
Pharmaceutical Blister Packaging Market
Pharmaceutical Blister Packaging Market by Material (Plastic Films, Aluminum Foils, Paper & Paperboard), Blister Type, Sealing Technology, Structure Type, Drug Type, Functionality, Application, End User, and Region - Global Forecast to 2030




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
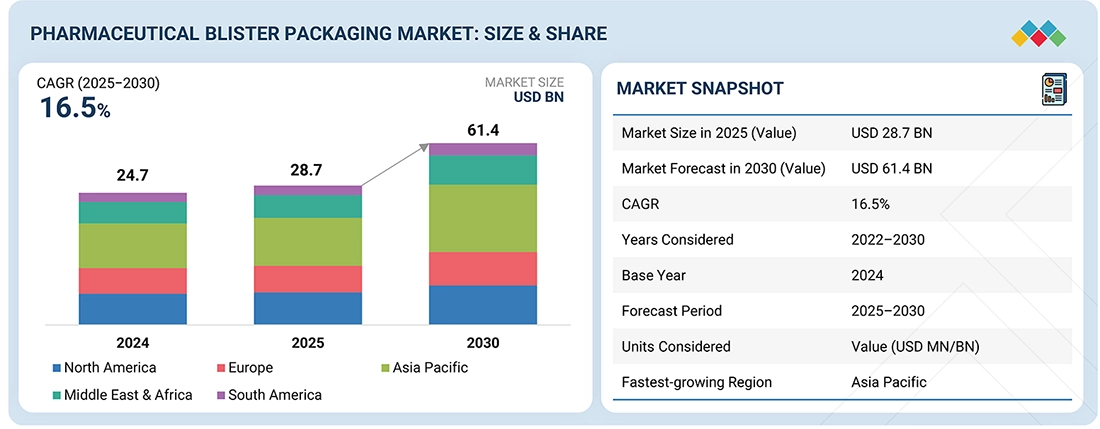
The pharmaceutical blister packaging market is projected to reach USD 61.4 billion by 2030 from USD 28.7 billion in 2025, at a CAGR of 16.5% from 2025 to 2030. Pharmaceutical blister packaging refers to pre-formed plastic cavities sealed with a backing material, typically aluminum foil or paper, designed to contain individual doses of medication. This packaging format is widely used for oral solid drugs such as tablets and capsules, providing protection against environmental factors, including moisture, light, and contamination. Blister packs offer multiple advantages including tamper evidence, child resistance, unit dose accuracy, and enhanced patient compliance through clear visibility of medication. The packaging serves as a critical component in pharmaceutical supply chains, ensuring product integrity from manufacturing through distribution to end-user consumption.
KEY TAKEAWAYS
-
BY REGIONThe Asia Pacific is expected to witness the highest growth rate, at a CAGR of 18.5% from 2025 to 2030, reaching USD 20.3 billion. North America remains the largest market, driven by strong healthcare infrastructure and innovations in drug delivery.
-
BY MATERIALThe plastic films segment held a 38.0% share of the pharmaceutical blister packaging market in 2024. Pharmaceutical blister packaging uses plastic films as its primary packaging material because these films provide multiple benefits at an economical price while maintaining operational efficiency.
-
BY APPLICATIONThe pharmaceutical blister packaging market reaches its highest value through prescription drugs because these medications need to be packaged in unit-dose and tamper-evident containers.
-
BY DRUG TYPETablets represent the largest segment in pharmaceutical blister packaging, owing to their widespread use, stability, and ease of handling. The segment's leading position exists because people worldwide consume solid oral medications and because tablet production and packaging processes need less financial resources.
-
COMPETITIVE LANDSCAPE - STAR PLAYERSAmcor and Constantia Flexibles were identified as top companies in the pharmaceutical blister packaging market, leveraging sustainable and innovative packaging solutions to maintain competitive advantages through substantial investments in research and development.
