
Chemiluminescence Immunoassay Market Size, Growth, Share & Trends Analysis
Chemiluminescence Immunoassay Market by Product (Instruments, Consumables), Technology (CLEIA, ECLI, Microparticle CLIA), Sample Type (Blood, Saliva), Application (Oncology, Cardiology), End User (Hospital, Clinical Laboratory) - Global Forecast to 2031




OVERVIEW
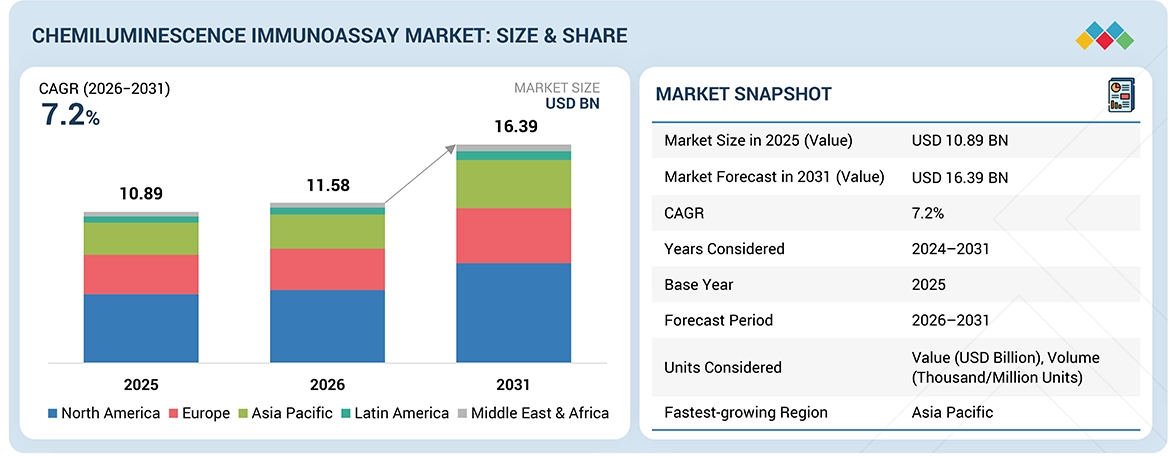
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
The Chemiluminescence Immunoassay market, valued at US$10.89 billion in 2024, stood at US$11.58 billion in 2025 and is projected to advance at a resilient CAGR of 7.2% from 2025 to 2031, culminating in a forecasted valuation of US$16.39 billion by the end of the period. The chemiluminescence immunoassay (CLIA) market is experiencing rapid growth because increasing rates of chronic and infectious diseases create a need for precise diagnostic tests. The implementation of automated laboratory systems combined with high-throughput analyzers, which enhance operational efficiency and decrease testing times, leads hospitals and diagnostic laboratories to adopt CLIA platforms. Market growth is supported by technological progress, including improved assay sensitivity, multiple testing capabilities, and integration of laboratory digital workflows. The increasing public knowledge about preventive healthcare, combined with the rise of screening initiatives and the development of healthcare systems in developing nations, has led to increased CLIA usage for both early disease diagnosis and standard medical testing.
KEY TAKEAWAYS
-
By RegionThe Asia Pacific market is expected to grow at the highest CAGR of 8.2% till 2031.
-
By Product TypeThe consumables segment is expected to register a CAGR of 7.6% during the forecast period.
-
By TechnologyThe chemiluminescence enzyme immunoassays (CLEIA) segment is expected to register strong growth during the forecast period.
-
By Sample TypeBy sample, the blood samples segment was projected to account for 64.1% of the market in 2025.
-
By ApplicationBy application, infectious diseases were projected to hold a share of 29.3% in 2025.
-
By End UserBy end user market, hospitals are expected to dominate the market.
-
Competitive Landscape - Key PlayersThe CLIA market is highly consolidated, dominated by key players such as F. Hoffmann-La Roche Ltd. (Switzerland), Abbott Laboratories (US), Siemens Healthineers (Germany), Danaher Corporation (US), and DiaSorin S.p.A. (Italy), which have extensive product portfolios and strong global presence.
-
Competitive Landscape - StartupsElabscience (China), Beijing Hotgen Biotech Co., Ltd. (China), and Shanghai Kehua Bio-Engineering Co., Ltd. (China) are some startups & SMEs in this market that specialize in CLIA devices.
