The research study involved the wide use of secondary sources, directories, and databases to identify and collect valuable information for the analysis of the global vaccine adjuvants market. Widespread interviews were conducted with various primary participants, including key industry members, subject-matter experts (SMEs), C-level managers of leading market players, and industry consultants, to obtain and verify qualitative and quantitative information and evaluate the growth scenarios of the market. The global market size is estimated through secondary research (top-down and bottom-up) followed by data triangulation with inputs from industry experts to arrive at the final market size.
Secondary Research
Secondary research was used mainly to identify and collect information for the technical, market-oriented, and commercial study of the vaccine adjuvants market. The secondary sources used for this study include World Health Organization (WHO), Centers for Disease Control and Prevention (CDC), European Medicines Agency (EMA), U.S. Food and Drug Administration (FDA), National Institutes of Health (NIH), Health Canada, European Centre for Disease Prevention and Control (ECDC), Academic Journals and Company Websites, corporate & regulatory filings, such as annual reports, SEC filings, investor presentations, and financial statements; business magazines & research journals; press releases; and trade, business, and professional associations among others. These secondary sources were also used to obtain major information about key market players, and market segmentation corresponding to industry trends, regional/country-level markets, market developments, and technology prospects. Secondary data was collected and analysed to arrive at the market size of the global vaccine adjuvants market, which was further validated through primary research.
Primary Research
Extensive primary research was conducted after acquiring basic knowledge about the global vaccine adjuvants market scenario through secondary research. Several primary interviews were conducted with market experts from the demand side, such as pharmaceutical & biopharmaceutical industries from the supply side, such as C-level and D-level executives, product managers, marketing & sales managers of key manufacturers, distributors, and channel partners. The primary interviews were conducted across six major regions, including the North America, Europe, Asia Pacific, Latin America, Middle East and Africa. Approximately 65% and 35% of the primary interviews were conducted with supply-side and demand-side participants, respectively. This primary data was collected through questionnaires, e-mails, online surveys, personal interviews, and telephonic interviews.
The following is a breakdown of the primary respondents:
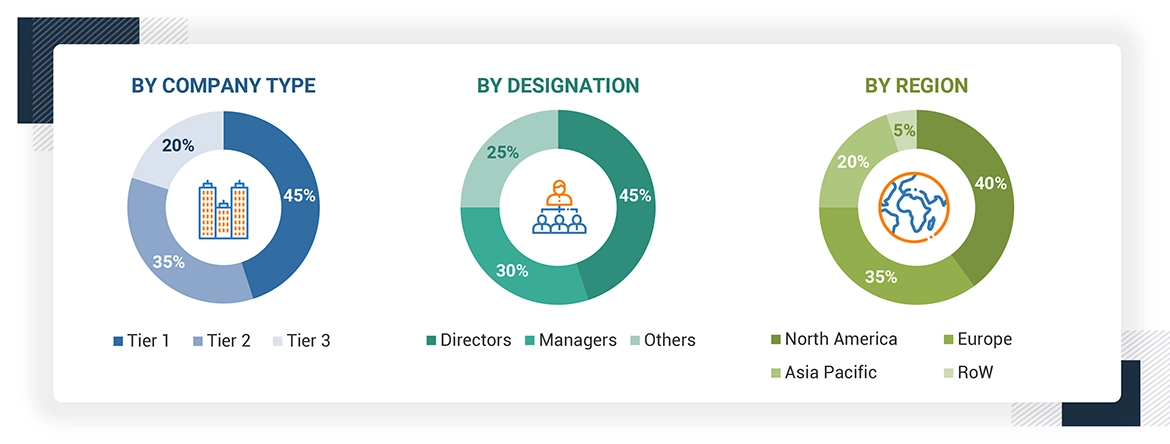
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
Both top-down and bottom-up approaches were used to estimate and validate the total size of the vaccine adjuvants market. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
Data Triangulation
After arriving at the market size from the market size estimation process explained above, the total market was divided into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics for all segments and subsegments, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Vaccine adjuvants are substances added to vaccines to enhance the body's immune response to the vaccine's active ingredient (the antigen). They work by stimulating or modulating the immune system, helping to produce a stronger, longer lasting, or more specific immune response. The market size includes vaccine adjuvants produced by vaccine adjuvant manufacturers and utilized by key market players in vaccine development and production. However, vaccine adjuvants developed and used inhouse by companies for their own vaccine development and manufacturing are excluded from the scope.
Stakeholders
-
Companies Providing Vaccine Adjuvants
-
Research Institutes and Academic Centers
-
Suppliers and Distributors of Vaccine adjuvants
-
Pharmaceutical and Biotechnology Companies
-
Veterinary companies
-
Business Research and Consulting Service Providers
-
Venture Capitalists
-
Contract Research Organizations and Contract Development Manufacturing Organizations
-
Market research and consulting firms
Report Objectives
-
To define, describe, and forecast the vaccine adjuvants market by value by product, adjuvant type, application, route of administration, vaccine type, disease type and region
-
To provide detailed information regarding the major factors influencing the market growth (such as drivers, restraints, opportunities, and challenges)
-
To analyze the micromarkets1 with respect to individual growth trends, prospects, and contributions to the overall vaccine adjuvants market
-
To analyze the opportunities for stakeholders and provide details of the competitive landscape for market leaders
-
To forecast the size of the market segments with respect to six regions: North America, Europe, the Asia Pacific, Latin America, Middle East and Africa
-
To profile the key players and analyze their market shares and core competencies2
-
To track and analyze competitive developments, such as product launches, partnerships, agreements, collaborations, and expansions
-
To benchmark players within the market using the proprietary “Company Evaluation Matrix” framework, which analyzes market players on various parameters within the broad categories of business and product excellence strategy



Growth opportunities and latent adjacency in Vaccine Adjuvants Market