-
COMPETITIVE LANDSCAPE - STARTUPSTekni Plex and Klöckner Pentaplast have distinguished themselves by advancing packaging technologies through strategic collaborations and product innovations, including digital traceability solutions and PVC-free thermoform blister systems that address sustainability trends.
The pharmaceutical blister packaging market is expanding rapidly, driven by the rising occurrence of chronic diseases, an aging population, and the growing need for convenient and protective packaging solutions. The increasing adoption of personalized medicine, combined with technological advancements in pharmaceutical packaging that enhance user safety and compliance, is further accelerating demand for high-performance blister packaging across the pharmaceutical industry. Sustainability trends are influencing manufacturers to adopt PVC-free blisters and recyclable packaging solutions, aligning with environmental regulations and consumer preferences for eco-friendly pharmaceutical products.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The pharmaceutical blister packaging market is currently experiencing a phase of innovation and rapid adoption. Healthcare systems and pharmaceutical manufacturers across North America, Europe, and the Asia Pacific are witnessing advancements in blister packaging technology and material science. This progress aligns with emerging trends in personalized medicine, patient-centric drug delivery, and sustainable packaging solutions. In the pharmaceutical industry, the use of PVC-free materials, recyclable substrates, and advanced barrier films is increasing among drug manufacturers and contract packaging organizations. These materials facilitate the development of packaging solutions that provide superior protection, reduce environmental impact, and align with regulatory requirements for child resistance and tamper evidence. Pharmaceutical companies and regulatory bodies are encouraging the adoption of smart packaging technologies, including digital traceability systems and serialization capabilities, which help combat counterfeit products and improve supply chain transparency. These initiatives support the development of automation-friendly, clinically validated blister packaging platforms tailored to the needs of diverse therapeutic categories and patient populations.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Growing demand for convenient, protective, and cost-effective packaging solutions

-
Rising occurrence of chronic diseases and aging population driving pharmaceutical consumption
Level
-
High costs associated with dynamic regulatory policies affecting overall packaging expenses
Level
-
Growing need for primary pharmaceutical packaging and introduction of personalized medicine
Level
-
Need to safeguard against counterfeit products while balancing innovation with cost efficiency
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Growing demand for convenient, protective, and cost-effective packaging solutions
The growing demand for convenient, protective, and cost-effective packaging solutions is a primary driver of the pharmaceutical blister packaging market. Blister packaging offers a robust solution with excellent barrier properties against moisture, UV rays, and contamination, ensuring medication stability and efficacy throughout the product lifecycle. The format provides unit dose accuracy, which enhances patient compliance and reduces medication errors, particularly important for chronic disease management and elderly patient populations. The cost-effectiveness of blister packaging, combined with its ability to extend shelf life and reduce waste, makes it an attractive option for pharmaceutical manufacturers seeking to optimize production efficiency while maintaining high quality standards.
Restraint: High costs associated with dynamic regulatory policies affecting overall packaging expenses
High costs associated with dynamic regulatory policies represent a significant restraint on the pharmaceutical blister packaging market. Pharmaceutical packaging must comply with stringent regulations that vary across different markets and jurisdictions, requiring manufacturers to invest in compliance infrastructure, testing protocols, and documentation systems. The evolving nature of regulatory requirements, including serialization mandates, sustainability standards, and safety specifications, creates ongoing cost pressures for packaging suppliers and pharmaceutical companies. These regulatory burdens can affect overall packaging costs and create barriers to market entry, particularly for smaller manufacturers and companies operating across multiple geographic regions with differing regulatory frameworks.
Opportunity: Growing need for primary pharmaceutical packaging and introduction of personalized medicine
The growing need for primary pharmaceutical packaging and the introduction of personalized medicine present significant growth opportunities in the pharmaceutical blister packaging market. As personalized medicine gains traction, there is increasing demand for flexible packaging solutions that can accommodate smaller batch sizes, customized dosing regimens, and patient-specific labeling requirements. Blister packaging is well-suited to these applications due to its versatility, scalability, and ability to provide individual dose protection. The expansion of specialty pharmaceuticals, biologics, and targeted therapies is creating new opportunities for innovative blister packaging designs that address unique stability requirements, administration protocols, and patient convenience factors.