The growth of the chemiluminescence immunoassay (CLIA) market is attributed to the rising incidence of chronic and infectious diseases, which creates a need for precise early diagnostic testing. The rising use of automated laboratory systems, which handle large testing volumes, brings better operational efficiency and faster test results to healthcare facilities that adopt CLIA testing systems. The testing process has become more dependable, and operational performance has improved because of continuous technological progress, which includes better sensitivity testing, multiplex assay capabilities, and digital system integration. The implementation of CLIA-based testing is increasing in developing countries because of expanding preventive healthcare programs, growing diagnostic screening initiatives, and better healthcare system development.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The CLIA market has seen technological advancements and evolving healthcare delivery methods. The healthcare industry experiences major disruptions due to the rapid adoption of automated systems that use AI to boost productivity, reduce errors, and improve analysis of test results. The development of miniature testing devices has enabled CLIA testing to reach emergency rooms, rural clinics, and mobile health units for point-of-care and immediate testing. The ability to conduct multiplex tests, which allows detection of multiple biomarkers simultaneously, has become a crucial development that aids research in oncology, autoimmune diseases, and infectious disease testing. Laboratories can now achieve greater operational efficiency by linking digital health systems, cloud services, and laboratory information systems. The increasing interest in precision medicine, together with biomarker-based diagnostic tests, has led to higher demand for high-sensitivity immunoassays, while laboratories use reagent rental systems and scalable platforms to control expenses and boost their operational capacity. The combination of these technological advancements and the transition to data-driven decentralized diagnostics systems transforms CLIA from a traditional laboratory instrument into a patient-focused diagnostic tool.
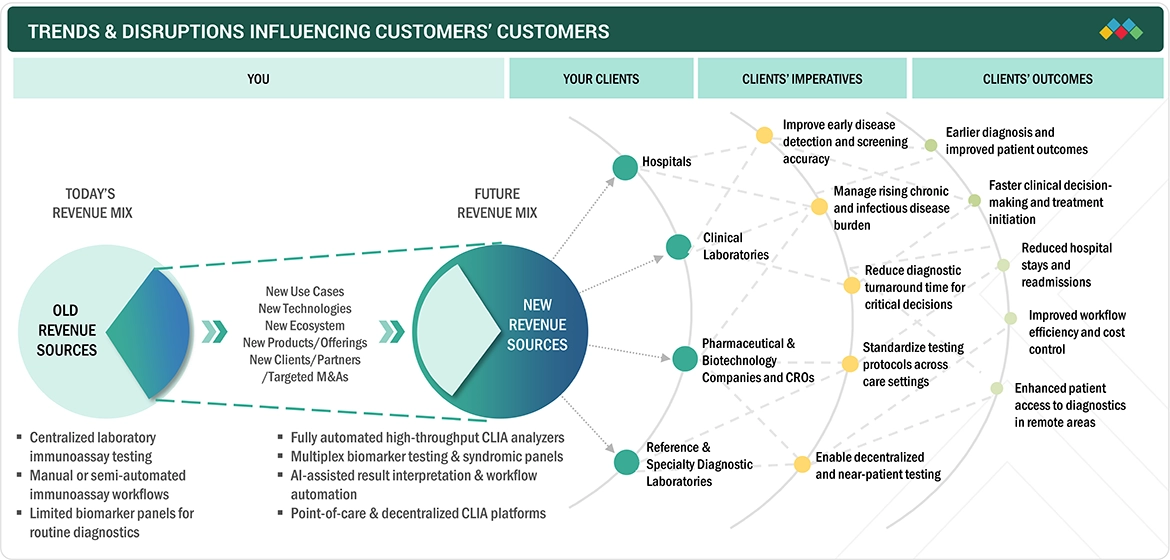
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Increasing incidences of chronic and infectious diseases globally

-
Advancements in improving chemiluminescence immunoassay technologies in recent years
Level
-
High cost of chemiluminescence systems and reagents
-
Lack of regular quality control procedures for monitoring and detecting cross-reactivity and interference
Level
-
High growth prospects for players in emerging economies
-
Increasing number of collaborations and partnerships
Level
-
Unfavorable reimbursement scenario and budgetary constraints in healthcare systems
-
Lack of skilled professionals and aging workforce
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Increasing incidences of chronic and infectious diseases
The increasing number of chronic and infectious diseases requiring precise and sensitive early diagnostic tests for effective management has become a key factor driving the growth of the chemiluminescence immunoassay market. The increasing incidence of cancers, along with cardiovascular diseases, thyroid disorders, diabetes, HIV, hepatitis, and respiratory infections, has created a greater requirement for biomarker-based testing and continuous health assessment. The CLIA technology offers high detection accuracy through its ability to identify disease markers, hormones, and infectious agents quickly. As healthcare systems expand and implement new screening programs, along with their focus on early diagnosis and disease surveillance, there has been greater use of CLIA platforms by laboratories and hospitals to enable quicker clinical decisions, which enhance patient results.
