
US Respiratory Care Devices Market Size, Growth, Share & Trends Analysis
US Respiratory Care Device Market by Product (PAP Devices, Ventilators, Humidifiers, Oxygen Concentrators, Spirometers, Peak Flow Meter, Masks), Disease Indication (Asthma, Infectious Disease), End User (Hospitals, Home Care Settings) - Forecast to 2031




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
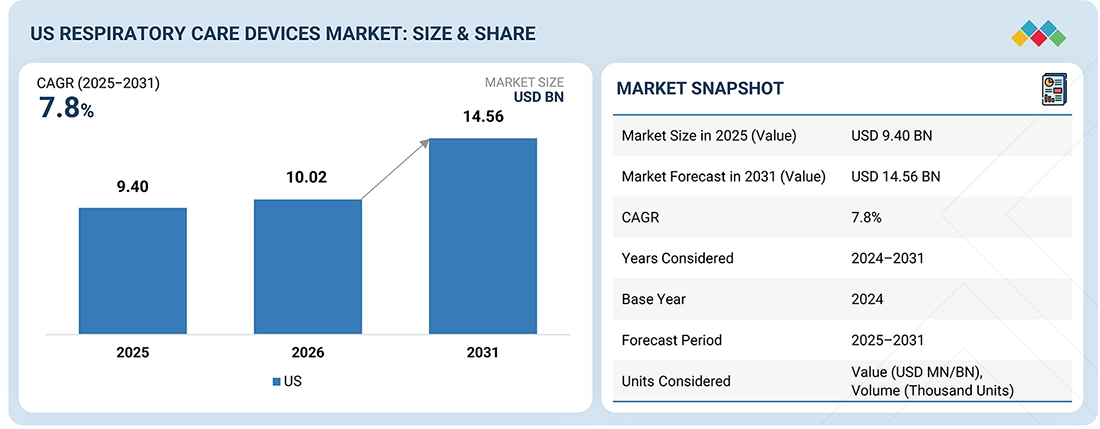
The US Respiratory Care Devices market, valued at US$9.40 billion in 2025, stood at US$10.02 billion in 2026 and is projected to advance at a resilient CAGR of 7.8% from 2026 to 2031, culminating in a forecasted valuation of US$14.56 billion by the end of the period. This growth is primarily driven by an increasing prevalence of chronic respiratory diseases such as COPD, asthma, and sleep apnea, along with an aging population that is more susceptible to breathing disorders. Factors like rising air pollution, a history of smoking, and respiratory complications following COVID-19 have also contributed significantly to the rising demand for long-term respiratory support. Advancements in technology, such as portable oxygen concentrators, smart inhalers, and connected ventilators, are expected to make these devices more user-friendly and support the trend toward home-based care. Additionally, favorable reimbursement policies and strong regulatory support from agencies like the Centers for Medicare & Medicaid Services and the US Food and Drug Administration, along with high healthcare spending and widespread adoption of advanced medical technologies, are further propelling market growth in hospitals, clinics, and home care settings.
KEY TAKEAWAYS
-
BY PRODUCTBased on product, the therapeutic respiratory care devices segment accounted for the largest share of 55.7% of the US respiratory care devices market in 2025.
-
BY DISEASE INDICATIONBased on disease indication, the COPD segment accounted for the largest share of the US respiratory care devices market in 2025.
-
BY END USERBased on end user, the hospitals segment accounted for the largest share of 68.1% of the US respiratory care devices market during the forecast period.
-
COMPETITIVE LANDSCAPEKoninklijke Philips N.V. and Medtronic were recognized as star players due to their strong product portfolios.
-
COMPETITIVE LANDSCAPETELESAIR Inc. has established itself as a startup due to its strong product portfolio and business strategy.
