
Medical Adhesives Market
Medical Adhesives Market by Technology (Water-Based, Solvent-Based, Solid & Hot Melt Based), Resin Type (Natural Resin, Synthetic & Semi-Synthetic Resin), Application (Dental, Surgery, Medical Device & Equipment), And Region - Global Forecast to 2031




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
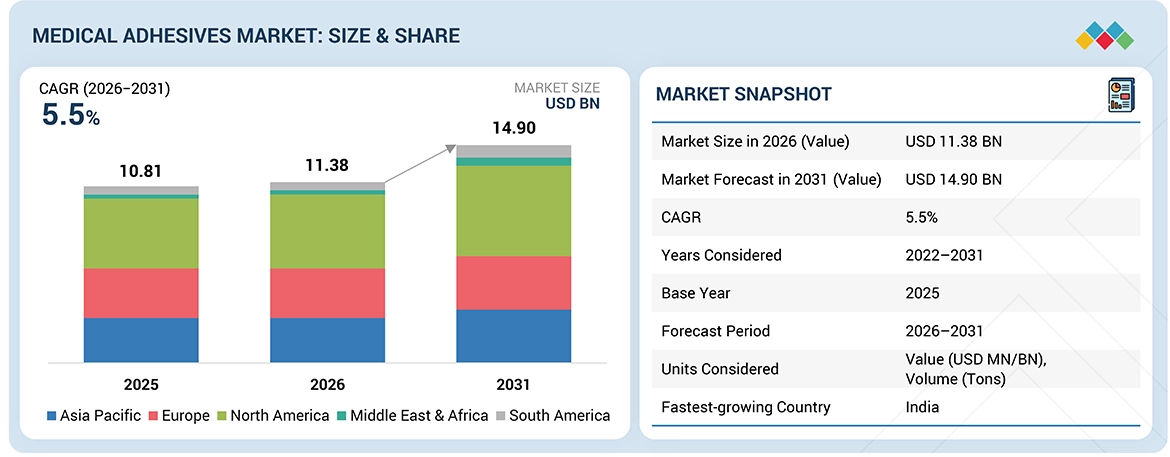
The medical adhesives market is projected to grow from USD 11.38 billion in 2026 to USD 14.90 billion in 2031, registering a CAGR of 5.5% during the forecast period. The demand for medical adhesives has been increasing due to the rising need for minimally invasive surgical procedures and advanced wound treatment methods, as well as the growing adoption of wearable medical technologies and systems that deliver drugs through the skin. The essential properties of adhesives, including strong bonding performance, biocompatibility, flexibility, and moisture resistance, make these materials ideal for use in vital medical fields that require components for surgical procedures, medical device manufacturing, and extended skin-contact applications. The market experiences accelerated growth due to three factors: the increase in chronic disease cases, the expansion of the elderly population, and the rise in healthcare spending across developing markets. The worldwide healthcare industry adopts advanced adhesive technologies because strict regulations require healthcare organizations to maintain patient safety standards and infection control measures and demonstrate their products meet quality standards.
KEY TAKEAWAYS
-
By RegionNorth America dominated the medical adhesives market, with a 36.8% share in terms of value in 2025.
-
By TechnologySolids & hot melt dominates the market, by technology, with a share of 62.15%, in terms of value, in 2024.
-
By Resin TypeBased on resin type, cyanoacrylate is the fastest-growing segment with a CAGR of 8.9% during the forecast period.
-
By ApplicationThe surgery segment holds the highest market share of 46.9%, in terms of value, during the forecast period.
-
COMPETITIVE LANDSCAPE- KEY PLAYERSCompanies such as Solventum, Henkel AG & Co. KGAA, and H.B. Fuller Company were identified as star players in the market, given their strong market share and product portfolios.
-
COMPETITIVE LANDSCAPE- STARTUPSMasterbond and Vivostat A/S have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas, underscoring their potential as emerging market leaders.