Restraint: High cost of chemiluminescence systems and reagents
The high cost of these assays and reagents restricts access for small laboratories, independent diagnostic centers, and medical facilities located in low- and middle-income countries. The financial requirements to acquire automated analyzers, together with their continuing costs for special reagents maintenance, calibration, and expert staff, create challenges for organizations with limited financial resources. The usage of reagent rental agreements and service contracts will result in increased expenses, which will discourage cost-sensitive users from adopting these services. The financial obstacles will prevent organizations from upgrading their technology, while they restrict the adoption of CLIA systems in resource-limited environments, which will result in slower market growth.
Opportunity: High growth prospects for players in emerging economies
Emerging economies provide new business opportunities through their developing healthcare systems, which enable people to access diagnostic tests. The healthcare industry is adopting advanced diagnostic technologies because of three factors, which include increased healthcare investments, expanded health insurance coverage, and government programs that develop laboratory networks. The need for dependable testing solutions that can handle large volumes of samples has increased because people now understand the importance of early disease detection and the rising prevalence of infectious and chronic diseases. The market players can enter price-sensitive markets through cost-effective analyzer models, reagent rental schemes, and localized manufacturing, which makes CLIA systems affordable and drives permanent market expansion.
Challenge: Unfavorable reimbursement scenario and budgetary constraints in healthcare systems
Unfavorable reimbursement scenarios create obstacles that prevent the chemiluminescence immunoassay (CLIA) market from growing because advanced diagnostic tests need continuous coverage, which healthcare providers find hard to adopt. In many regions, reimbursement rates may not fully cover the costs of CLIA testing, including reagents, maintenance, and laboratory operations, placing financial pressure on hospitals and diagnostic laboratories. The laboratory experience faces two main issues: complex billing procedures and delayed claim approvals, which create serious cash flow problems and operational efficiency gaps. The healthcare facilities face two obstacles, which include the need to invest in CLIA testing services and the need to expand those services. This situation causes market penetration to decrease even after the technology has demonstrated its clinical benefits.
CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Roche provides high-throughput CLIA platforms through its cobas e series system, which performs immunoassay testing for oncology, cardiology, infectious diseases, and endocrine disorders. The systems enable laboratories to run automated processes while testing a wide range of tests. | High sensitivity and specificity enable early disease detection and reliable monitoring. The combination of automation and standardized reagents with scalable throughput enables laboratories to achieve efficient testing operations and faster results in high-volume testing environments. |
 |
Danaher supplies CLIA-based immunoassay analyzers through its subsidiary, Beckman Coulter Diagnostics, including the Access and DxI series analyzers that detect cardiac markers and hormones, infectious diseases, and therapeutic drug levels. | Fast processing, combined with integrated automation systems, increases laboratory capacity, helping doctors make medical decisions. Advanced assay precision supports critical care diagnostics, while workflow integration reduces manual intervention and operational errors. |
 |
The Abbott ARCHITECT and Alinity i CLIA platforms provide immunoassay testing for infectious diseases, oncology markers, fertility, thyroid function, and cardiac biomarkers, supporting both standard and advanced diagnostic procedures. | The broad assay menu provides testing capabilities that cover all required tests through one testing system. The laboratory achieves greater operational efficiency through high-throughput capabilities and dependable system performance, which, combined with its strict quality control measures and standardized processes, deliver reliable clinical outcomes and better patient treatment management. |
 |
Siemens Healthineers offers CLIA-based systems through its ADVIA Centaur and Atellica IM analyzers, which provide automated immunoassay testing capabilities for endocrine, cardiac, infectious disease, allergy, and oncology testing applications. | Laboratory workflows become more efficient through the use of complete automation systems and digital network connections. The diagnostic process relies on analytical tests that deliver precise results, enabling medical professionals to determine a patient's health conditions. |
 |
DiaSorin provides CLIA testing services through its LIAISON platform, which offers testing for infectious diseases, autoimmune disorders, vitamin D levels, and specialized tests at both decentralized and expert laboratories. | The company's specialized testing abilities have helped develop high-value diagnostic tests for specific medical needs. The testing system allows for flexible operations because it can perform random-access tests and deliver accurate results for infectious diseases and autoimmune disorders, which helps doctors make clinical decisions. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The CLIA market functions within a multifaceted healthcare diagnostic system, which depends on various stakeholders who must work together. Instrument and reagent manufacturers lead the market because they develop essential components for testing, which include analyzers, reagents, and assay menus. Hospitals, reference laboratories, diagnostic chains, and research institutes represent the main market for these systems because they conduct tests at high volume, which results in increased reagent use. The US Food and Drug Administration and the Centers for Medicare & Medicaid Services control market entry through their regulatory processes, quality requirements, and reimbursement procedures, while distributors and service providers handle system setup, equipment upkeep, and product distribution. CLIA platforms serve as testing grounds for pharmaceutical and biotechnology companies, which use their capabilities to develop biomarker validation and companion diagnostic tests for their products. The CLIA ecosystem and value chain progression result from ongoing testing material requirements, government control, technological advancements, and the growth of laboratory networks in the industry.