The growth of the respiratory care devices market in US is primarily driven by the rising incidence of chronic respiratory diseases, such as COPD, asthma, and sleep apnea. Additionally, the rapidly aging population is more vulnerable to breathing disorders. Factors like increasing air pollution, patients' previous smoking habits, and respiratory complications following COVID-19 have contributed to a larger patient pool in need of long-term respiratory care. Technological innovations, including portable oxygen concentrators, smart inhalers, and connected ventilators, have improved the usability of respiratory devices and supported the transition to home-based care. Furthermore, attractive reimbursement plans and strong regulatory support from agencies like the Centers for Medicare & Medicaid Services and the US Food and Drug Administration, coupled with high healthcare spending and the swift adoption of advanced medical technologies, continuously drive growth in the inpatient, outpatient, and home care markets.
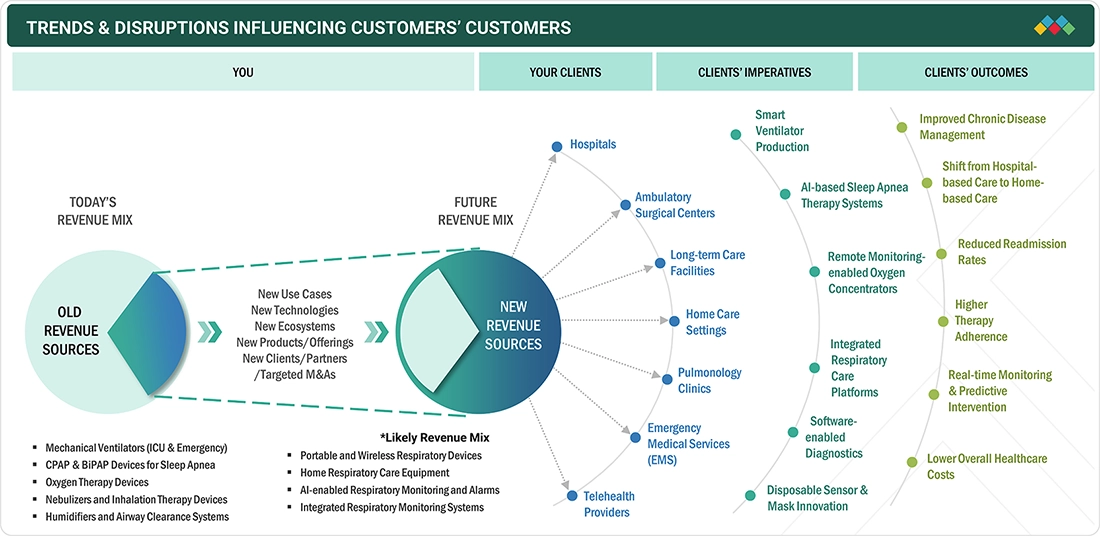
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
A large portion of the US respiratory care devices market is undergoing a transformation in response to changing customer needs. The nature of respiratory care is shifting from traditional hospital-centered models to more intelligent, portable, and digitalized products. Medical professionals are increasingly focused on developing methods to provide precise monitoring that can detect respiratory issues at an early stage, allowing for more effective treatment of chronic respiratory diseases across various care settings. Furthermore, the emphasis on patient safety during anesthesia and sedation, along with the need to streamline clinical workflows, is driving the demand for advanced respiratory technology. As a result, trends such as remote monitoring, AI-powered systems, and home-based respiratory care are becoming more standard. These developments are leading to improved patient outcomes, reduced ICU and hospital stays, and greater operational efficiency for hospitals, ambulatory surgery centers, specialty clinics, and home healthcare providers.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Rising prevalence of chronic respiratory diseases

-
Increasing demand for home-based and non-invasive respiratory care
Level
-
High cost of advanced respiratory care devices
-
Strict regulatory and approval processes
Level
-
Integration of AI and digital health in respiratory devices
-
Development of portable and user-friendly devices
Level
-
Intense competition and pricing pressure
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Rising prevalence of chronic respiratory diseases
The increasing prevalence of chronic respiratory diseases is a key factor driving the rapid growth of the US respiratory care devices market. Conditions such as chronic obstructive pulmonary disease (COPD), asthma, and sleep apnea are on the rise due to an aging population, long-term exposure to air pollution, smoking history, and lifestyle-related risks like obesity. Additionally, post-COVID respiratory complications have significantly heightened the number of individuals needing continuous or long-term breathing support. Such cases typically require regular diagnosis, monitoring, and treatment, which can be effectively managed with devices like ventilators, oxygen therapy systems, CPAP/BiPAP machines, nebulizers, and respiratory monitoring equipment. As the number of chronic respiratory patients continues to rise, there is an increasing demand for both acute and home-based respiratory care devices across various healthcare settings, including hospitals, clinics, and home care.