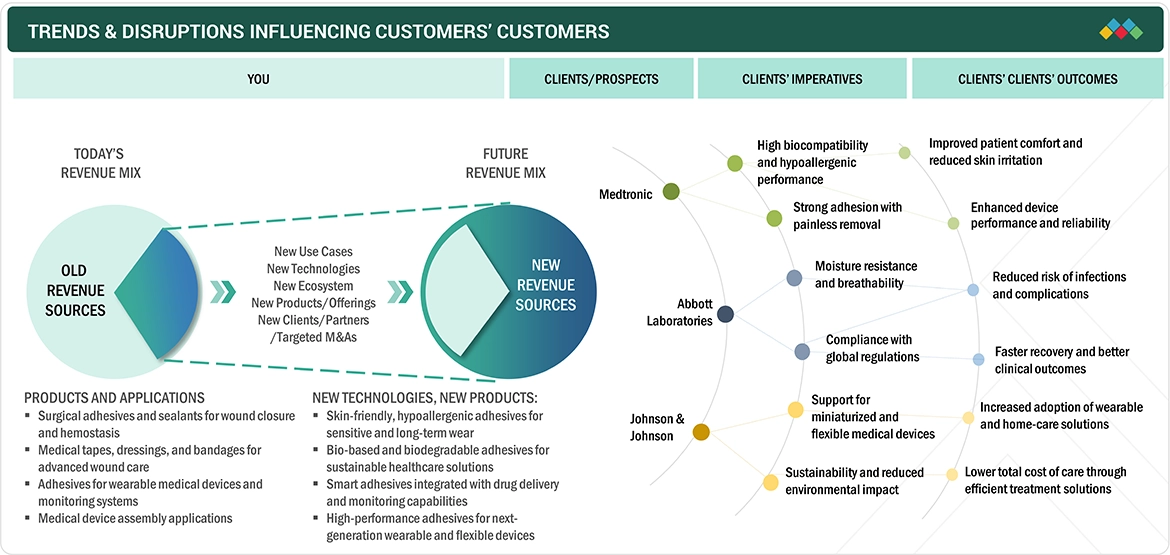
The medical adhesives market is steadily advancing, driven by the rising demand for advanced healthcare solutions and the rapid expansion of the medical devices and wound care sectors. As surgical procedures increase and the use of medical devices, including transdermal drug delivery systems and wearable medical devices, grows, these three critical elements drive market expansion. Hospitals achieve better product performance through their ongoing healthcare infrastructure funding, and medical device companies work with adhesive manufacturers to ensure patient comfort and safety. Researchers are currently developing new adhesive formulas that will provide biocompatible and skin-friendly performance and extended durability. The medical industry is driven by two factors: sustainable, low-toxicity product development and strict medical-grade product regulations.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The medical adhesives market is evolving through advanced wound care and wearable medical devices and minimally invasive surgical procedures, which are now using these materials because of technological advancements that include better formulation methods, biocompatibility improvements, and the creation of pressure-sensitive and bio-based adhesives that perform at high levels. The demand for drug delivery systems using transdermal patches has increased due to these advancements. The market changes due to strict regulatory standards that protect patient safety and product effectiveness, the increasing demand for skin-safe and hypoallergenic products, and the industry shift toward low-VOC, sustainable adhesive solutions. The healthcare market progresses through supply chain improvements and increased healthcare research funding and the creation of durable strong adhesives which can be easily detached from surfaces.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Rising demand for minimally invasive surgeries

-
Growth in advanced wound care and wearable devices
Level
-
Stringent regulatory approvals
-
Risk of skin irritation and allergic reactions
Level
-
Increasing demand for bio-based and sustainable adhesive
-
Expansion in emerging healthcare markets
Level
-
Complex product development and high R&D costs
-
Performance limitations in extreme conditions
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Rising demand for minimally invasive surgeries processing industries
The demand for advanced wound care solutions together with minimally invasive surgical procedures drives the medical adhesives market. Healthcare systems select medical adhesives because they help reduce patient recovery time while minimizing scarring and decreasing infection risks which traditional sutures and staples cannot provide. The product exists as an ideal choice for contemporary medical environments because it enables doctors to successfully close wounds while delivering patients better comfort and speedier recovery results. The demand for strong and skin-compatible adhesives reached new heights because of the quick growth of wearable medical devices which includes continuous glucose monitors and cardiac patches. The increasing rates of chronic illnesses such as diabetes and heart disease have created a need for adhesive-based long-term medical care solutions. The healthcare sector sees increased spending which creates a greater need for innovative adhesive solutions that result from healthcare facilities implementing new medical devices and the development of new adhesive technologies. The medical adhesives market continues to expand because of these elements which work together with better biocompatibility and improved moisture resistance and flexible product design.
Restraint: Stringent regulatory approvals
The medical adhesives market faces multiple restraints which will prevent it from achieving its potential growth. The medical-grade product market faces its biggest challenge because of its strict rules which control all aspects of product development. Healthcare adhesives must achieve both safety requirements and biocompatibility standards and performance benchmarks which regulatory authorities established. This situation presents particular difficulties for businesses operating at small and medium size because they lack sufficient financial backing. The development of certain adhesive products leads to heightened risk of skin irritation and allergic responses and cytotoxicity which poses particular challenges for patients who have delicate skin or who need to use adhesive products over extended periods. Brand reputation and market acceptance face further risks from product recalls which occur when adverse events are reported. Advanced medical adhesives require higher financial investment than traditional wound closure methods which creates a barrier for their use in budget-restricted healthcare systems found in developing areas. The factors mentioned above create obstacles which prevent the widespread acceptance of products and which cause delays in their development and marketing activities within the industry.
