
Contract Research Organization (CRO) Services Market
Contract Research Organization (CRO) Services Market by Type (Early Phase, Clinical, Lab, Consulting), Therapeutic Area [Oncology (Breast, Lung), Infectious, Neurology, Vaccines], Modality, Model (FSO, FSP), End User, Competition - Global Forecast to 2031




CRO SERVICES MARKET SIZE, SHARE & GROWTH SNAPSHOT
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
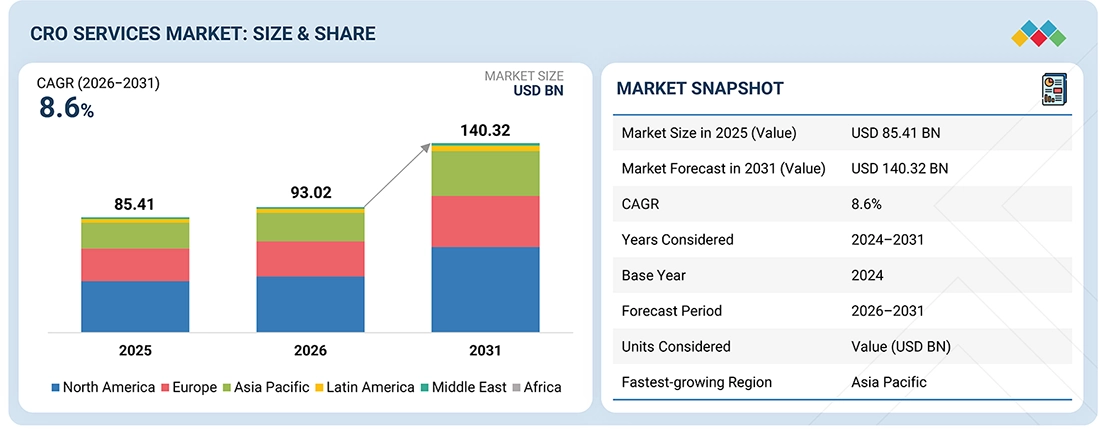
The global contract research organization (CRO) services market is projected to grow from USD 93.02 billion in 2026 to USD 140.32 billion by 2031, at a CAGR of 8.6% during the forecast period. The market was valued at USD 85.41 billion in 2025. Factors such as the increasing complexity & the growing volume of trials, the rising focus on patient-centric clinical trials such as Decentralized Clinical trials (DCTs), and the flexibility of services offered by CROs are supporting the growth of this market. Growth is also supported by the increasing outsourcing of drug development and clinical research activities by pharmaceutical, biopharmaceutical, and medical device companies.
KEY TAKEAWAYS
-
BY REGIONNorth America is the largest market, accounting for a share of 44.5% in 2025.
-
BY SERVICE TYPEClinical research services accounted for the largest share, at 57.6%, in 2025.
-
BY THERAPEUTIC AREAOncology dominates the market with 35.5% share in 2025.
-
BY END USERMedical device companies are expected to grow at the fastest rate during the forecast period of 2026 to 2031.
-
COMPETITIVE LANDSCAPEMajor market players have adopted both organic and inorganic strategies, including partnerships and investments. Thermo Fisher Scientific Inc. launched the international CorEvitas Adolescent Alopecia Areata (AA) Registry to address a critical unmet need for real-world, adolescent-specific evidence and data on this autoimmune disease.