Restraint: High cost of advanced respiratory care devices
The high cost of advanced respiratory care devices is a major barrier to the growth of the US respiratory care devices market. For instance, smart ventilators, portable oxygen concentrators, and AI-enabled respiratory monitoring systems incur significant expenses related to manufacturing, research and development, and regulatory compliance, which result in premium pricing. The initial purchase of these devices, along with ongoing expenses such as maintenance, software updates, and consumables, can be a substantial financial burden, especially for smaller hospitals, ambulatory surgery centers, and home care agencies. Additionally, some advanced devices are not fully covered by reimbursement policies, leading to affordability issues for patients requiring long-term respiratory support. These pricing challenges may hinder adoption rates and limit the integration of new respiratory technologies, particularly in cost-sensitive care settings.
Opportunity: Integration of AI and digital health in respiratory devices
The combination of artificial intelligence (AI) and digital health technologies is driving significant growth in the US respiratory care devices market. AI-powered respiratory machines have the ability to process large amounts of patient data in real time, allowing them to detect early signs of respiratory failure, effectively predict exacerbations of chronic conditions like COPD and asthma, and provide clinical support for timely interventions. Digital health platforms, such as connected inhalers, smart ventilators, and remote monitoring systems, enable continuous monitoring of patients' medication adherence, oxygen levels, and breathing patterns, even outside of clinical settings. This approach supports healthcare models that allow patients to receive care at home and in virtual environments, helping to prevent hospital admissions and reduce overall treatment costs. Furthermore, the trend of healthcare professionals making data-driven decisions and utilizing telehealth services is increasing. The integration of AI and digital health technologies with respiratory care devices is seen as a key to the future, with the US market poised to benefit significantly in terms of clinical outcomes, operational efficiencies, and accelerated growth.
Challenge: Intense competition and pricing pressure
The US respiratory care devices market faces significant challenges due to intense rivalry and price-cutting measures. The presence of numerous established global manufacturers, alongside a growing number of local and emerging competitors, has heightened competition across various product segments, including ventilators, oxygen therapy devices, and sleep apnea equipment. This competitive landscape forces companies to continually innovate while maintaining prices that satisfy customers, which ultimately leads to reduced profit margins. Additionally, group purchasing organizations (GPOs), hospital networks, and home care providers are increasingly demanding cost-effective solutions and volume discounts. This trend limits the ability to differentiate pricing strategies. As a result, manufacturers find themselves in a difficult position; they must invest heavily in research and development while also offering products at relatively low prices. Balancing these demands makes it challenging to achieve consistent profits over time while remaining technologically competitive.