Opportunity: Increasing demand for bio-based and sustainable adhesive
The medical adhesives market creates multiple business opportunities because material science developments and healthcare industry needs combine to open fresh business paths. The market opportunity exists because bio-based adhesives were developed to create products which biodegrade while meeting international environmental standards and current market requirements.Manufacturers allocate research investments to develop adhesive products which achieve high performance standards while guaranteeing safety protection for users and environmental safety. The growing demand for home healthcare solutions and remote patient monitoring systems has generated new markets for adhesive technologies which medical wearable devices use to stay attached to skin for long periods. The Asia Pacific and Latin America and Middle East markets show strong growth potential because healthcare systems improve while people learn about modern medical treatments and increase their spending. The market will gain new business opportunities through technological progress which involves the development of smart adhesives that provide medication delivery and environmental protection functions. Market growth will occur during the next years because strategic partnerships between healthcare providers and manufacturers will work together with recent industry developments.
Challenge: Complex product development and high R&D costs
The medical adhesives market needs constant innovation together with strategic planning because it faces multiple challenges. The development of adhesives which need to fulfill multiple clinical needs through their ability to stick to surfaces and their flexible and breathable properties and their easy removal needs to be solved because it creates major difficulties which must be addressed to ensure patient safety. The industry faces challenges in developing solutions that function effectively on various skin types and different environmental conditions and through multiple usage periods. The main problem in this situation involves finding adhesive solutions that maintain their strength while enabling users to remove them without causing skin damage which poses challenges for both elderly patients and young children. Manufacturers face production and pricing challenges because supply chain disruptions and raw material availability fluctuations create market uncertainty. Market players must invest continuously in research and development because of the high competition level which results in financial strain for their organizations. The organization faces operational difficulties because it needs to meet changing regulatory requirements which differ between various regions. The solution to these challenges needs the development of new formulation technologies together with better testing procedures and a commitment to designing products that prioritize patient needs.
MEDICAL ADHESIVES MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
A leading provider of medical adhesive solutions for wound care, surgical applications, and wearable medical devices, offering advanced pressure-sensitive adhesives, tapes, and dressings designed for long wear and patient comfort. | Strong skin adhesion with gentle removal, high breathability, reduced risk of skin damage, and reliable performance in long-term wearable applications and clinical environments. |
 |
Develops high-performance medical-grade adhesives for device assembly, wound closure, and drug delivery systems, leveraging technologies such as light-curing, cyanoacrylates, and acrylic-based adhesives. | Excellent bonding strength across substrates, high precision in device assembly, sterilization resistance, and enhanced durability for critical healthcare applications. |
 |
Offers a broad portfolio of medical adhesive technologies for applications such as wound closure, medical device assembly, filtration systems, PPE, and skin-contact wearables, including cyanoacrylate and pressure-sensitive adhesives. (hbfuller.com) | High biocompatibility, strong adhesion to diverse substrates, support for sterilization processes, and reliable performance in both skin-contact and structural medical applications. |
 |
Provides medical adhesive tapes, films, and advanced wound care solutions used in diagnostics, wearable devices, and long-term patient monitoring applications, focusing on customized healthcare materials. | Skin-friendly adhesion, flexibility for conformability, moisture management, and suitability for extended wear in sensitive applications. |
 |
Through its medical devices segment (including Ethicon), develops surgical adhesives and topical skin adhesives for wound closure, infection prevention, and post-operative care. | Rapid wound closure, microbial barrier protection, reduced need for sutures, improved patient comfort, and enhanced healing outcomes in surgical settings. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The medical adhesives market ecosystem is structured around raw material suppliers and specialty chemical manufacturers that produce key components which include acrylics and silicones and polyurethanes and cyanoacrylates used in medical-grade formulations. The adhesive manufacturers use these materials to create multiple products which include pressure-sensitive adhesives and surgical glues and tapes and sealants and coatings that people use for wound care and medical device assembly and wearable applications. The finished products of converters and medical product manufacturers use these adhesives which include dressings and transdermal patches and surgical drapes and diagnostic devices that need high biocompatibility and flexibility and durability. Distributors and healthcare suppliers play a critical role in ensuring global availability while they provide regulatory compliance support and deliver products to hospitals and clinics on time. North America and Europe and Asia Pacific function as main centers for production and consumption because their healthcare systems are advanced and their regulatory systems are strong and their research and development activities for patient-friendly adhesive technologies receive continuous funding.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Medical Adhesives Market, By Technology
The medical adhesives market divides into three segments which include water-based adhesives and solvent-based adhesives and solid & hot melt adhesives. Water-based adhesives lead the market because they provide excellent biocompatibility and low toxicity and produce minimal volatile organic compound emissions which make them suitable for skin-contact and wound care applications. Medical tapes and dressings and wearable devices use these adhesives because they ensure patient safety and comfort during use. The bonding strength and durability of solvent-based adhesives remain strong yet their market growth continues to decline because of environmental regulations which restrict solvent emissions. Solid and hot melt adhesives now see increased usage for medical device assembly and hygiene purposes because they provide fast curing times and simple processing methods and effective adhesion to multiple materials. The water-based adhesive market exists as the dominant force because rising regulations mandate environmentally friendly products which lack toxic components and because healthcare demands require both safe and effective solutions.