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
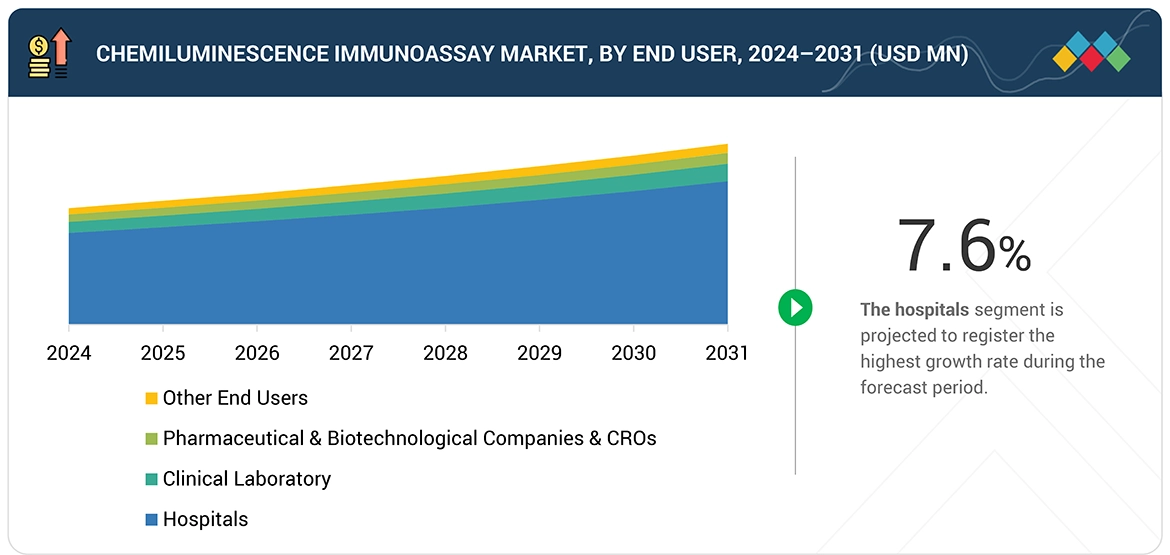
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Chemiluminescence Immunoassay Market, By Product Type
In 2025, consumables held the largest share in the CLIA market. The growth of the chemiluminescence immunoassay (CLIA) market is driven by increased demand for consumable products because hospitals and diagnostic laboratories perform high test volumes, creating ongoing needs for reagents, calibrators, controls, and reaction cartridges. The rising use of automated CLIA analyzers, along with expanded testing capabilities for infectious diseases, oncology, and endocrine disorders, results in higher reagent consumption. The combination of reagent-rental agreements with instrument–reagent bundling models fosters an environment where manufacturers gain predictable income through continuous product usage.
Chemiluminescence Immunoassay Market, By Technology
Chemiluminescence enzyme immunoassays (CLEIA) accounted for the largest share. The growth of the segment is driven by its superior sensitivity, rapid detection capability, and strong signal amplification, which make it suitable for detecting early disease symptoms and identifying biomarkers present at minimal levels. The rising need for automated high-throughput testing in infectious diseases, oncology, and endocrine disorders drives clinical laboratories to adopt CLEIA testing methods.
Chemiluminescence Immunoassay Market, By Sample Type
Based on sample type, the market is segmented into blood, saliva, urine, and other sample types. Blood samples account for the largest market share, as they offer dependable results and enable testing for various immunoassay procedures for cardiac markers, hormones, infectious diseases, and oncology biomarkers. The system achieves widespread use in clinical diagnostics because of its capability to deliver precise quantitative results through established blood-based testing procedures, which are supported by existing collection networks.
Chemiluminescence Immunoassay Market, By Application
Infectious diseases account for the largest share of the applications market. Infectious diseases are growing rapidly as an application of CLIA due to the rising global incidence of infections and the need for sensitive and early detection methods for diseases such as HIV, hepatitis, tuberculosis, and respiratory viruses. The testing method of CLIA provides essential components for disease surveillance and blood safety testing and outbreak management through its ability to detect diseases with high accuracy and its automated functions and its use in large-scale testing.
