TYPE, 2024–2031 (USD MILLION)
TABLE 388
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 389
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 390
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 391
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 392
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 393
REST OF LATIN AMERICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 394
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 395
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION)
TABLE 396
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET FOR IMMUNOASSAYS, BY TYPE, 2024–2031 (USD MILLION)
TABLE 397
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 398
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE, 2024–2031 (USD MILLION)
TABLE 399
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY POINT OF TESTING, 2024–2031 (USD MILLION)
TABLE 400
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY ASSAY FORMAT, 2024–2031 (USD MILLION)
TABLE 401
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 402
MIDDLE EAST & AFRICA: CARDIAC MARKER TESTING MARKET FOR LABORATORY TESTING FACILITIES, BY TYPE, 2024–2031 (USD MILLION)
TABLE 403
OVERVIEW OF STRATEGIES DEPLOYED BY KEY COMPANIES IN CARDIAC MARKER TESTING MARKET, JANUARY 2023−MARCH 2026
TABLE 404
CARDIAC MARKER TESTING MARKET: DEGREE OF COMPETITION
TABLE 405
CARDIAC MARKER TESTING MARKET: REGION FOOTPRINT
TABLE 406
CARDIAC MARKER TESTING MARKET: PRODUCT FOOTPRINT
TABLE 407
CARDIAC MARKER TESTING MARKET: TECHNOLOGY FOOTPRINT
TABLE 408
CARDIAC MARKER TESTING MARKET: POINT OF TESTING FOOTPRINT
TABLE 409
CARDIAC MARKER TESTING MARKET: DETAILED LIST OF KEY STARTUPS/SMES
TABLE 410
CARDIAC MARKER TESTING MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES (1/2)
TABLE 411
CARDIAC MARKER TESTING MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES (2/2)
TABLE 412
CARDIAC MARKER TESTING MARKET: PRODUCT APPROVALS, JANUARY 2023–JANUARY 2026
TABLE 413
CARDIAC MARKER TESTING MARKET: DEALS, JANUARY 2023–JANUARY 2026
TABLE 414
F. HOFFMANN-LA ROCHE AG: COMPANY OVERVIEW
TABLE 415
F. HOFFMANN-LA ROCHE AG: PRODUCTS OFFERED
TABLE 416
F. HOFFMANN-LA ROCHE AG: PRODUCT APPROVALS, JANUARY 2023–JANUARY 2026
TABLE 417
F. HOFFMANN-LA ROCHE AG: DEALS, JANUARY 2023–JANUARY 2026
TABLE 418
ABBOTT LABORATORIES: COMPANY OVERVIEW
TABLE 419
ABBOTT LABORATORIES: PRODUCTS OFFERED
TABLE 420
DANAHER CORPORATION: COMPANY OVERVIEW
TABLE 421
DANAHER CORPORATION: PRODUCTS OFFERED
TABLE 422
SIEMENS HEALTHINEERS AG: COMPANY OVERVIEW
TABLE 423
SIEMENS HEALTHINEERS AG: PRODUCTS OFFERED
TABLE 424
THERMO FISHER SCIENTIFIC: COMPANY OVERVIEW
TABLE 425
THERMO FISHER SCIENTIFIC: PRODUCTS OFFERED