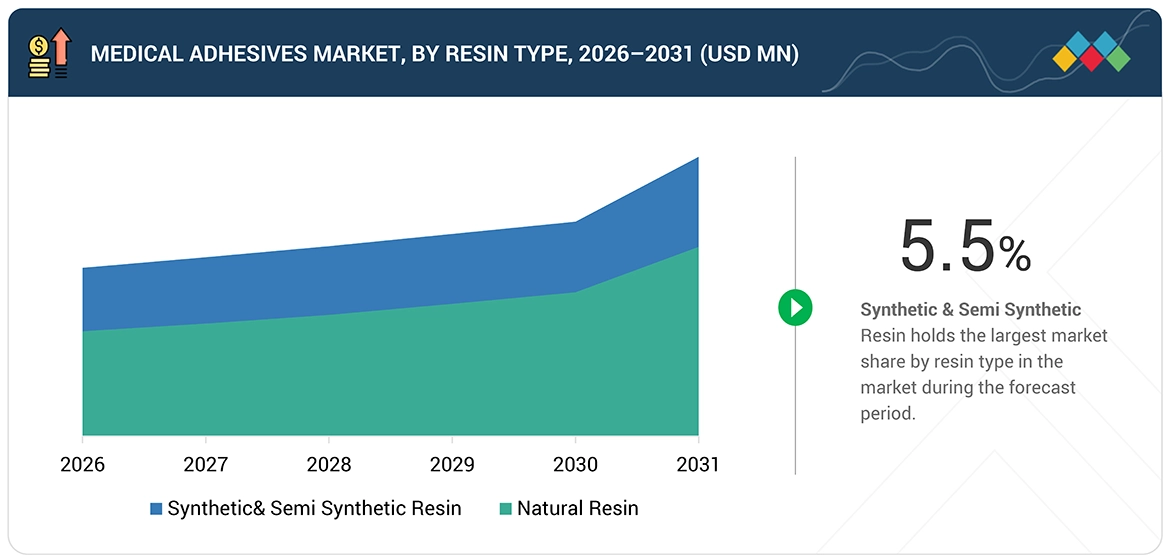
Medical Adhesives Market, By Resin Type
The medical adhesives market is categorized into three segments: fibrin, collagen, and other medical adhesive products. Fibrin-based adhesives maintain the largest market share because they provide better biocompatibility and function as natural components of the body's healing process. Fibrin adhesives, derived from human or animal plasma components, provide a complete blood coagulation system that surgeons use for hemostasis, tissue sealing, and wound closure. The medical community uses these products because they help patients heal faster while controlling blood loss and decreasing the chances of negative health effects during cardiovascular, orthopedic, and general surgical operations. Collagen-based adhesives are becoming more popular because they provide excellent tissue compatibility which helps cells to grow especially in wound care and regenerative medicine fields. Specialized applications which need strong bonding and controlled degradation use synthetic adhesives which include cyanoacrylates and polyethylene glycol (PEG)-based formulations as their preferred resin types. Fibrin adhesives represent the preferred choice because they provide safe usage, effective results in emergency medical situations and growing demand for biologically based medical solutions in contemporary healthcare.