Chemiluminescence Immunoassay Market, By End User
Hospitals accounted for the largest market share in 2025. The CLIA market is experiencing growth in the hospitals sector because hospitals need their emergency departments to provide high-speed diagnostic tests, which can handle multiple tests at once, and their medical staff needs to monitor patients, and their doctors need to conduct regular disease tests. The implementation of CLIA standards in hospital laboratories is being driven by three factors, which include the rising use of automated immunoassay analyzers and their expanding testing capabilities, and the requirement for precise and fast test outcomes that support clinical choices.
REGION
Asia Pacific to register the highest growth in the CLIA market during the forecast period.
Healthcare facilities in APAC countries like China, India, and Japan are seeing rapid growth due to their high healthcare investments and efforts to make diagnostic services more available. The demand for precise and timely diagnostic tests is rising because people increasingly suffer from chronic medical conditions, infectious diseases, and health problems. Government programs that build laboratory networks, expand universal health coverage, and establish disease-testing facilities accelerate the adoption of automated immunoassay systems. The public now understands preventive healthcare better, while private diagnostic chains expand their services and local companies produce affordable CLIA reagents and analyzers. This combination creates a market that grows rapidly, positioning APAC as one of the most dynamic regional markets.

CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMPANY EVALUATION MATRIX
The CLIA market demonstrates intense rivalry between major diagnostic companies, which include F. Hoffmann-La Roche Ltd., Abbott Laboratories, Siemens Healthineers, Danaher Corporation, and DiaSorin S.p.A. Roche maintains a strong market position due to its extensive immunoassay menu, high-throughput cobas platforms, and its global installed base. Abbott and Siemens Healthineers compete through automation and workflow integration, and their extensive test portfolios, which meet hospital and reference laboratory requirements. Danaher, through Beckman Coulter Diagnostics, emphasizes precision diagnostics and scalable automation solutions, while DiaSorin differentiates itself through specialty immunoassays and strong expertise in infectious and autoimmune disease testing. The global CLIA market continues to evolve as companies focus on innovation, menu expansion, strategic partnerships, and efforts to enter new markets.
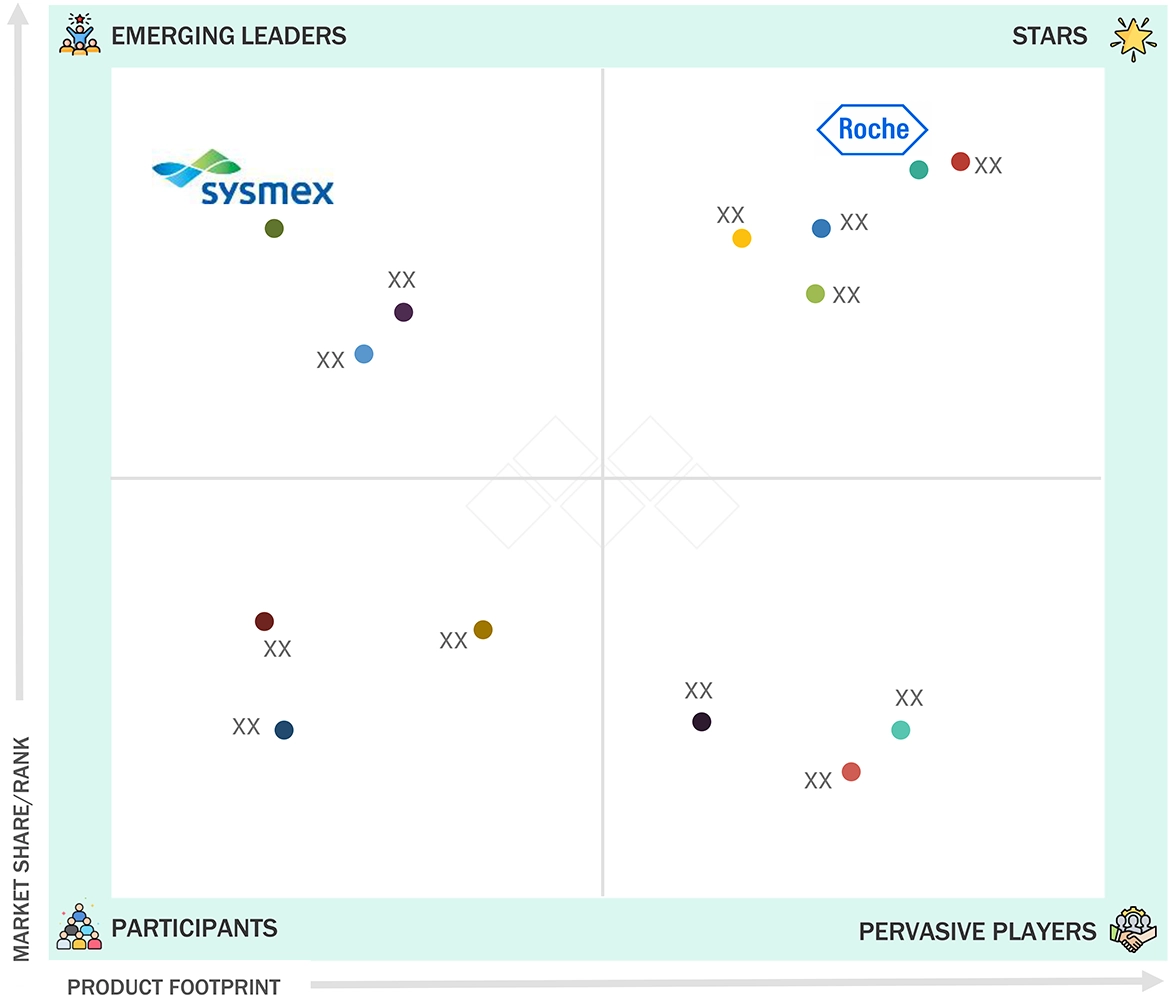
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- F. Hoffmann-La Roche (Switzerland)
- Danaher Corporation (US)
- Siemens Healthineers AG (Germany)
- DiaSorin S.p.A. (Italy)
- Abbott Laboratories (US)
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (China)
- Werfen (Spain)
- Sysmex Corporation (Japan)
- QuidelOrtho Corporation (US)
- HUMAN Gesellschaft für Biochemica und Diagnostica (Germany)
- EUROIMMUN Medizinische Labordiagnostika AG (Germany)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 10.89 Billion |
| Market Forecast in 2031 (Value) | USD 16.39 Billion |
| Growth Rate | CAGR of 7.2% from 2026–2031 |
| Years Considered | 2024–2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Billion), Volume (Thousand/Million Units) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Regions Covered | North America, Asia Pacific, Europe, Latin America, Middle East & Africa |