The CRO services market is witnessing steady growth, driven by factors such as the increasing complexity & the growing volume of trials; the rising focus on patient-centric clinical trials such as Decentralized Clinical trials (DCTs); and the flexibility of services offered by CROs are supporting the growth of this market. Moreover, the upcoming therapeutic drugs patent cliff is fueling pharmaceutical companies to invest heavily in R&D activities and outsource their clinical trials to CROs. Additionally, the growing regulatory focus on patient diversity and data analysis offerings that favor customer reimbursement & market access strategies are other key growth opportunities.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The impact on biopharma clients’ businesses in the CRO services market arises from evolving sponsor demands and changes in global R&D strategy. Pharmaceutical, biotechnology, medical device, and diagnostics companies are the largest users of CRO solutions, with efficient clinical development and regulatory compliance as primary focus areas. Trends such as increased outsourcing, adoption of decentralized and digital clinical trials, and stricter regulatory requirements now directly affect project timelines, budgets, and commercial launch success for sponsors.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Rising trial volume and complexity

-
Growing focus on decentralized/patient-centric trials
Level
-
No restraints
Level
-
Growing regulatory stringency on the diversity of the patient pool
-
Risk-based monitoring (RBM) and risk-based quality management (RBQM) as a service
Level
-
Patient retention
-
Cybersecurity and intellectual property challenges
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Rising trial volume and complexity
The steady rise in global clinical trial volume and increasing protocol complexity are key drivers of CRO market growth. More trials target niche populations in oncology, rare diseases, and cell & gene therapies, requiring specialized expertise and broader geographic reach. Protocol designs demand larger datasets, adaptive methodologies, biomarker integration, and real-world evidence collection, significantly expanding operational demands. These complexities push sponsors to outsource to CROs that can provide global site networks, advanced data analytics, and regulatory alignment. By alleviating the resource and infrastructure burden for sponsors, CROs are positioned as essential partners in accelerating trial execution and ensuring compliance across multiple geographies. As pipelines expand and therapeutic science evolves, the reliance on CRO services is expected to deepen, reinforcing their central role in modern drug development.
Opportunity: Growing regulatory stringency on the diversity of the patient pool
Regulatory agencies like the FDA and EMA are increasing requirements for diversity in clinical trials to ensure effective treatments for all populations. The FDA’s 2024 guidelines require trial sponsors to develop Diversity Action Plans (DAPs) to include underrepresented groups, while the EMA’s Clinical Trials Regulation (EU) No. 536/2014 promotes broader participant inclusion. In Europe, the UK HRA/MHRA is piloting Inclusion & Diversity Plans for clinical trials, thereby widening opportunities for CRO partners with community outreach, decentralized models, and real-world data to deliver on these plans across geographies. In response, CROs are enhancing their offerings, creating an opportunity for growth over the forecast period.
Challenge: Patient retention
Patient retention remains a consistent challenge mainly due to burdensome visit schedules, complex protocols, and long study durations. These can derail endpoints. Thus, CROs are building patient-centric programs explicitly to boost engagement and keep participants enrolled. For instance, IQVIA is tackling attrition with digital and in-home support. Its Health Research Space patient app earned a 2025 MedTech Breakthrough Award for engagement, and its Decentralized Trial Suite added Mobile Research Nursing & Phlebotomy to bring visits to the patient, reducing the friction that drives dropouts.
CONTRACT RESEARCH ORGANIZATION SERVICES MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
ICON plc provided clinical trial services to the Pfizer and BioNTech SE investigational COVID-19 vaccine programme. | Provided high level of remote clinical monitoring and source data verification, safeguarding data quality and integrity in the evolving pandemic environment |
 |
IQVIA collaborated with Janssen Research & Development, LLC on the Phase 3 COVID-19 vaccine clinical trials | The client leveraged IQVIA's suite of decentralized trial solutions. Employment of telehealth technologies, virtual oversight, and digital patient engagement strategies. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The CRO services market ecosystem comprises contract research organizations (such as IQVIA, ICON, Syneos Health), specialized service providers (for clinical trials, data management, regulatory affairs), and sponsor clients, including pharmaceutical, biotechnology, and medical device companies (like Pfizer, Novartis, and Medtronic). CROs provide essential expertise and infrastructure to design, execute, and manage clinical studies, advancing new therapies from early discovery through regulatory approval. Sponsors rely on CRO partners for cost efficiency, faster timelines, and access to global patient populations, while CROs deliver operational excellence and regulatory compliance. Collaborative partnerships across this value chain are essential for accelerating innovation, lowering development costs, and ensuring success in a highly regulated, competitive life sciences market.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
CRO Services Market, By Service Type
In 2025, clinical research services held the largest share of the CRO services market and will continue leading the market through 2025. The large share of this segment is attributed to the Phase I-IV trial execution, which is the most resource-intensive and cost-driving stage of drug development. Sponsors increasingly rely on CROs for clinical monitoring, site management, patient recruitment, medical writing, pharmacovigilance, and trial operations to accelerate timelines and meet regulatory demands.