TABLE 426
BIO-RAD LABORATORIES : COMPANY OVERVIEW
TABLE 427
BIO-RAD LABORATORIES : PRODUCTS OFFERED
TABLE 428
BIOMÉRIEUX SA : COMPANY OVERVIEW
TABLE 429
BIOMÉRIEUX SA : PRODUCTS OFFERED
TABLE 430
BIOMÉRIEUX SA: DEALS, JANUARY 2023–JANUARY 2026
TABLE 431
BECTON, DICKINSON AND COMPANY: COMPANY OVERVIEW
TABLE 432
BECTON, DICKINSON AND COMPANY: PRODUCTS OFFERED
TABLE 433
DIASORIN SPA: COMPANY OVERVIEW
TABLE 434
DIASORIN SPA: PRODUCTS OFFERED
TABLE 435
REVVITY: COMPANY OVERVIEW
TABLE 436
REVVITY: PRODUCTS OFFERED
TABLE 437
TOSOH CORPORATION: COMPANY OVERVIEW
TABLE 438
TOSOH CORPORATION: PRODUCTS OFFERED
TABLE 439
QUIDEL CORPORATION: COMPANY OVERVIEW
TABLE 440
QUIDEL CORPORATION: PRODUCTS OFFERED
TABLE 441
QUIDEL CORPORATION: PRODUCT APPROVALS, JANUARY 2023–JANUARY 2026
TABLE 442
QUIDEL CORPORATION: DEALS, JANUARY 2023–JANUARY 2026
TABLE 443
LSI MEDIENCE CORPORATION (PHC HOLDINGS CORPORATION): COMPANY OVERVIEW
TABLE 444
RANDOX LABORATORIES: COMPANY OVERVIEW
TABLE 445
GUANGZHOU WONDFO BIOTECH CO., LTD.: COMPANY OVERVIEW
TABLE 446
BODITECH MED INC.: COMPANY OVERVIEW
TABLE 447
BTNX INC.: COMPANY OVERVIEW
TABLE 448
RESPONSE BIOMEDICAL: COMPANY OVERVIEW
TABLE 449
ALFA SCIENTIFIC DESIGNS, INC.: COMPANY OVERVIEW
TABLE 450
CTK BIOTECH: COMPANY OVERVIEW
TABLE 451
CREATIVE DIAGNOSTICS: COMPANY OVERVIEW
TABLE 452
LIFESIGN LLC: COMPANY OVERVIEW
TABLE 453
DIASYS DIAGNOSTIC SYSTEMS GMBH: COMPANY OVERVIEW
TABLE 454
ATLAS MEDICAL GMBH: COMPANY OVERVIEW
TABLE 455
GETEIN BIOTECH INC.: COMPANY OVERVIEW
TABLE 456
STUDY ASSUMPTIONS
TABLE 457
CARDIAC MARKER TESTING MARKET: RISK ASSESSMENT
FIGURE 1
CARDIAC MARKER TESTING MARKET SEGMENTATION & REGIONAL SCOPE
FIGURE 2
KEY INSIGHTS & MARKET HIGHLIGHTS
FIGURE 3
GLOBAL CARDIAC MARKER TESTING MARKET, 2024–2031
FIGURE 4
MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CARDIAC MARKER TESTING MARKET, 2023–2025
FIGURE 5
DISRUPTIVE TRENDS IMPACTING GROWTH OF CARDIAC MARKER TESTING MARKET
FIGURE 6
HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS IN CARDIAC MARKER TESTING MARKET, 2025
FIGURE 7
ASIA PACIFIC TO REGISTER HIGHEST GROWTH DURING FORECAST PERIOD
FIGURE 8
RISING CASES OF CARDIOVASCULAR DISEASES TO DRIVE MARKET
FIGURE 9
REAGENTS & KITS SEGMENT TO DOMINATE MARKET DURING FORECAST PERIOD
FIGURE 10
IMMUNOASSAYS SEGMENT TO LEAD MARKET DURING FORECAST PERIOD
FIGURE 11
MYOCARDIAL INFARCTION SEGMENT TO LEAD MARKET DURING FORECAST PERIOD
FIGURE 12
TROPONIN I & T SEGMENT TO LEAD MARKET DURING FORECAST PERIOD
FIGURE 13
LAB TESTS SEGMENT TO LEAD MARKET DURING FORECAST PERIOD
FIGURE 14
SINGLEPLEX ASSAYS SEGMENT TO LEAD MARKET DURING FORECAST PERIOD
FIGURE 15
LABORATORY TESTING FACILITIES SEGMENT TO COMMAND LARGEST SHARE DURING FORECAST PERIOD