WHAT IS IN IT FOR YOU: CHEMILUMINESCENCE IMMUNOASSAY MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Product Analysis | The evaluation investigated all system components of CLIA through testing of immunoassay analyzers, all associated reagents, calibrators, and controls, and clinical diagnostics consumables. The analysis evaluated technological progress through assessments of high-throughput automation, improved chemiluminescent signal detection, multiplex testing, AI-based workflow optimization, and laboratory information system connectivity. The analysis examined how top manufacturers use regulatory pathways, quality standards, assay-sensitivity enhancements, and product-differentiation methods to develop innovative products and establish performance standards. | Healthcare providers and stakeholders gained clarity on selecting high-performance CLIA platforms by correlating assay accuracy, turnaround time, throughput, and workflow efficiency with clinical outcomes. The insights enabled organizations to optimize their portfolio through technology adoption while aligning with preventive screening, precision diagnostics, and decentralized testing trends, which enhanced their diagnostic reliability and operational efficiency. |
| Company Information | The research analyzed global and regional CLIA manufacturers, including F. Hoffmann-La Roche Ltd., Abbott Laboratories, Siemens Healthineers, Danaher, Beckman Coulter Diagnostics, and DiaSorin S.p.A. It evaluated their operations, product range, testing services, technologies, market presence, research projects, authorizations, production capacity, alliances, and product releases. The study also examined competitor positioning in hospital labs, reference labs, and diagnostic areas, as well as recent partnerships, acquisitions, and new product development. | The analysis showed how competition operated in CLIA testing environments, which technologies led the industry, and which partnerships formed the testing ecosystem. Clients were able to identify collaboration opportunities, acquisition targets, distribution partnerships, and differentiation strategies in high-growth areas such as infectious disease diagnostics, oncology biomarkers, and specialty immunoassays. |
| Geographic Analysis | The CLIA market was examined across North America, Europe, Asia Pacific, Latin America, the Middle East, and Africa. The assessment considered disease burden, diagnostic testing volumes, healthcare infrastructure maturity, reimbursement environments, regulatory frameworks, and levels of laboratory automation adoption. The study assessed regional manufacturing hubs and local reagent production, expanding diagnostic chains, and government screening programs, in addition to conducting country-level deep dives and global benchmarking. | The regional analysis results shared with strategic planners showed which markets would experience the most rapid growth, which market demand centers, which reimbursement policies, and which expansion paths. The clients received support to develop their market entry plans, improve their distribution systems, choose their main healthcare partners, and exploit the increasing need for advanced immunodiagnostic tests in both developing and established markets. |
RECENT DEVELOPMENTS
- October 2025 : Roche announced that it has received the CE mark for its Elecsys Dengue Ag test, a high-throughput, fully automated immunoassay intended as an aid in the diagnosis of acute dengue virus infection.