Challenge: Need to safeguard against counterfeit products while balancing innovation with cost efficiency
Safeguarding against counterfeit products while balancing innovation with cost efficiency remains a key challenge in the pharmaceutical blister packaging market. The pharmaceutical industry faces ongoing threats from counterfeit medications, which pose serious risks to patient safety and brand integrity. Blister packaging manufacturers must incorporate anti-counterfeiting features such as holograms, serialization codes, and tamper-evident seals while maintaining cost competitiveness. The challenge lies in implementing advanced security technologies and traceability systems without significantly increasing packaging costs or complicating manufacturing processes. Companies must invest in research and development to create innovative solutions that enhance product authentication and supply chain visibility while remaining economically viable for widespread adoption across diverse pharmaceutical applications and market segments.
PHARMACEUTICAL BLISTER PACKAGING MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Amcor Plc has secured a commercial order for its AmSky PVC-free thermoform blister system, which provides sustainable packaging solutions for pharmaceutical manufacturers seeking to eliminate PVC materials while maintaining superior barrier properties and product protection for oral solid drugs. | Delivers environmental sustainability through PVC-free materials, maintains excellent moisture and oxygen barrier performance, supports regulatory compliance, and enables pharmaceutical companies to meet sustainability goals while ensuring medication stability and patient safety. |
 |
Constantia Flexibles developed its Flexible Blister solution, a next-generation pharmaceutical blister format that combines traditional thermoformed blister functionality with strip packaging flexibility. The solution allows pharmaceutical manufacturers to implement anti-counterfeiting protections and printed patient information and customizable opening mechanisms, which include child-resistant formats directly into their tablet and capsule blister packaging while preserving their current manufacturing processes. | The system achieves several objectives by implementing its integrated packaging system, which enhances product security and anti-counterfeiting security and improves patient accessibility and medication adherence through its portable information-rich design. It also achieves safety compliance through its customizable safety features, which include child-resistant and senior-friendly options and the system decreases capital expenses through its ability to connect with current packaging systems while maintaining complete supply chain protection of drug stability. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The pharmaceutical blister packaging market is a collaborative ecosystem where material suppliers, packaging manufacturers, pharmaceutical companies, contract packaging organizations, and healthcare providers work together to develop and implement packaging solutions that ensure medication safety and efficacy. Companies in the market supply a variety of materials and technologies, including thermoformed plastics, aluminum foils, barrier films, and printing systems. Contract packaging organizations and pharmaceutical manufacturers utilize these materials to create blister packs that meet stringent quality standards and regulatory