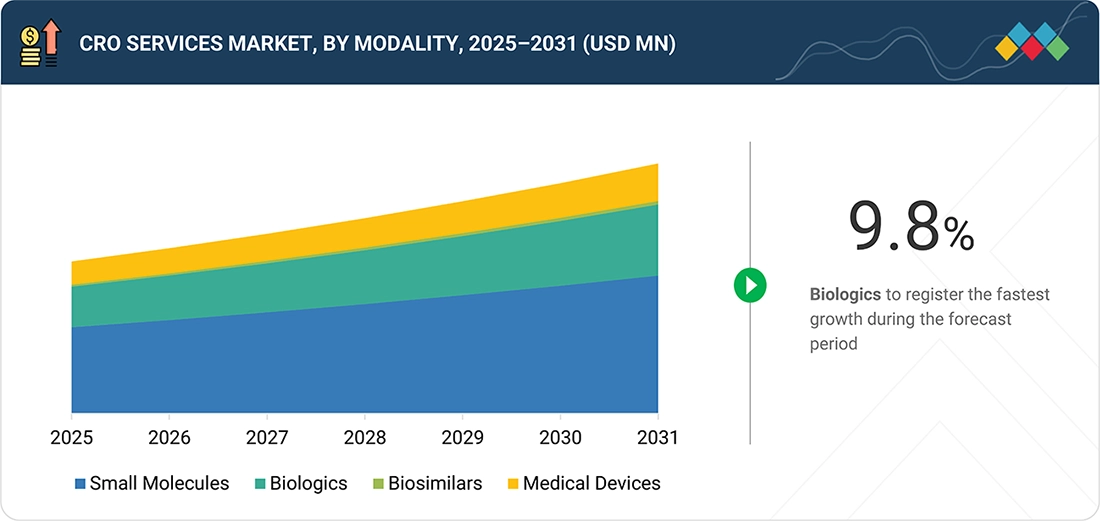
CRO Services Market, By Modality
In 2025, small molecules dominated the market. Their large share can be attributed to factors such as the dominance of the pipeline by volume and the loss of exclusivity for patented products. Additionally, the generics and bioequivalence market adds recurring study volume.
CRO Services Market, By Therapeutic area
In 2025, the oncology therapeutic area is expected to dominate the CRO services market. Oncology commands the largest share of CRO services because it remains the most innovation-intensive area in drug development, with pipelines expanding and clinical development productivity improving, fueling sustained trial demand.
REGION
Asia Pacific to be fastest-growing region in global CRO services market during forecast period
The Asia Pacific CRO services market is expected to register the highest CAGR during the forecast period, driven by growth in the pharmaceutical and biopharmaceutical industries, the low cost of conducting clinical trials in the region, favorable government policies, and the rising number of CROs.