- November 2025 : Abbott acquired cancer test maker Exact Sciences (EXAS.O) in a deal valued at up to USD 23 billion, including debt, marking one of its largest purchases in nearly a decade and a bold entry into the fast-growing cancer diagnostics market.
- December 2025 : ALZpath, Inc., a leading developer of innovative diagnostic tools for Alzheimer's disease and related dementias, announced a licensing agreement with Siemens Healthineers for the use of its proprietary pTau217 antibody in the development of a pTau217 assay in the expanding menu of Siemens Healthineers in vitro diagnostic (IVD) tests for Alzheimer's disease.
Table of Contents

Methodology
This study involved four major activities in estimating the size of the CLIA market. Exhaustive secondary research was done to collect information on the market, peer market, and parent market. The next step was to validate these findings, assumptions, and sizing with industry experts across value chains through primary research. The bottom-up approach was employed to estimate the overall market size. After that, market breakdown and data triangulation were used to estimate the market size of segments and sub-segments.
Secondary Research
The secondary research process involved the widespread use of secondary sources, directories, databases (such as Bloomberg Businessweek, Factiva, and D&B Hoovers), white papers, annual reports, company house documents, investor presentations, and companies' SEC filings. Secondary research was used to identify and collect information useful for the extensive, technical, market-oriented, and commercial study of the CLIA market. It was also used to obtain important information on key players, market classification, and segmentation aligned with industry trends to the bottom-most level, and key developments from market and technology perspectives. A database of the key industry leaders was also prepared using secondary research.
Primary Research
Extensive primary research was conducted after secondary research provided information on the CLIA market. Several primary interviews were conducted with market experts from both the demand and supply sides across major countries of North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. Primary data was collected through questionnaires, emails, and telephonic interviews. The primary sources from the supply side included various industry experts, such as Chief Executive Officers (CXOs), Vice Presidents (VPs), Directors from business development, marketing, and product development/innovation teams, as well as related key executives from manufacturers and distributors operating in the CLIA market.; and key opinion leaders.
Primary interviews were conducted to gather insights into market statistics, revenue from products and services, market breakdowns, market size estimates, market forecasting, and data triangulation. Primary research also helped in understanding the various trends in technology, applications, verticals, and regions. Stakeholders from the demand side, customers/end users who are using infection control products, were interviewed to understand the buyer’s perspective on the suppliers, products, and their current usage and the future outlook of their business, which will affect the overall market.
The following is a breakdown of the primary respondents:
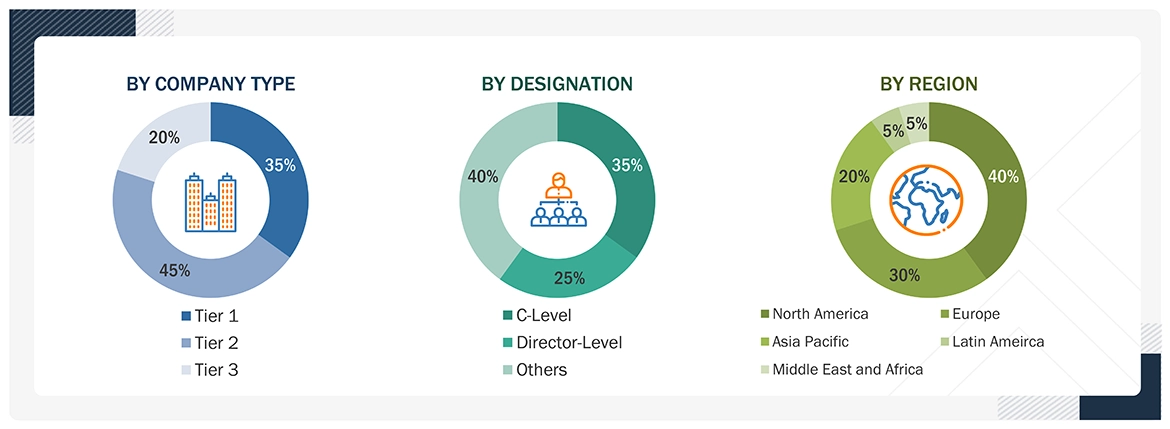
Note 1: C-level primaries include CEOs, COOs, CTOs, and VPs.