requirements. The ecosystem includes equipment manufacturers that provide blister packaging machinery, inspection systems, and serialization technologies. Regulatory bodies and industry associations establish standards and guidelines that govern packaging performance, safety features, and environmental impact. Ultimately, healthcare institutions, pharmacies, and patients benefit from blister packaging solutions that provide medication protection, dosing accuracy, and ease of use, supported by comprehensive quality assurance and supply chain management systems.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
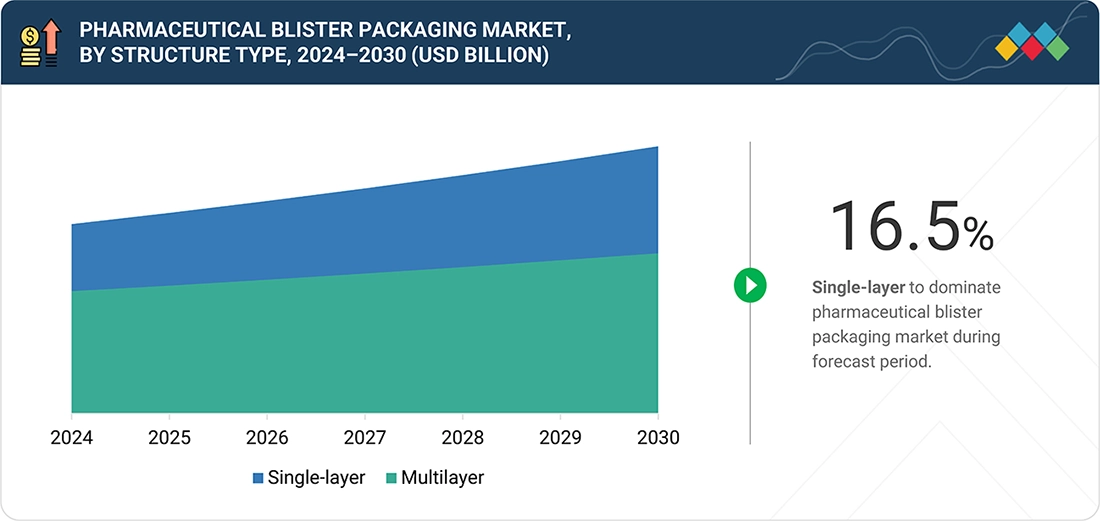
Pharmaceutical Blister Packaging Market, by Material
The pharmaceutical blister packaging market experiences its highest growth rate through plastic films, which provide the best combination of affordable production costs and easy processing and effective product performance. The market for packaging materials is dominated by PVC and PET and advanced barrier-coated films because these materials provide excellent thermoformability and transparent properties and they safeguard moisture-sensitive medications when they are used with PVDC or Aclar coatings. The segment experiences rapid expansion because of rising demand for solid oral dosage forms, the growing capacity of thermoforming operations, and the continuous development of PVC-free recyclable mono-material films, which help pharmaceutical packaging companies meet their sustainability targets and environmental regulations.
Pharmaceutical Blister Packaging Market, by Application
Biopharmaceutical products are projected to be the fastest-growing segment in pharmaceutical blister packaging. This growth is driven by the rapid expansion of biologics, specialty drugs, and personalized medicines, which require high-barrier, contamination-resistant, and unit-dose packaging formats. Blister packaging is increasingly being adapted for select oral biologics and adjunct therapies, supporting stability and patient compliance. Additionally, rising investments in biopharma R&D, increasing approvals of complex therapies, and the need for secure, traceable, and premium packaging solutions are accelerating demand in this segment, outpacing traditional categories like OTC and generics.
REGION
Asia Pacific to be fastest-growing region in market during forecast period
The Asia Pacific is the fastest-growing region in the pharmaceutical blister packaging market, at a CAGR of 18.5% from 2025 to 2030, reaching USD 20.3 billion. This rapid expansion is driven by increasing pharmaceutical manufacturing capacity, growing healthcare expenditure, and rising demand for quality packaging solutions across emerging markets. The region benefits from expanding middle-class populations with greater access to healthcare, increasing prevalence of chronic diseases, and government initiatives to improve pharmaceutical quality standards. Countries like China, India, Japan, and South Korea are experiencing significant growth in pharmaceutical production and consumption, creating substantial demand for blister packaging solutions. Additionally, the presence of cost-competitive manufacturing facilities, growing contract packaging services, and increasing adoption of international quality standards are facilitating the swift expansion of blister packaging utilization across diverse therapeutic categories and distribution channels throughout the Asia Pacific region.