US RESPIRATORY CARE DEVICES MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Sleep apnea masks are patient interfaces that can be used with a CPAP or BiPAP device to deliver continuous positive airway pressure and keep the airway open during sleep in patients with obstructive sleep apnea. These masks are intended for long-term nightly use, both at home and in clinical sleep therapy settings. | Improve airway patency and reduce apnea events, enhance patient comfort and therapy compliance, and support effective long-term management of sleep-disordered breathing. |
 |
Pulse oximetry devices are non-invasive sensors that continuously measure and monitor the patient's blood oxygen saturation (SpO2) and pulse rate, enabling patient monitoring in hospital wards, ICUs, emergency departments, and home care environments. | Allow early detection of hypoxemia, facilitate timely clinical intervention, increase patient safety, and help clinicians to monitor respiratory and cardiovascular status in real time. |
 |
Mechanical ventilators are used in assisted or controlled breathing modes to support patients who have respiratory failure or are unable to breathe adequately due to lung damage or other reasons in intensive care units and emergency care settings. | Provide sufficient oxygenation and ventilation, reduce breathing work, support recovery in critical cases, and enable accurate control of respiratory parameters for optimal patient care. |
 |
Humidifiers are respiratory therapy devices used to add heat and moisture to inhaled gases, preventing airway drying and increasing patient comfort during oxygen therapy and ventilation. | Increase patient comfort, lessen airway irritation, facilitate secretion clearance, and help the respiratory therapy be more effective and of longer duration. |
 |
Emergency and transport ventilators are devices that provide portable respiratory support to patients during pre-hospital care, in-hospital care, inter-facility transfer, and emergency response situations. | Provide stable ventilation during transport, allow quick response in emergencies, increase patient survival and safety, and maintain continuity of care outside traditional hospital settings. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
Inside the US, the respiratory care device market is a complicated and very close network of different players such as manufacturers, distributors, end users, and regulators, who together help to continuously invent, provide and safely adopt respiratory technologies. Leading manufacturers like Philips, Medtronic, and ResMed are the main innovators of ventilators, oxygen therapy units, and sleep apnea devices, whereas nationally acting major distributors like Cardinal Health, McKesson, Medline, and US Med take care of the supply and logistics all over the country. The use of these products is mainly to hospitals and healthcare systems like Mayo Clinic and Johns Hopkins Medicine for the treatment of acute and chronic respiratory diseases. The regulatory and standardization agencies in the US include, Food and Drug Administration, Centers for Medicare & Medicaid Services, and Federal Communications Commission, which among other things, assure the safety of medical devices, facilitate the reimbursement process, and regulate the compliance of connected technologies. All these actors together make up a strong and collaborative ecosystem that allow the continuous advancement and update of the respiratory care field in the United States.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
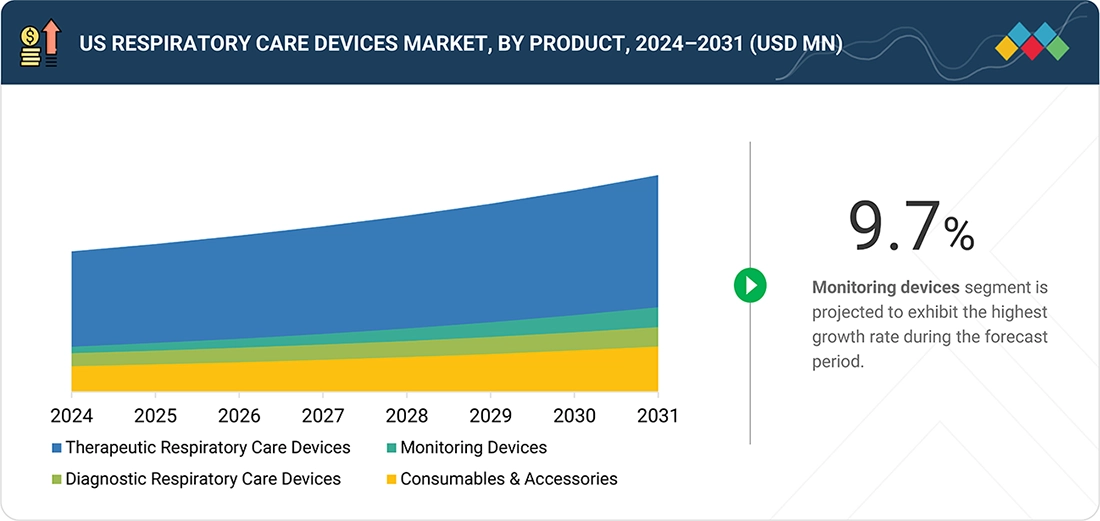
US Respiratory Care Devices Market, By Product
Among the different product segments in the US respiratory care devices market, therapeutic respiratory care devices hold the largest share primarily because of their indispensable contribution to the treatment and subsequent management of chronic and acute respiratory disorders. For instance, ventilators, CPAP and BiPAP machines, oxygen therapy systems, and nebulizers are commonly used for the treatment of diseases like COPD, asthma, sleep apnea, and respiratory failure. The widespread occurrence of these illnesses as well as an increase in the elderly population and the emergence of new post, COVID respiratory complications are major demand drivers of such therapeutic means. Besides that, a home, based care approach gaining momentum had led to a higher uptake of portable, simple, to, use therapeutic devices, especially for long, term oxygen therapy and sleep apnea management. Their regular use in both hospital and homecare settings, coupled with strong reimbursement backing and continuous innovation, has made therapeutic respiratory care devices the US market leader.