CONTRACT RESEARCH ORGANIZATION SERVICES MARKET: COMPANY EVALUATION MATRIX
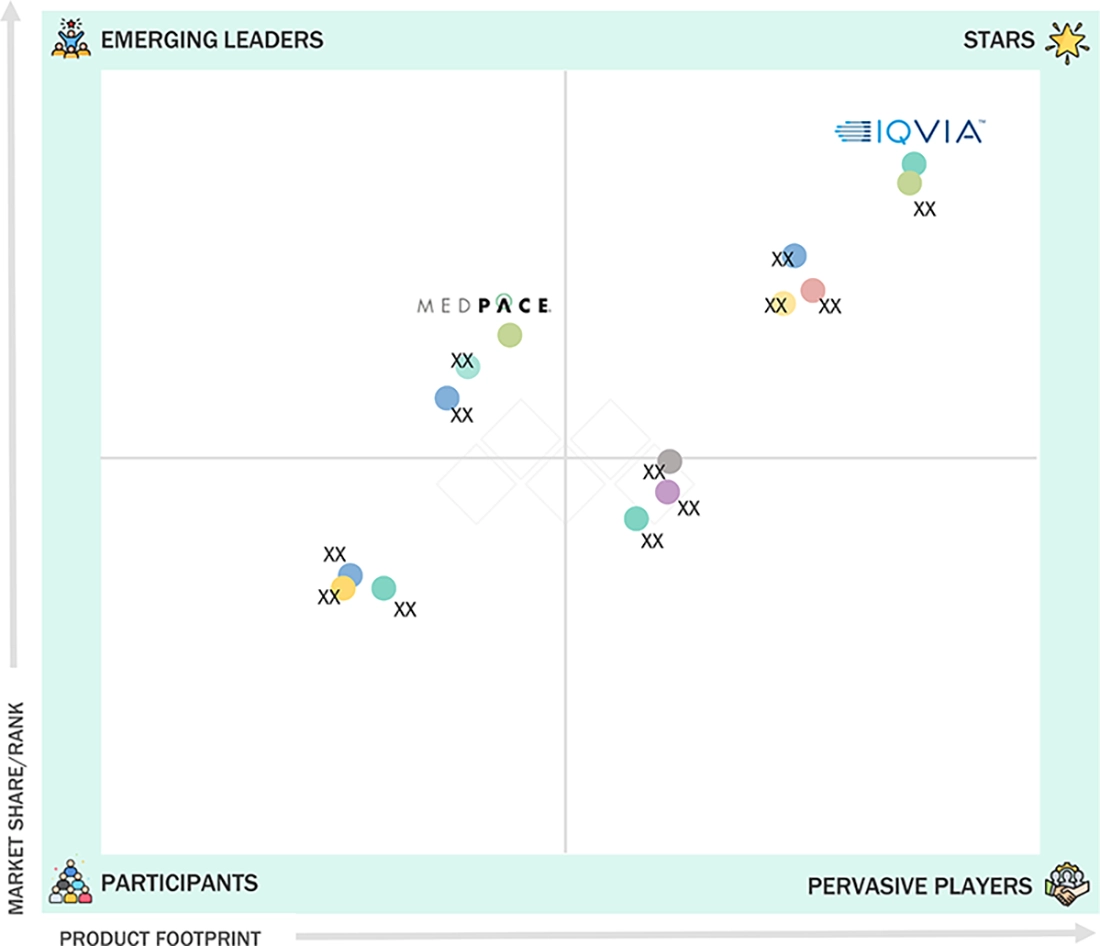
In the CRO services market matrix, IQVIA (Star) leads with commanding market share and global operational reach, supported by its advanced data analytics, technology-driven clinical trial solutions, and deep expertise serving top pharmaceutical and biotech sponsors. IQVIA’s integrated offerings and extensive global site network make it the preferred partner for large-scale, multi-regional clinical programs. Medpace (Emerging Leader) is rapidly gaining recognition for its specialized focus in specific therapeutic areas, hands-on project management, and high-touch service model tailored to emerging biotechs and mid-sized pharma clients. Through quality execution and niche expertise, Medpace continues to strengthen its competitive position and expand its footprint in key markets.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- IQVIA, Inc.
- ICON Plc.
- Thermo Fisher Scientific Inc.
- Fortrea
- Syneos Health
- LabCorp (US)
- Medpace
- WuXi AppTec
- Frontage Labs
- Pharmaron
- Tigermed
- SGS Société Générale De Surveillance SA.