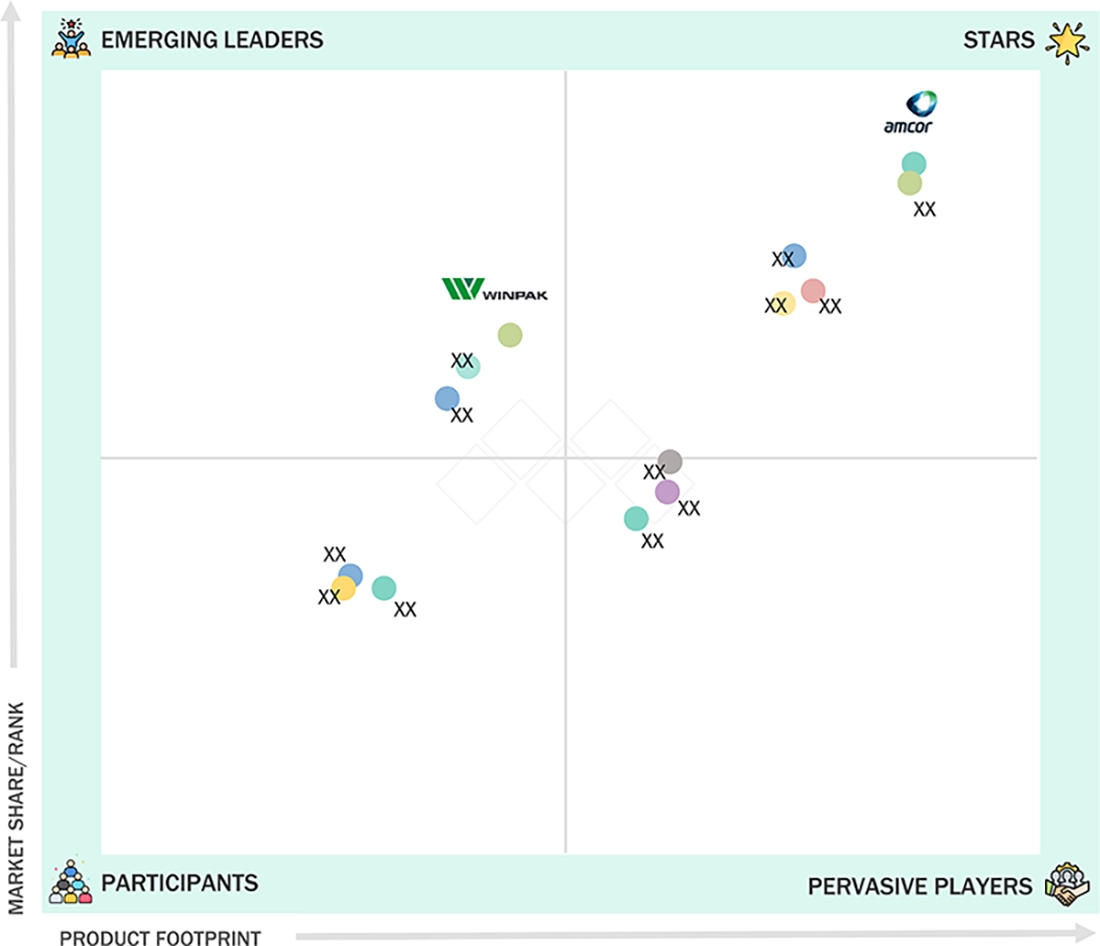
PHARMACEUTICAL BLISTER PACKAGING MARKET: COMPANY EVALUATION MATRIX
Amcor Plc, recognized as a leading player in the pharmaceutical blister packaging market, offers a diverse portfolio that includes PVC-free thermoform blister systems, sustainable packaging materials, and advanced barrier solutions for pharmaceutical applications. The company boasts a strong global manufacturing presence and collaborates with pharmaceutical companies and contract packaging organizations across North America, Europe, and the Asia Pacific. Another significant player in the market is Constantia Flexibles, identified as a top company strengthening its position by introducing digital traceability solutions, serialization-enabled packaging systems, and specialty glass and plastic containers designed for use in pharmaceutical, biotechnology, and healthcare applications.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Amcor Plc (Switzerland)
- Constantia Flexibles (Austria)
- Tekni-Plex (US)
- Sonoco Products Company (US)
- UFlex Ltd. (India)
- Winpak Ltd. (Canada)
- ACG (India)
- WestRock (US)
- Honeywell International (US)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 28.7 Billion |
| Market Forecast in 2030 (Value) | USD 61.4 Billion |
| Growth Rate | CAGR of 16.5% from 2025–2030 |
| Years Considered | 2022–2030 |
| Base Year | 2024 |
| Forecast Period | 2025–2030 |
| Units Considered | Value (USD Million/Billion) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Regions Covered | North America, Europe, Asia Pacific, South America, and Middle East & Africa |
WHAT IS IN IT FOR YOU: PHARMACEUTICAL BLISTER PACKAGING MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Understanding demand patterns for pharmaceutical blister packaging across global healthcare systems | Assessed blister packaging adoption trends across key pharmaceutical markets, including North America, Europe, and the Asia Pacific, highlighting differences in material preferences, regulatory requirements, and application focus across oral solid drugs, chronic disease management, and self-administered drug delivery systems. | Helps clients align product portfolios with region-specific regulatory frameworks, sustainability requirements, and therapeutic category demand drivers. |
| Identifying material suppliers and contract packaging opportunities | Mapped regional blister packaging suppliers, contract packaging organizations, and pharmaceutical manufacturers across North America, Europe, and Asia Pacific, with insights into thermoforming capabilities, barrier film technologies, serialization systems, and sustainable material alternatives, including PVC-free solutions. | Supports supply chain optimization, reduces material sourcing risks, enables sustainability transitions, and improves speed-to-market for new pharmaceutical products. |
