This research study involved the extensive use of secondary sources, directories, and databases to identify and collect valuable information for the analysis of the global continuous bioprocessing market. In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, to obtain and verify critical qualitative and quantitative information and assess the growth prospects of the market. The global market size estimated through secondary research was then triangulated with inputs from primary research to arrive at the final market size.
Secondary Research
Secondary research was used mainly to identify and collect information for the extensive, technical, market-oriented, and commercial study of the continuous bioprocessing market. The secondary sources used for this study include World Health Organization (WHO), National Institutes of Health (NIH), United States Food and Drug Administration (US FDA), National Institute for Bioprocessing Research and Training (NIBRT), Society for Biological Engineering (SBE), Bio-Process Systems Alliance (BPSA), European Patent Office (EPO), India Brand Equity Foundation (IBEF), Biotechnology Innovation Organization (BIO), National Center for Biotechnology Information (NCBI), BioPharm International, EvaluatePharma, ScienceDirect, Eurostat, research journals; corporate filings such as annual reports, SEC filings, investor presentations, and financial statements; press releases; trade, business, professional associations and among others. These sources were also used to obtain key information about major players, market classification, and segmentation according to industry trends, regional/country-level markets, market developments, and technology perspectives.
Primary Research
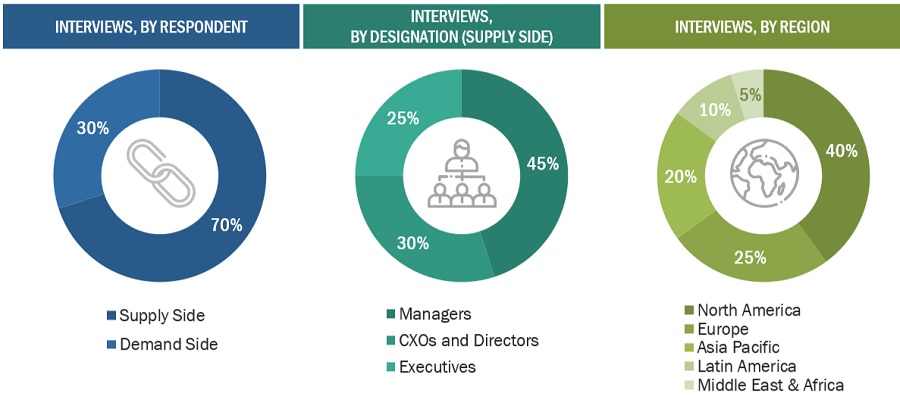
Extensive primary research was conducted after acquiring basic knowledge about the global continuous bioprocessing market scenario through secondary research. Several primary interviews were conducted with market experts from the demand side, such as pharmaceutical and biopharmaceutical companies, CROs, CMOs, and academic & research institutes, and experts from the supply side, such as C-level and D-level executives, product managers, marketing & sales managers of key manufacturers, distributors, and channel partners. These interviews were conducted across five major regions, including the Asia Pacific, North America, Europe, Latin America, and the Middle East & Africa. Approximately 70% and 30% of the primary interviews were conducted with supply-side and demand-side participants, respectively. This primary data was collected through questionnaires, e-mails, online surveys, personal interviews, and telephonic interviews.
The following is a breakdown of the primary respondents:
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
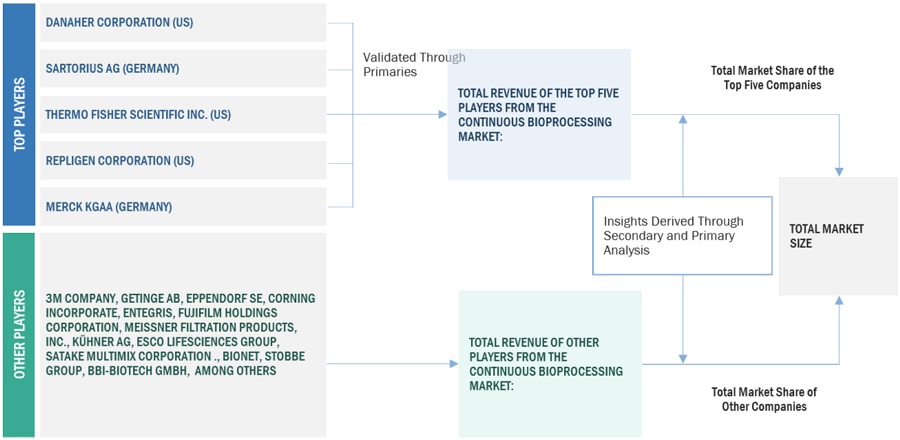
Both top-down and bottom-up approaches were used to estimate and validate the total size of the continuous bioprocessing market. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
Bottom-up Approach
-
The key players in the industry and market have been identified through extensive secondary research
-
The revenues generated from the single-use assemblies business of leading players have been determined through primary and secondary research
-
All percentage shares, splits, and breakdowns have been determined using secondary sources and verified through primary sources
To know about the assumptions considered for the study, Request for Free Sample Report
Top-down Approach
After arriving at the overall market size from the market size estimation process, the total market was split into several segments and subsegments.

Data Triangulation
After arriving at the market size from the market size estimation process explained above, the total market was divided into several segmentsand subsegments. To complete the overall market engineering process and arrive at the exact statistics for all segments and subsegments, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Continuous bioprocessing is a method used in the biopharmaceutical industry for the production of biologically derived products, such as biopharmaceuticals, vaccines, and other biologics, through continuous and uninterrupted operations. In the context of continuous bioprocessing, perfusion refers to a method of cell culture and bioproduction where a constant flow of fresh culture media is supplied to a bioreactor, and a corresponding volume of spent media, cells, or bioproducts is continuously removed. This is in contrast to traditional batch bioprocessing, where the culture is grown in a single vessel and harvested at the end of the production cycle.
Stakeholders
-
Continuous bioprocessing product manufacturers and distributors
-
Pharmaceutical and biotechnology companies
-
Healthcare service providers (including hospitals and clinics)
-
R&D companies
-
Academic institutes
-
Government associations
-
Market research and consulting firms
-
Venture capitalists and investors
Report Objectives
-
To define, describe, and forecast the global continuous bioprocessing market based on the product, scale of operation, process, application, end user and region
-
To provide detailed information regarding the major factors influencing the growth of the market (such as drivers, restraints, opportunities and challenges)
-
To strategically analyze micro-markets with respect to individual growth trends, future prospects, and contributions to the overall continuous bioprocessing market
-
To analyze opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders
-
To forecast the size of the market segments with respect to five main regions, namely, North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa
-
To strategically profile the key players and comprehensively analyze their product portfolios, market positions, and core competencies
-
To track and analyze competitive developments such as acquisitions, product launches, expansions, and R&D activities in the continuous bioprocessing market.
Available Customizations
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for this report:
Portfolio Assessment
-
Product Matrix, which gives a detailed comparison of the product portfolios of the top three companies.
Company Information
-
Detailed analysis and profiling of additional market players (up to three).
Geographical Analysis
-
A further breakdown of the Rest of Asia Pacific continuous bioprocessing market into countries
-
A further breakdown of the Rest of European continuous bioprocessing market into countries
-
A further breakdown of the Rest of Latin American continuous bioprocessing market into countries



Growth opportunities and latent adjacency in Continuous Bioprocessing Market