Medical Adhesives Market, By Application
The medical adhesives market is divided into three segments based on its applications: dental, surgical, and medical device and equipment uses, as well as other unidentified uses. The surgical segment dominates the market because it requires adhesives for wound closure and hemostasis, as well as tissue sealing, across various surgical operations. Medical adhesives are increasingly preferred over traditional sutures and staples because they allow faster operations and create less scarring and lower infection risks, and they improve patient comfort and recovery outcomes. Their extensive use in cardiovascular, orthopedic, general, and cosmetic surgeries further strengthens their dominance in this segment. Dental applications also contribute significantly because dentists use adhesives for bonding, restorations, and orthodontic treatment. The medical device and equipment market expands because people need biocompatible bonding solutions to connect their growing collection of wearable devices, diagnostic tools, and disposable medical items. The company provides additional applications through its development of wound care dressings and transdermal drug delivery systems. The surgical segment maintains its dominance because surgical procedures have increased and adhesive technologies have advanced and less invasive surgical methods have become more popular.
REGION
Asia Pacific to be fastest-growing region during forecast period
The medical adhesives market experiences its highest growth rate in Asia Pacific because of three factors: healthcare infrastructure expansion, increased medical tourism, and growing healthcare spending in China, India, and Southeast Asian countries. The region needs advanced wound care and surgical procedures because its increasing number of elderly people and rising chronic disease cases create a higher requirement for medical adhesives. The market continues to expand due to increasing use of wearable medical devices and rising public knowledge about minimally invasive medical procedures. Regional growth accelerates through three factors which include government support and better regulatory systems and manufacturing and research development investments from both international and domestic companies. Asia Pacific serves as a preferred center for medical adhesive production and advancement because it offers lower manufacturing expenses and access to training facilities for professional workers.

MEDICAL ADHESIVES MARKET: COMPANY EVALUATION MATRIX
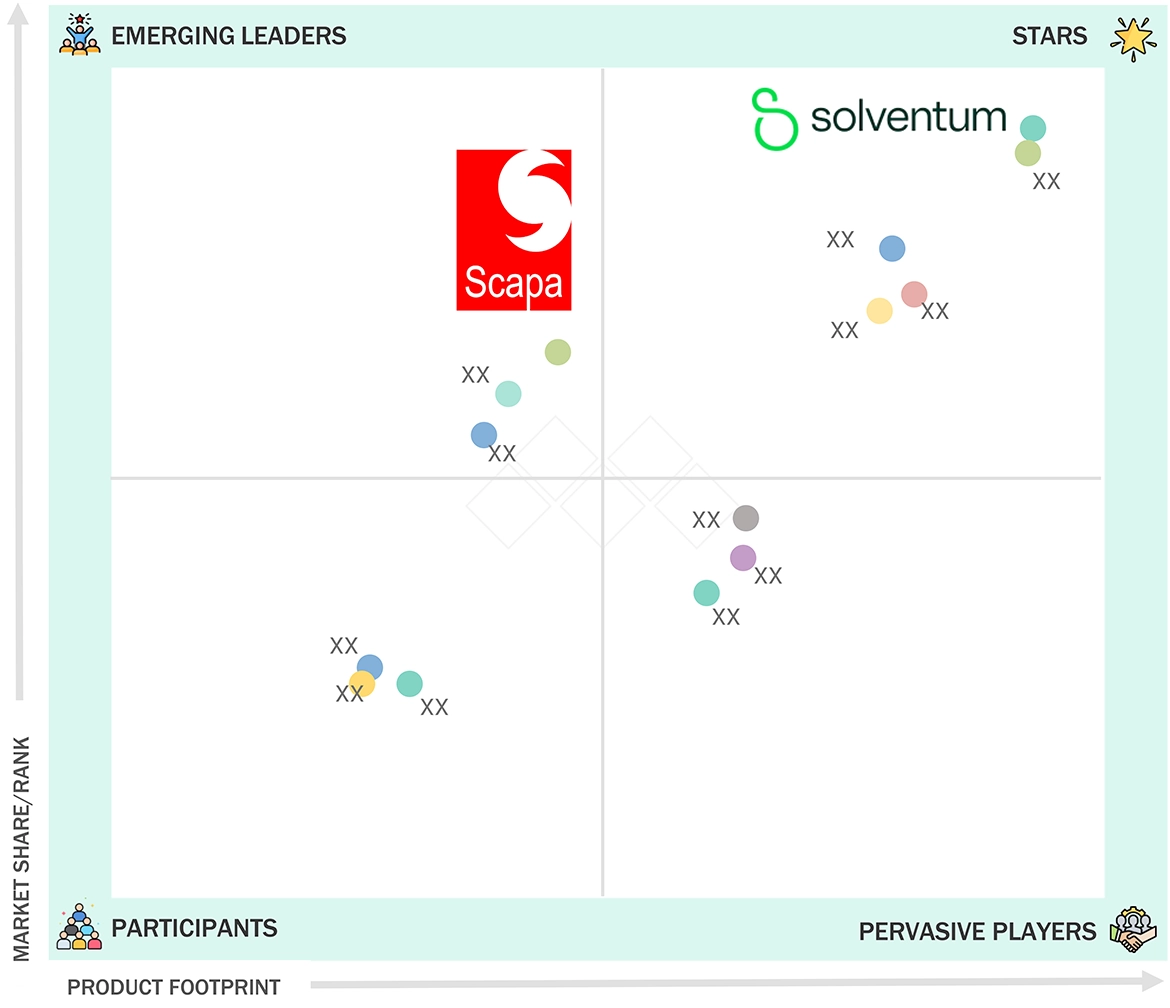
Solventum (Star) holds a leading position in the global medical adhesives market through its extensive range of innovative adhesive products which include pressure-sensitive adhesives and surgical tapes and wound care products. The company benefits from its powerful research and development resources together with its ongoing development of skin-compatible long-lasting adhesives and its cooperative partnerships with medical device manufacturers. The company establishes its market dominance through its international operations and emphasis on high-performance biocompatible materials which support multiple uses in wearable technology and surgical operations and sophisticated wound treatment methods. Scapa Healthcare (Emerging Player) is gaining prominence through its expertise in customized healthcare adhesive solutions, particularly for wearable devices and skin-contact applications. The company develops adhesive technologies which focus on hypoallergenic properties and patient needs and environmental sustainability. The company experiences growth through its increasing research and development investments while establishing strong relationships with medical device manufacturers which help it compete in the medical adhesives market as an upcoming player.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Solventum (US)