US Respiratory Care Devices Market, By Disease Indication
Among the disease indications segment, chronic obstructive pulmonary disease (COPD) holds the largest share in the US respiratory care devices market, mainly because of the high prevalence and long, term nature of the disease. COPD is one of the principal causes of sickness and death in the US, which is mostly due to the aging population, a history of smoking, exposure to occupational pollutants, and rising levels of air pollution. Many COPD patients generally need oxygen therapy systems, nebulizers, ventilators, and non, invasive ventilation devices on a regular basis, or certainly more than once, to control their symptoms and avoid the disease getting worse. Moreover, the increasing number of hospital stays and the rising trend of providing respiratory care at home for COPD patients have helped to increase the demand for devices. The continuing and worsening nature of COPD, together with its heavy reliance on therapeutic and monitoring devices, has made it the leading disease indication in the US respiratory care devices market.
US Respiratory Care Devices Market, By End User
Hospitals make up the largest portion of the distributor segment of the respiratory care devices market in the US because they are the main centers for the treatment of acute and severe respiratory cases. Hospitals serve as the main point of care for a large number of respiratory patients such as those experiencing respiratory failure, COPD exacerbations, pneumonia, trauma, and post, surgical respiratory complications. Hence, they have at their disposal the most recent respiratory support devices like ventilators, oxygen therapy systems, and monitoring equipment. The respiratory care in Intensive care units (ICUs) and emergency departments is thus largely dependent on the use of the latest respiratory care devices for managing critical patients. Moreover, hospitals with their skilled healthcare professionals, state, of, the, art infrastructure, and high budgets for medical equipment can buy and upgrade to the most technologically advanced respiratory devices faster than any other healthcare facility. The ongoing prevalence of respiratory diseases together with the demand for immediate, high, acuity care make hospitals the predominant end users in the respiratory care devices market in the US.