FIGURE 16
ASIA PACIFIC TO REGISTER HIGHEST GROWTH DURING FORECAST PERIOD
FIGURE 17
CARDIAC MARKER TESTING MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
FIGURE 18
CARDIAC MARKER TESTING MARKET: PORTER’S FIVE FORCES ANALYSIS
FIGURE 19
CARDIAC MARKER TESTING MARKET: SUPPLY CHAIN ANALYSIS
FIGURE 20
CARDIAC MARKER TESTING MARKET: VALUE CHAIN ANALYSIS
FIGURE 21
CARDIAC MARKER TESTING MARKET: ECOSYSTEM ANALYSIS
FIGURE 22
CARDIAC MARKER TESTING MARKET: TRENDS & DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES
FIGURE 23
NUMBER OF DEALS & FUNDING ACTIVITIES IN CARDIAC MARKER TESTING MARKET
FIGURE 24
PATENT ANALYSIS FOR CARDIAC MARKER TESTING, JANUARY 2016–DECEMBER 2025
FIGURE 25
MARKET POTENTIAL OF AI IN CARDIAC MARKER TESTING MARKET
FIGURE 26
INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS, BY END USER
FIGURE 27
KEY BUYING CRITERIA, BY END USER
FIGURE 28
NORTH AMERICA: CARDIAC MARKER TESTING MARKET SNAPSHOT
FIGURE 29
ASIA PACIFIC: CARDIAC MARKER TESTING MARKET SNAPSHOT
FIGURE 30
REVENUE ANALYSIS OF KEY PLAYERS IN CARDIAC MARKER TESTING MARKET, 2023–2025 (USD MILLION)
FIGURE 31
CARDIAC MARKER TESTING MARKET SHARE ANALYSIS, 2025 (GLOBAL)
FIGURE 32
US: CARDIAC MARKER TESTING MARKET SHARE ANALYSIS, 2025
FIGURE 33
CARDIAC MARKER TESTING MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
FIGURE 34
CARDIAC MARKER TESTING MARKET: COMPANY FOOTPRINT
FIGURE 35
CARDIAC MARKER TESTING MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
FIGURE 36
EV/EBITDA OF KEY VENDORS
FIGURE 37
YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
FIGURE 38
CARDIAC MARKER TESTING MARKET: BRAND/PRODUCT COMPARISON FOR CARDIAC MARKER TESTING
FIGURE 39
F. HOFFMANN-LA ROCHE AG: COMPANY SNAPSHOT (2025)
FIGURE 40
ABBOTT LABORATORIES: COMPANY SNAPSHOT (2025)
FIGURE 41
DANAHER CORPORATION: COMPANY SNAPSHOT (2025)
FIGURE 42
SIEMENS HEALTHINEERS AG: COMPANY SNAPSHOT (2025)
FIGURE 43
THERMO FISHER SCIENTIFIC: COMPANY SNAPSHOT (2025)
FIGURE 44
BIO-RAD LABORATORIES : COMPANY SNAPSHOT (2025)
FIGURE 45
BIOMÉRIEUX SA : COMPANY SNAPSHOT (2025)
FIGURE 46
BECTON, DICKINSON AND COMPANY: COMPANY SNAPSHOT (2025)
FIGURE 47
DIASORIN SPA: COMPANY SNAPSHOT (2024)
FIGURE 48
REVVITY: COMPANY SNAPSHOT (2025)
FIGURE 49
TOSOH CORPORATION: COMPANY SNAPSHOT (2025)
FIGURE 50
QUIDEL CORPORATION: COMPANY SNAPSHOT (2025)
FIGURE 51
RESEARCH DESIGN
FIGURE 52
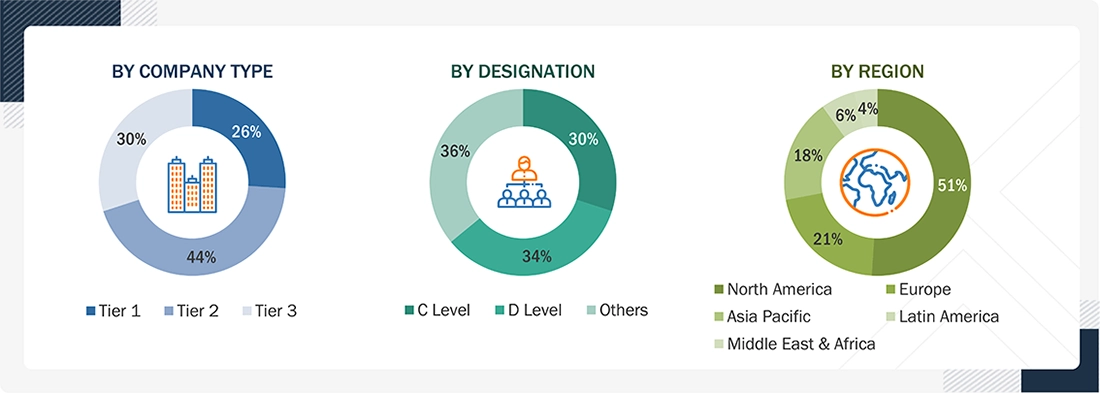
BREAKDOWN OF PRIMARY INTERVIEWS: BY COMPANY TYPE, DESIGNATION, AND REGION
FIGURE 53
TOP-DOWN APPROACH
FIGURE 54
DATA TRIANGULATION
FIGURE 55
PARAMETRIC ASSUMPTIONS


































Growth opportunities and latent adjacency in Cardiac Marker Testing Market