The study involved four major activities in estimating the current size of the Transdermal Patches Market. Exhaustive secondary research was done to collect information on the market, peer market, and parent market. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. After that, market breakdown and data triangulation were used to estimate the market size of segments and subsegments.
Secondary Research
Secondary sources referred to for this research study include financial statements of companies offering Transdermal Patches and information from various trade, business, and professional associations. The secondary data was collected and analyzed to arrive at the overall size of the Transdermal Patches market, which was validated by primary respondents.
Primary Research
Extensive primary research was conducted after obtaining information regarding the Transdermal Patches market scenario through secondary research. Several primary interviews were conducted with market experts from both the demand and supply sides across major countries of North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. Primary data was collected through questionnaires, emails, and telephonic interviews. The primary sources from the supply side included various industry experts, such as Chief X Officers (CXOs), Vice Presidents (VPs), Directors from business development, marketing, product development/innovation teams, and related key executives from Transdermal Patches manufacturers; distributors; and key opinion leaders.
Primary interviews were conducted to gather insights such as market statistics, data on revenue collected from the products and services, market breakdowns, market size estimations, market forecasting, and data triangulation. Primary research also helped in understanding the various trends related to technology, application, vertical, and region. Stakeholders from the demand side customers/end users who are using Transdermal Patches were interviewed to understand the buyer’s perspective on the suppliers, products, and their current usage of microfluidics and the future outlook of their business, which will affect the overall market.
A breakdown of the primary respondents for the Transdermal Patches market is provided below:
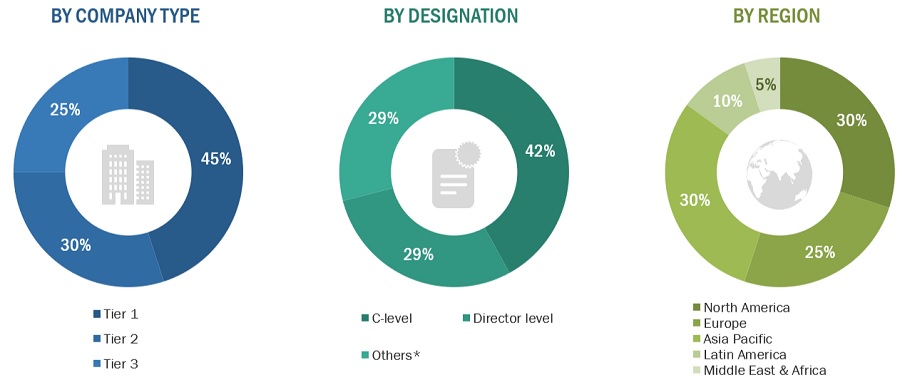
Note 1: C-level primaries include CEOs, COOs, CTOs, and VPs.
Note 2: Other primaries include Others include sales managers, marketing managers, and product managers.
Note 3: Companies are classified into tiers based on their total revenue. As of 2020: Tier 1=>USD 1 billion, Tier 2 = USD 500 million to USD 1 billion, Tier 3=<USD 500 million
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The research methodology used to estimate the size of the Transdermal Patches market includes the following details.
The market sizing of the market was undertaken from the global side.
Country-level Analysis: The size of the Transdermal Patches market was obtained from the annual presentations of leading players and secondary data available in the public domain. The share of products and services in the overall Transdermal Patches market was obtained from secondary data and validated by primary participants to arrive at the total Transdermal Patches market. Primary participants further validated the numbers.
Geographic market assessment (by region & country): The geographic assessment was done using the following approaches:
Approach 1: Geographic revenue contributions/splits of leading players in the market (wherever available) and respective growth trends
Approach 2: Geographic adoption trends for individual product segments by end users and growth prospects for each of the segments (assumptions and indicative estimates validated from primary interviews)
At each point, the assumptions and approaches were validated through industry experts contacted during primary research. Considering the limitations of data available from secondary research, revenue estimates for individual companies (for the overall Transdermal Patches market and geographic market assessment) were ascertained based on a detailed analysis of their respective product offerings, geographic reach/strength (direct or through distributors or suppliers), and the shares of the leading players in a particular region or country.
Global Transdermal Patches Market Size: Top-Down Approach

To know about the assumptions considered for the study, Request for Free Sample Report
Global Transdermal Patches Market Size: Bottom-Up Approach

Data Triangulation
After arriving at the overall size from the market size estimation process explained above, the total market was split into several segments and subsegments. The data triangulation and market breakdown procedures explained below were implemented, wherever applicable, to complete the overall market engineering process and arrive at the exact statistics for various market segments and subsegments. The data was triangulated by studying various factors and trends from the demand and supply sides. Along with this, the market size was validated using both the top-down and bottom-up approaches.
Market Definition
Transdermal patches are medicated adhesive patches that are placed on the skin to deliver a specific dose of medication into the bloodstream. These patches are promising alternatives to oral formulations as they offer improved bioavailability, continuous drug delivery, and reduced side effects, among other advantages. Transdermal patches are especially useful for patients who require constant drug levels.
Key Stakeholders
-
Senior Management
-
Finance Department
-
Procurement Department
Objectives of the Study
-
To describe, analyze, and forecast the transdermal patches market, by patch type, adhesive type, application, distribution channel, end user and region.
-
To describe and forecast the transdermal patches market for key regions, namely, North America, Europe, the Asia Pacific, Latin America, and the Middle East and Africa
-
To provide detailed information regarding the drivers, restraints, opportunities, and challenges influencing the growth of the transdermal patches market
-
To strategically analyze the ecosystem, regulations, patenting trend, value chain, Porter’s five forces, and prices pertaining to the market under study
-
To strategically analyze micro-markets with respect to individual growth trends, prospects, and contributions to the overall market
-
To analyze market opportunities for stakeholders and provide details of the competitive landscape for market players.
-
To profile key players and comprehensively analyze their market shares and core competencies in the transdermal patches market.
-
To analyze competitive developments such as collaborations, acquisitions, product launches, expansions, and R&D activities in the transdermal patches market.
Available Customizations
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for the report:
Geographic Analysis
-
Further breakdown of the Rest of Europe Transdermal Patches market into the Netherlands, Switzerland, Austria, Belgium, and others
-
Further breakdown of the Rest of Asia Pacific Transdermal Patches market into Singapore, Malaysia, and others
Company Information
-
Detailed analysis and profiling of additional market players



Growth opportunities and latent adjacency in Transdermal Patches Market