- Henkel AG & Co. KGAA (Germany)
- H.B. Fuller Company (US)
- Scapa Healthcare (US)
- Johnson & Johnson (US)
- Permabond (UK)
- B.Braun (Germany)
- Chemence Medical Inc (US)
- Artivion (US)
- Dymax (US)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2026 (Value) | USD 11.38 Billion |
| Market Forecast in 2030 (value) | USD 14.90 Billion |
| Growth Rate | CAGR of 5.5% from 2026-2031 |
| Years Considered | 2022-2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Million/Billion), Volume (Tons) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Regions Covered | North America, Asia Pacific, Europe, South America, Middle East & Africa |
WHAT IS IN IT FOR YOU: MEDICAL ADHESIVES MARKET REPORT CONTENT GUIDE
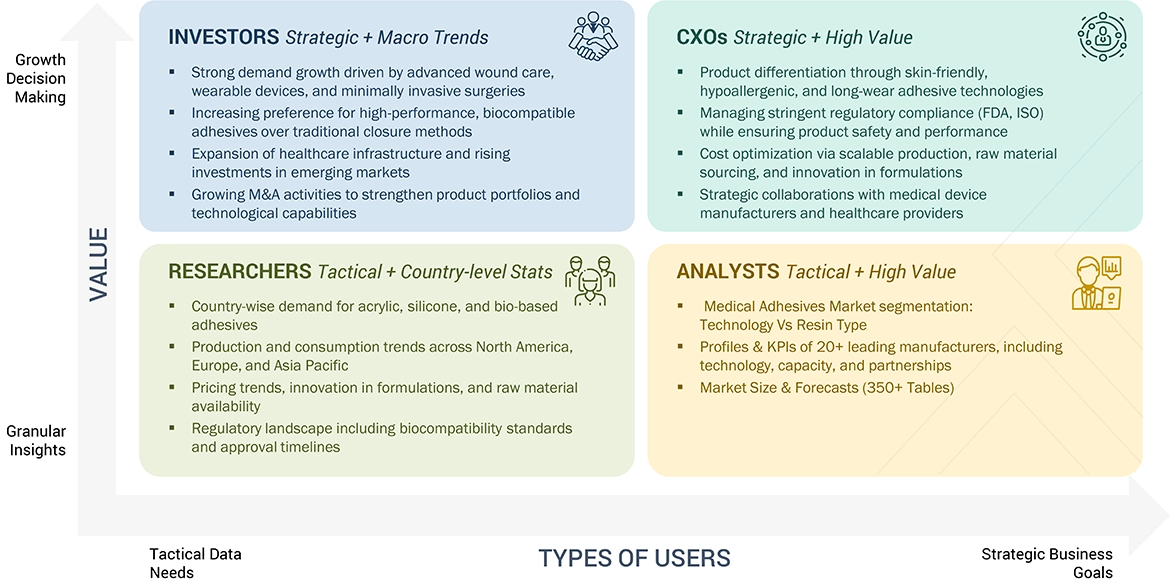
DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Demand for country-specific market insights | Provided detailed analysis of medical adhesives demand, pricing trends, regulatory landscape, and key end-use applications across regions such as North America, Europe, and Asia Pacific. | Helps clients identify high-growth healthcare markets, understand regulatory requirements, and plan strategic regional expansion. |
| Request for competitor benchmarking | Delivered comparative assessment of leading medical adhesive manufacturers, including product portfolios, technology focus (water-based, hot melt, etc.), R&D capabilities, and strategic initiatives. | Enables better competitive positioning, identification of innovation gaps, and benchmarking against industry leaders. |
| Application-specific insights | Provided insights into adhesive usage across surgery, dental, medical devices, wound care, and wearable applications, highlighting performance requirements and adoption trends. | Supports product development, targeted marketing strategies, and alignment with evolving healthcare needs. |
| Custom material/formulation support | Evaluated and recommended suitable adhesive types such as acrylic, silicone, and bio-based formulations for specific medical and skin-contact applications. | Reduces development time, enhances product performance, and ensures compatibility with clinical requirements. |
| Technical feasibility & product guidance | Delivered guidance on adhesion performance, biocompatibility, sterilization compatibility, and durability for various medical applications. | Improves product reliability, ensures patient safety, and enhances end-use performance. |
| Regulatory & compliance support | Provided analysis of global medical regulations (FDA, EMA) and standards related to biocompatibility, safety, and quality compliance. | Ensures adherence to regulatory standards, reduces approval risks, and supports successful product commercialization. |
RECENT DEVELOPMENTS
- December 2025 : Solventum completed the acquisition of Acera Surgical to expand its MedSurg portfolio into synthetic tissue matrices for advanced wound care.