- Eurofins Scientific
- Linical
- Parexel International Corporation
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 85.41 Billion |
| Market Forecast in 2031 (Value) | USD 140.32 Billion |
| Growth Rate | CAGR of 8.6% from 2026-2031 |
| Years Considered | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD Billion) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East, Africa |
WHAT IS IN IT FOR YOU: CONTRACT RESEARCH ORGANIZATION SERVICES MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Leading CRO |
|
|
| Leading CRO |
|
|
| SME CRO |
|
|
RECENT DEVELOPMENTS
- March 2026 : IQVIA launched IQVIA.ai, a Unified Agentic AI Platform Powered by NVIDIA to improve efficiency and decision-making across life sciences.
- March 2025 : ICON plc and Mural Health Technologies, Inc. partnered to use the participant management and payments platform, Mural Link.
- January 2024 : Thermo Fisher Scientific Inc. expanded its global laboratory services capabilities with a new bioanalytical lab at GoCo Health Innovation City in Gothenburg, Sweden, serving pharmaceutical and biotech customers with advanced laboratory services and instrumentation across all phases of pharmaceutical development.
Table of Contents

Methodology
This research study extensively used secondary sources, directories, and databases to identify and collect valuable information to analyze the global CRO services market. In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, to obtain and verify critical qualitative and quantitative information and assess the growth prospects of the market. The global market size estimated through secondary research was then triangulated with inputs from primary research to arrive at the final market size.
Secondary Research
The market for companies offering CRO services is derived from secondary data from paid and unpaid sources, analysis of the service portfolios of leading companies in the ecosystem, and rating of companies by performance and quality. Various sources were consulted during the secondary research process to identify & collect information for this study. The secondary sources include annual reports, company investor presentations, white papers, journals, certified publications, articles by recognized authors, directories, and databases. The secondary research was used to obtain critical information on the industry’s value chain, the total pool of key players, market classification, and segmentation from market & technology-oriented perspectives. Secondary data were analyzed to determine the overall size of the global CRO services market, and the findings were validated through primary research.
Primary Research
Extensive primary research was conducted after acquiring basic knowledge about the global CRO services market scenario through secondary research. Several primary interviews were conducted with market experts from the demand side, such as pharmaceutical and medical device companies, and experts from the supply side, such as C-level and D-level executives, and trial managers. These interviews were conducted across regions, namely, North America, Europe, the Asia Pacific, and the Rest of the World (including Latin America, the Middle East, and Africa). This primary data was collected through questionnaires, e-mails, online surveys, personal interviews, and telephonic interviews.
In the complete market engineering process, the top-down & bottom-up approaches were extensively used, along with several data triangulation methods, to perform the market estimation and market forecasting for the overall market segments and subsegments listed in this report. Extensive qualitative and quantitative analyses were performed on the complete market engineering process to present key information/insights throughout the report.
BREAKDOWN OF PRIMARIES
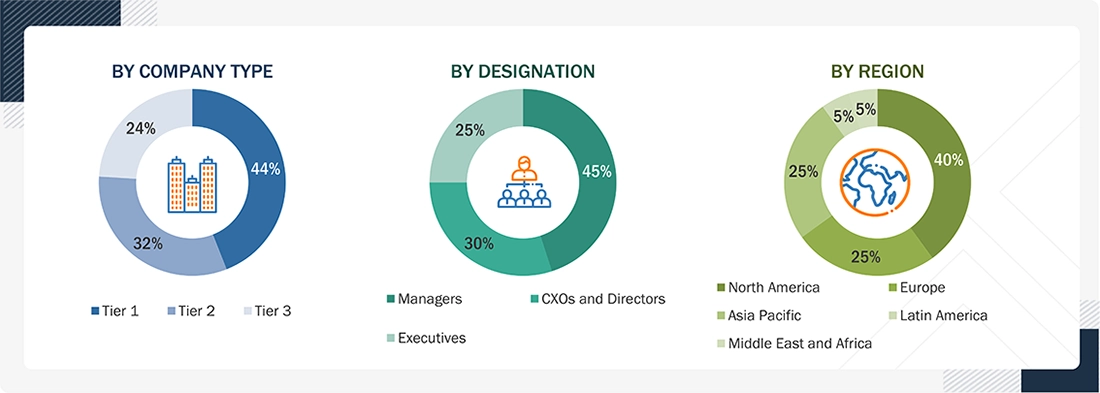
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
Both top-down & bottom-up approaches were used to estimate and validate the total size of the CRO services market. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
Bottom-up & Top-down Approach
The total market was divided into several segments and subsegments after determining the overall market size through estimation.