US RESPIRATORY CARE DEVICES MARKET: COMPANY EVALUATION MATRIX
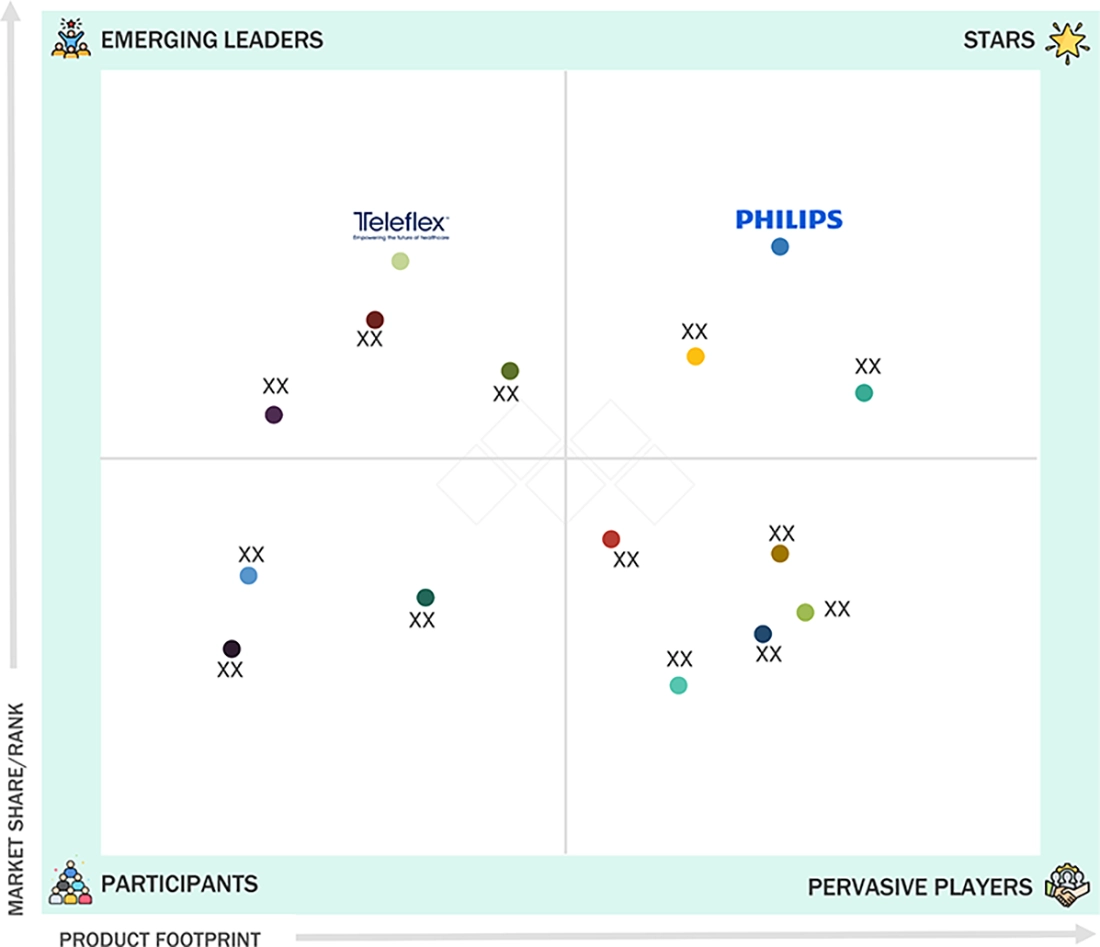
In the North America respiratory care devices market matrix, Koninklijke Philips N.V. (Star) leads with scale, extensive distribution, and a comprehensive portfolio of monitoring solutions. Masimo (Emerging Leader) is gaining momentum by offering innovative capnography devices that enhance clinical accuracy, improve patient safety, and support efficient respiratory monitoring. While Koninklijke Philips N.V. dominates through its regional reach and established presence, Masimo's focus on technological innovation positions it for rapid growth toward the leaders’ quadrant.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 9.40 Billion |
| Market Forecast in 2031 (Value) | USD 14.56 Billion |
| Growth Rate | CAGR of 7.8% from 2025–2031 |
| Years Considered | 2024–2031 |
| Base Year | 2024 |
| Forecast Period | 2025–2031 |
| Units Considered | Value (USD Million/Billion), Volume (Thousand Units) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Country Covered | US |
| Parent & Related Segment Reports |
Respiratory Care Devices Market Europe Respiratory Care Device Market Asia Pacific Respiratory Care Device Market North America Respiratory Care Device Market |
WHAT IS IN IT FOR YOU: US RESPIRATORY CARE DEVICES MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Volume Analysis | Market assessment by volume (units) for dental equipment used in digital dentures ecosystem |
|
| Product Analysis | Further breakdown of other applications in the market | Insights on other applications involved in the market |
| Company Information | Market share analysis, by country (US), and competitive leadership mapping for established players in the market | Insights on market share analysis by country |
RECENT DEVELOPMENTS
- December 2025 : ResMed received FDA approval for its new Smart Comfort feature, which uses AI. This feature collects actual sleep data and employs machine learning to recommend optimal comfort settings for CPAP therapy, tailored to each individual's needs. Smart Comfort is part of a new sleep device that automatically calibrates to the user's requirements. It helps new sleep apnea patients gradually acclimate to their treatment by determining the best settings that balance pressure and comfort. This approach also increases the likelihood that patients will adhere to their treatment over the long term. The launch of the AirSense 11 devices and the myAir app in the US is scheduled for early 2026, and Smart Comfort will be included in this rollout.