RECENT DEVELOPMENTS
- 2024 : Amcor Plc secured a commercial order for its AmSky PVC-free thermoform blister system, demonstrating the company's commitment to sustainable packaging solutions that eliminate PVC materials while maintaining superior barrier properties and product protection for pharmaceutical applications. This innovation addresses growing demand from pharmaceutical manufacturers seeking environmentally responsible packaging alternatives that comply with evolving sustainability regulations and corporate environmental goals.
- 2023 : Constantia Flexibles launched REGULA CIRC, a recyclable cold-form aluminum blister foil designed for pharmaceutical applications. This solution delivers full barrier protection against moisture, oxygen, and light while addressing sustainability requirements by enabling recyclability, marking a significant step toward circular blister packaging formats in the pharma industry.
Table of Contents

Methodology
The study involved four major activities to estimate the current size of the Pharmaceutical Blister Packaging Market. Exhaustive secondary research was carried out to collect information on the market, the peer product market, and the parent product group market. The next step was to validate these findings, assumptions, and sizes with the industry experts across the value chain of pharmaceutical packaging through primary research. The top-down and bottom-up approaches were employed to estimate the overall size of the Pharmaceutical Blister Packaging Market. After that, market breakdown and data triangulation procedures were used to determine the size of different segments and sub-segments of the market.
Secondary Research
The market for the companies offering pharmaceutical packaging is arrived at by secondary data available through paid and unpaid sources, analyzing the product portfolios of the major companies in the ecosystem, and rating the companies by their performance and quality. Various secondary sources, such as Business Standard, Bloomberg, World Bank, and Factiva, were referred to in order to identify and collect information for this study on the Pharmaceutical Blister Packaging Market. In the secondary research process, various secondary sources were referred to identify and collect information related to the study. Secondary sources included annual reports, press releases, and investor presentations, forums, certified publications, and whitepapers. The secondary research was used to obtain critical information on the industry’s value chain, the total pool of key players, market classification, and segmentation from the market and technology-oriented perspectives.