Data Triangulation
After arriving at the market size from the estimation process explained above, the total market was divided into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics for all segments and subsegments, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
CROs outsource research services to several pharmaceutical & biotechnology companies, medical device companies, and academic institutes. These services include clinical research, early-phase development, laboratory services, consulting, and data management. A sponsor (i.e., a company investigating the safety and efficacy of new drugs, therapies, or medical devices) partners with a CRO on a contractual or project-by-project basis. The CRO provides advice & guidance for planning, designing, and executing clinical trials; it offers a comprehensive and diverse range of services for each trial phase.
Key Stakeholders
- Contract Research Organizations (CROs)
- Clinical Trial Service Providers
- Pharmaceutical & Biopharmaceutical Companies
- Medical Device Manufacturing Companies
- Academic & Research Institutes
- Venture Capitalists & Investors
- Market Research & Consulting Firms
- Government Associations
- Medical Institutions & Universities
Report Objectives
- To define, describe, and forecast the CRO services market based on service type, therapeutic area, modality, delivery model, end user, and region
- To provide detailed information regarding the primary factors influencing market growth, such as drivers, restraints, opportunities, and challenges
- To strategically analyze the micromarkets concerning individual growth trends, prospects, and contributions to the overall CRO services market
- To analyze the opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders
- To analyze subsegments of the market concerning individual growth trends, prospects, and contributions to the overall market
- To forecast the revenue of the market segments concerning six regions, namely, North America (US and Canada), Europe (Germany, UK, France, Italy, Spain, and the Rest of Europe), the Asia Pacific (China, Japan, India, Australia, South Korea and the Rest of Asia Pacific), Latin America (Brazil, Mexico, and Rest of Latin America, the Middle East (GCC and Rest of Middle East), and Africa
- To profile the key market players and comprehensively analyze their market shares and core competencies
- To track & analyze competitive developments such as service launches, agreements, partnerships, acquisitions, business expansions, and research & development activities in the CRO services market.
Available customizations:
With the given market data, MarketsandMarkets offers customizations to meet the company’s needs. The following customization options are available for this report:
Company Information
- Detailed analysis & profiling of additional market players (up to three).
Geographic Analysis
- A further breakdown of the Rest of Asia Pacific’s CRO services market into countries
- A further breakdown of the Rest of Europe’s CRO services market into countries
- A further breakdown of the Rest of Latin America’s CRO services market into countries
- A further breakdown of the Rest of the Middle East’s CRO services market into countries
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Contract Research Organization (CRO) Services Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS
Growth opportunities and latent adjacency in Contract Research Organization (CRO) Services Market





















James
Jun, 2022
Which are the driving factors of the contract research organization services market?.
John
Jun, 2022
Who are the major players operating in the contract research organization services market?.
Kahlill
Jun, 2022
Which region will lead the global contract research organization services market?.
Emily
Jun, 2026
After going through the Contract Research Organization Services Market report, I found the shift toward strategic outsourcing and integrated service models particularly relevant for mid-sized pharma companies trying to optimize development timelines without expanding internal infrastructure..
Daniel
Jun, 2026
What stood out to me in the Contract Research Organization Services Market analysis is how clinical trial complexity and regulatory pressure are driving demand for specialized CRO capabilities, especially in areas like oncology and rare diseases where expertise directly impacts trial success rates..
Lucas
Jun, 2026
From an investment perspective, reviewing the Contract Research Organization Services Market gave me a clearer view of how consolidation trends and partnerships are shaping competitive positioning, making CROs with end-to-end offerings more attractive for long-term growth..