- September 2025 : Henkel AG & Co. KGaA launched new Loctite LED-curable adhesives (AA 3951, AA 3953, AA 3961, AA 3963) for medical devices, offering fast curing, regulatory compliance, and improved efficiency, strengthening its position in the medical adhesives market.
- February 2024 : H.B. Fuller is expanding its Medical Adhesive Technologies (MAT) business with the acquisitions of Medifill Ltd., an Irish maker of wound closure adhesives, and GEM S.r.l., an Italian medical adhesive and device manufacturer.
- January 2022 : Chemence Medical received FDA clearance for the Exofin Precision Pen, a pen-style topical skin adhesive device for wound closure. It offers precise, controlled application with an adjustable tip supporting multiple delivery modes, improving versatility and ease of use.
- April 2021 : 3M introduced a new silicone adhesive solution, the 2484 single-coated medical film tape, designed to balance strength, flexibility, and comfort. It is breathable, conformable, supports up to seven-day wear, and minimizes skin cell damage, making it ideal for fragile skin applications.
Table of Contents

Methodology
The study involves two major activities in estimating the current market size for the medical adhesives market. Exhaustive secondary research was done to collect information on the market, peer market, and parent market. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. Afterward, market breakdown and data triangulation were used to estimate the market size for segments and subsegments.
Secondary Research
Secondary sources used for this research include the financial statements of companies that offer medical adhesives and information from various trade, business, and professional associations. Secondary research has been used to obtain critical information on the industry’s value chain, the total pool of key players, market classification, and segmentation by industry trends down to the bottom-most level and regional markets. Secondary data were collected and analyzed to determine the overall size of the medical adhesives market, and the results were validated by primary respondents.
Primary Research
Extensive primary research was conducted following secondary research on the medical adhesives market. Several primary interviews were conducted with market experts from both the demand and supply sides across major countries of North America, Europe, Asia Pacific, the Middle East & Africa, and South America. Primary data was collected through questionnaires, emails, and telephonic interviews. The primary sources from the supply side included various industry experts, such as Chief X Officers (CXOs), Vice Presidents (VPs), Directors from business development, marketing, product development/innovation teams, and related key executives from medical adhesives industry vendors; material providers; distributors; and key opinion leaders. Primary interviews were conducted to gather insights into market statistics, revenue from products and services, market breakdowns, market size estimates, market forecasting, and data triangulation. Primary research also helped in understanding the various trends related to technology, applications, end users, and regions. Stakeholders from the demand side, such as CIOs, CTOs, CSOs, and installation teams of the customer/end users who are seeking medical adhesives services, were interviewed to understand the buyer’s perspective on the suppliers, products, component providers, and their current usage of medical adhesives and future outlook of their business, which will affect the overall market.