- April 2025 : Drägerwerk AG & Co. KGaA received US FDA 510(k) clearance for its VentStar Resus and VentStar Autobreath heated emergency ventilators. This clearance indicates that these devices are fundamentally similar to other legally marketed products. With this approval, the company is allowed to sell these powered emergency ventilators as Class II devices in the US. However, they must comply with general regulatory requirements, including labeling, manufacturing quality, and device registration.
Table of Contents

Methodology
This study involved four major activities in estimating the current US respiratory care devices market size. First, extensive secondary research was conducted to gather information on the market, including related and parent markets. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. After that, market breakdown and data triangulation were used to estimate the market size of segments and subsegments.
Secondary Research
The secondary research process involved the use of secondary sources, directories, databases (such as Bloomberg Businessweek, Factiva, and D&B Hoovers), white papers, annual reports, company house documents, investor presentations, and companies' SEC filings. Secondary research was used to identify and collect information useful for the extensive, technical, market-oriented, and commercial study of the US respiratory care devices market. It was also used to obtain important information on key players, market classification, and segmentation aligned with industry trends to the bottom-most level, and key developments from market and technology perspectives. A database of the key industry leaders was also prepared using secondary research.
Primary Research
In the primary research process, various sources from both the supply and demand sides were interviewed to obtain qualitative and quantitative information for this report. The primary sources on the supply side include industry experts such as CEOs, vice presidents, marketing and sales directors, technology & innovation directors, and other key executives from various companies and organizations operating in the US respiratory care devices market. The primary sources on the demand side included industry experts, purchasing & sales managers, doctors, and personnel from research organizations. Primary research was conducted to validate the market segmentation, identify key players, and gather insights into key industry trends and market dynamics.
A breakdown of the primary respondents for the US respiratory care devices market is provided below:
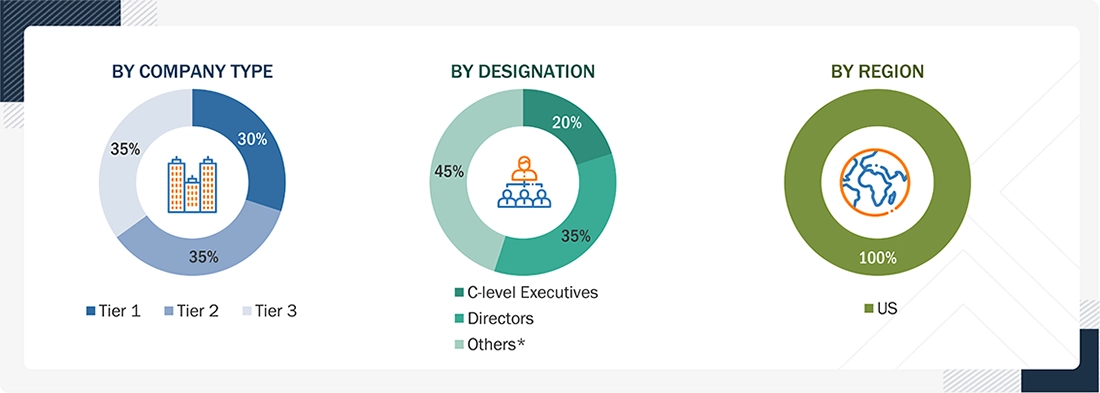
*Others include sales managers, marketing managers, business development managers, product managers, distributors, and suppliers.