Note 2: Other primaries include sales managers, marketing managers, and product managers.
Note 3: Companies are classified into tiers based on their total revenue.
As of 2023: Tier 1=>USD 1 billion, Tier 2 = USD 500 million to USD 1 billion, Tier 3=<USD 500 million.
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The research methodology used to estimate the size of the CLIA market includes the following details.
The market sizing was conducted on a global basis.
Country-level Analysis: The size of the CLIA market was obtained from the annual presentations of leading players and secondary data available in the public domain. The share of products in the overall CLIA market was obtained from secondary data and validated by primary participants to arrive at the total CLIA market. Primary participants further validated the numbers.
Geographic market assessment (by region & country): The geographic assessment was done using the following approaches:
Approach 1: Geographic revenue contributions/splits of leading players in the market (wherever available) and respective growth trends
Approach 2: Geographic adoption trends for individual product segments by end user and growth prospects for each of the segments (assumptions and indicative estimates validated from primary interviews)
At each point, the assumptions and approaches were validated through industry experts contacted during primary research. Considering the limitations of data available from secondary research, revenue estimates for individual companies (for the overall CLIA market and geographic market assessment) were ascertained based on a detailed analysis of their respective product offerings, geographic reach/strength (direct or through distributors or suppliers), and the shares of the leading players in a particular region or country.
Global Chemiluminescence Immunoassay Market : Top-Down and Bottom-Up Approach

Data Triangulation
The market was split into several segments and subsegments after determining the overall market size, using the market sizing processes. Data triangulation and market breakdown procedures were employed to complete the overall market engineering process and determine the exact statistics for each market segment and subsegment. The data was triangulated by examining various factors and trends on both the demand and supply sides of the CLIA market.
Market Definition
Chemiluminescent immunoassay (CLIA) is an immunoassay technique in which the label, i.e., the indicator of the analytical reaction, is a luminescent molecule. CLIA is a variation of the standard enzyme immunoassay (EIA), a biochemical technique used in immunology. This variation technique can be used for medical diagnosis and in several other industries for various applications. In recent years, CLIA has attracted increasing attention across fields such as clinical diagnosis, life sciences, food safety, environmental monitoring, and pharmaceutical analysis, due to its high specificity, high sensitivity, broad applicability, simple instrumentation, and wide linear range.
Key Stakeholders
- CLIA manufacturers and distributors
- CLIA analyzers & consumables manufacturers
- Healthcare institutions (hospitals, medical schools, group practices, individual surgeons, and governing bodies)
- Healthcare institutions (hospitals and outpatient clinics)
- Distributors and suppliers
- Research institutes
- Health insurance payers
- World Health Organization (WHO)
- Organisation for Economic Co-operation and Development (OECD)
- National Institutes of Health (NIH)
- Centers for Disease Control and Prevention (CDC)
- Annual Reports/SEC Filings, Investor Presentations, and Press Releases of Key Players
- White Papers, Journals/Magazines, and News Articles
- Paid Databases, such as Factiva, D&B Hoovers, and Bloomberg Business
Report Objectives
- To define, describe, segment, and forecast the global CLIA market by product type, technology, sample type, application, end user, and region.
- To provide detailed information about the factors influencing market growth (such as drivers, restraints, opportunities, and challenges)
- To analyze micromarkets concerning individual growth trends, prospects, and contributions to the overall CLIA market.
- To analyze market opportunities for stakeholders and provide details of the competitive landscape for key players.
- To forecast the size of the CLIA market in five main regions, along with their respective key countries, namely, North America, Europe, the Asia Pacific, Latin America, and the Middle East & Africa
- To profile key players in the global CLIA market and comprehensively analyze their core competencies and market shares.
- To track and analyze competitive developments such as acquisitions, expansions, partnerships, agreements, and collaborations; and product launches and approvals.
- To benchmark players within the CLIA market using the "Competitive Leadership Mapping" framework, which analyzes market players on various parameters within the broad categories of business and product strategy.
Available customizations:
Based on the given market data, MarketsandMarkets offers customizations tailored to the company’s specific needs. The following customization options are available for the report:
Geographic Analysis
- Further breakdown of the Rest of Asia Pacific CLIA market into Thailand, Indonesia, Philippines, Vietnam, Hong Kong, and other countries
- Further breakdown of the Rest of Europe CLIA market into Belgium, Russia, the Netherlands, Switzerland, and other countries.
- Further breakdown of the Rest of Latin America CLIA market into Argentina, Peru, and other countries.
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Chemiluminescence Immunoassay Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS




















Growth opportunities and latent adjacency in Chemiluminescence Immunoassay Market