The following is the breakdown of primary respondents:
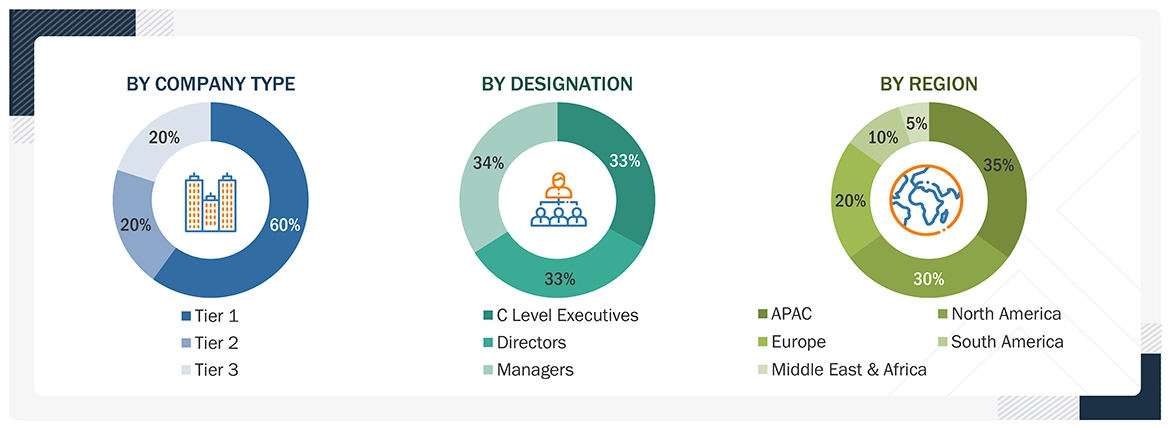
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The research methodology used to estimate the size of the medical adhesives market includes the following details. The market sizing of the market was undertaken from the demand side. The market was upsized based on regional demand for medical adhesives across different end-use industries. Such procurements provide information on the demand aspects of the medical adhesives industry for each end-use industry. For each end-use, all possible market segments were integrated and mapped.
Medical Adhesives Market : Top-Down and Bottom-Up Approach

Data Triangulation
After arriving at the overall size from the market size estimation process explained above, the total market was split into several segments and subsegments. The data triangulation and market breakdown procedures explained below were implemented, as applicable, to complete the overall market engineering process and obtain exact statistics for various market segments and subsegments. The data was triangulated by examining various factors and trends on both the demand and supply sides. Additionally, the market size was validated using both top-down and bottom-up approaches.
Market Definition
Medical adhesives are specialized bonding materials used by healthcare professionals to secure medical devices, bond tissues, and protect wounds. The medical adhesives are designed for safe skin and internal use because they maintain effective adhesion without causing any skin problems. The adhesives include cyanoacrylates and fibrin-based sealants, acrylics, and silicone-based systems, which medical professionals use to meet specific treatment requirements. Medical adhesives find extensive application in wound closure, surgical procedures, and the attachment of wearable devices and transdermal drug delivery systems. The products serve as necessary replacements for traditional sutures and staples because they deliver both flexible performance and durable construction, along with simple application.
Key Stakeholders
- Medical Adhesives Manufacturers
- Medical Adhesives Distributors and Suppliers
- End-use Industries
- Universities, Governments, and Research Organizations
- Associations and Industrial Bodies
- R&D Institutes
- Environmental Support Agencies
- Investment Banks and Private Equity Firms
- Research and Consulting Firms
Report Objectives
- To define, describe, and forecast the market size in terms of volume and value
- To provide detailed information regarding the key factors, such as drivers, restraints, opportunities, and challenges influencing market growth
- To analyze and forecast the market by technology, resin type, application, and region
- To forecast the market size concerning five main regions (along with country-level data), namely, North America, Europe, Asia Pacific, Middle East & Africa, and South America, and analyze the significant region-specific trends
- To strategically analyze micromarkets with respect to individual growth trends, prospects, and contributions of the submarkets to the overall market
- To analyze the market opportunities and the competitive landscape for stakeholders and market leaders
- To assess recent market developments and competitive strategies, such as agreements, contracts, acquisitions, partnerships & collaborations, and new product developments/new product launches, to draw the competitive landscape
- To strategically profile the key market players and comprehensively analyze their core competencie
Available customizations:
MarketsandMarkets offers the following customizations for this market report:
- Additional country-level analysis of the medical adhesives market
Product Analysis
- Product matrix, which provides a detailed comparison of the product portfolio of each company in the market
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Medical Adhesives Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS


















Growth opportunities and latent adjacency in Medical Adhesives Market