Note 1: C-level executives include CEOs, CFOs, COOs, and VPs.
Note 2: Companies are classified into tiers based on their total revenue. As of 2025, Tier 1 = >USD 10.00 billion, Tier 2 = USD 1.00 billion to USD 10.00 billion, and Tier 3 = <USD 1.00 billion.
Source: MarketsandMarkets Analysis
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
For the regional market value, annual revenues were calculated using revenue mapping for major product manufacturers and OEMs active in the US respiratory care devices market. All the major product manufacturers were identified at the country level. Revenue mapping for the respective business segments/subsegments was done for the major players. Also, the US respiratory care devices market was split into various segments and subsegments based on:
- List of major players operating in the products market at the country level
- Product mapping of various US respiratory care device manufacturers at the country level
- Mapping of annual revenue generated by listed major players from US respiratory care devices (or the nearest reported business unit/product category)
- Revenue mapping of major players covered
- Extrapolation of the revenue mapping of the listed major players to derive the US market value of the respective segments/subsegments
- Summation of the market value of all segments/subsegments to arrive at the US respiratory care devices market
The consolidated data was enhanced with detailed insights and analysis from MarketsandMarkets and presented in this report.
Top-Down and Bottom-Up Approach

Data Triangulation
After determining the overall size of the US respiratory care devices market using the above-mentioned methodology, the market was split into several segments and subsegments. The data triangulation and market breakdown procedures were employed, as applicable, to complete the overall market engineering process and obtain exact market value data for the key segments and subsegments. The extrapolated market data was triangulated by studying various macroindicators and regional trends from both demand- and supply-side participants.
Market Definition
Respiratory care is a healthcare specialty that provides diagnostic evaluation, management, control, therapy, and monitoring of cardiopulmonary system diseases and disorders. Respiratory care devices are widely used in general & surgical wards and ICUs as a life-support measure.
Key Stakeholders
- Respiratory care device manufacturers and distributors
- Healthcare institutions (hospitals, laboratories, medical schools, and outpatient clinics)
- Research institutes
- Contract manufacturing organizations (CMOs)
- Government associations
- Market research and consulting firms
- Venture capitalists and investors
Report Objectives
- To define, describe, and forecast the US respiratory care devices market based on product, disease indication, and end user
- To forecast the market size in the US
- To strategically analyze industry trends, technology trends, pricing analysis, regulatory landscape, supply/value chain, ecosystem mapping, Porter’s Five Forces, patent analysis, key stakeholders, buying criteria, and conference & events
- To provide detailed information regarding the major factors influencing the growth of this market (such as drivers, restraints, opportunities, and challenges)
- To analyze the micromarkets with respect to individual growth trends, prospects, and contributions to the overall US respiratory care devices market
- To analyze the opportunities for stakeholders and provide details of the competitive landscape for market leaders
- To strategically profile key players in the US respiratory care devices market and comprehensively analyze their core competencies
- To track and analyze competitive developments, including product launches & approvals, partnerships, agreements, collaborations, joint ventures, expansions, and acquisitions, across the overall US respiratory care devices market
- To benchmark players within the market using the proprietary “Company Evaluation Matrix” framework, which analyzes market players on various parameters within the broad categories of business and product/service strategy
- To analyze the impact of generative AI on the US respiratory care devices market
Available customizations:
Based on the given market data, MarketsandMarkets offers customizations tailored to your company’s specific needs. The following customization options are available for the report:
COMPETITIVE LANDSCAPE ASSESSMENT
- Market share analysis, by country (US), which provides market shares of the top 3–5 key players in the US respiratory care devices market
- Competitive leadership mapping for established players in the US
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the US Respiratory Care Device Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS




















Growth opportunities and latent adjacency in US Respiratory Care Device Market