Primary Research
In the primary research process, various primary sources from both the supply and demand sides were interviewed to obtain qualitative and quantitative information for this report. The primary sources from the supply side included industry experts, such as Chief Executive Officers (CEOs), Vice Presidents (VPs), marketing directors, technology and innovation directors, and related key executives from several key companies and organizations operating in the Pharmaceutical Blister Packaging Market. After the complete market engineering (calculations for market statistics, market breakdown, market size estimations, market forecasting, and data triangulation), extensive primary research was conducted to gather information and verify and validate the critical numbers arrived at. Primary research was also conducted to identify the segmentation types, industry trends, competitive landscape of pharmaceutical packaging offered by various market players, and key market dynamics, such as drivers, restraints, opportunities, challenges, industry trends, and key player strategies. In the complete market engineering process, the top-down and bottom-up approaches and several data triangulation methods were extensively used to perform the market estimation and market forecasting for the overall market segments and subsegments listed in this report. Extensive qualitative and quantitative analysis was performed on the complete market engineering process to list the key information/insights throughout the report.
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The top-down and bottom-up approaches were used to estimate and validate the size of the Pharmaceutical Blister Packaging Market. These approaches were also used extensively to estimate the size of various dependent market segments. The research methodology used to estimate the market size included the following:

Data Triangulation
After arriving at the overall market size using the market size estimation processes, the market was split into several segments and subsegments. The data triangulation procedure was employed, wherever applicable, to complete the overall market engineering process and arrive at the exact statistics of each market segment and subsegment. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Pharmaceutical packaging pertains to the materials and systems designed to protect, preserve, and deliver pharmaceutical products for oral, injectable, inhalation, and topical drug delivery modalities. It plays an important role in the stability and sterility of a drug, avoids contamination, and provides accurate dosing for the life of the product. Pharmaceutical packaging denotes the numerous varieties of available options, like plastic bottles, blister packs, vials, ampoules, prefillable syringes, tubes, pouches, and cartons to meet drug formulation requirements and regulatory obligations. Some of the significant functionalities of pharmaceutical packaging include moisture, oxygen, and light barrier protection, tamper evidence, patient convenience, and functionality with active pharmaceutical ingredients (APIs).
Areas of interest include ISO 15378 relating specifically to primary packaging materials, ISO 9001 regarding quality management systems, and GMP compliance. The DSCSA and the EU FMD identified serialized, tracking, and anti-counterfeiting programs, creating an opportunity for the packaging industry to help with drug quality and patient safety.
Stakeholders
- Pharmaceutical packaging manufacturers
- Raw material suppliers
- Converters & processors
- Distributors and traders
- Industry associations and regulatory bodies
- End users
Report Objectives
- To define, describe, and forecast the size of the Pharmaceutical Blister Packaging Market, based on type, packaging type, drug delivery, raw material, and region in terms of value and volume
- To provide detailed information on the significant drivers, restraints, opportunities, and challenges influencing the market
- To strategically analyze micromarkets concerning individual growth trends, prospects, and their contribution to the market
- To assess the growth opportunities in the market for stakeholders and provide details on the competitive landscape for market leaders
- To forecast the market size of segments and subsegments for North America, Europe, Asia Pacific, South America, and the Middle East & Africa
- To strategically profile key players and comprehensively analyze their market shares and core competencies
- To analyze competitive developments such as acquisitions, product launches, expansions, partnerships, and agreements in the Pharmaceutical Blister Packaging Market
- To provide the impact of AI/Gen AI on the market
Available Customizations
With the given market data, MarketsandMarkets offers customizations according to client-specific needs.
The following customization options are available for the Pharmaceutical Blister Packaging Market report:
Product Analysis
- A product matrix that gives a detailed comparison of the product portfolio of each company
Geographic Analysis as per Feasibility
- A further breakdown of the Pharmaceutical Blister Packaging Market for additional countries
Company Information
- Detailed analysis and profiling of additional market players (up to five)
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Pharmaceutical Blister Packaging Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS
















Growth opportunities and latent adjacency in Pharmaceutical Blister Packaging Market