4
MARKET OVERVIEW
Emerging CAR T-cell therapies poised to revolutionize cancer treatment despite cost and safety hurdles.
44
4.2.1.1
TECHNOLOGICAL ADVANCEMENTS IN CAR T-CELL THERAPIES
4.2.1.2
LABEL EXPANSIONS AND STRONG EFFICACY SIGNALS IN HEMATOLOGIC CANCERS
4.2.1.3
GROWING INVESTMENT IN CAR T-CELL THERAPY DEVELOPMENT
4.2.2.1
HIGH COST AND REIMBURSEMENT CHALLENGES FOR CAR T-CELL THERAPY
4.2.3.1
EXPANDING BEYOND HEMATOLOGIC MALIGNANCIES INTO SOLID TUMORS AND NEW DISEASE AREAS
4.2.3.2
FOCUS SHIFT ON ALLOGENEIC CAR-T AND CAR-NK
4.2.3.3
GROWING COLLABORATIONS AMONG BIOPHARMACEUTICAL COMPANIES, ACADEMIC INSTITUTIONS, AND RESEARCH ORGANIZATIONS
4.2.4.1
LONG-TERM SAFETY SURVEILLANCE AND EVOLVING LABELING REQUIREMENTS
4.3
UNMET NEEDS & WHITE SPACES
4.4
INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
4.5
STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5
INDUSTRY TRENDS
Navigate competitive pressures and pricing shifts in the evolving global CAR-T-cell therapy landscape.
55
5.1
PORTER’S FIVE FORCES ANALYSIS
5.1.1
THREAT OF NEW ENTRANTS
5.1.2
THREAT OF SUBSTITUTES
5.1.3
BARGAINING POWER OF BUYERS
5.1.4
BARGAINING POWER OF SUPPLIERS
5.1.5
INTENSITY OF COMPETITIVE RIVALRY
5.2
MACROECONOMIC OUTLOOK
5.2.2
GDP TRENDS AND FORECASTS
5.2.3
TRENDS IN GLOBAL CAR-T-CELL THERAPY MARKET
5.5.1
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY TYPE, 2025
5.5.2
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY KEY PLAYER, 2025
5.5.3
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY REGION, 2025
5.6
KEY CONFERENCES & EVENTS, 2026–2027
5.7
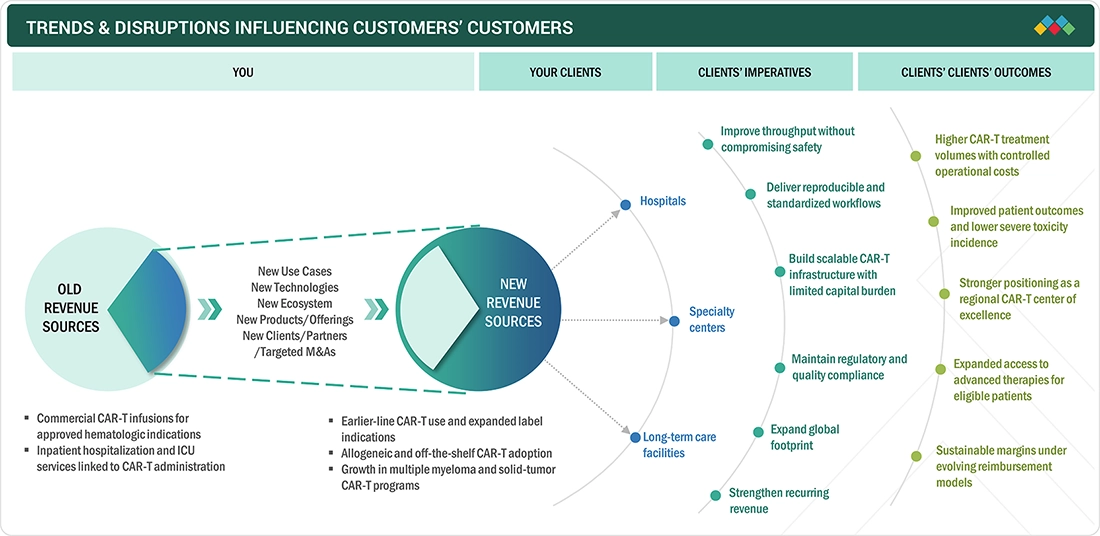
TRENDS/DISRUPTIONS IMPACTING CUSTOMER’S BUSINESS
5.8
INVESTMENT/FUNDING SCENARIO
5.9
IMPACT OF 2025 US TARIFF ON CAR T-CELL THERAPY MARKET
5.9.2
PRICE IMPACT ANALYSIS
5.9.3
IMPACT ON COUNTRY/REGION
5.9.4
IMPACT ON END-USE INDUSTRIES
5.9.4.2
SPECIALTY ONCOLOGY CENTERS
5.9.4.3
LONG-TERM CARE FACILITIES
6
TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
AI-driven innovations revolutionize CAR T-cell therapy with cutting-edge gene editing and imaging technologies.
72
6.1.1.1
CAR DESIGN AND OPTIMIZATION
6.1.1.2
VIRAL VECTOR TECHNOLOGY
6.1.1.3
CELL CULTURE AND EXPANSION TECHNIQUES
6.1.2
COMPLEMENTARY TECHNOLOGIES
6.1.2.1
GENE EDITING TECHNOLOGY (CRISPR-CAS9-BASED GENOME EDITING)
6.1.3
ADJACENT TECHNOLOGIES
6.1.3.1
MONITORING AND IMAGING TECHNOLOGIES
6.2
TECHNOLOGY/PRODUCT ROADMAP
6.4
IMPACT OF AI/GEN AI ON CAR T-CELL THERAPY MARKET
7
SUSTAINABILITY AND REGULATORY LANDSCAPE
Navigate complex global regulations to leverage eco-standards and optimize CAR T-cell therapy compliance.
77
7.1
REGIONAL REGULATIONS AND COMPLIANCE
7.1.1
REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
7.1.2
REGULATORY FRAMEWORK
7.2
SUSTAINABILITY IMPACT AND REGULATORY POLICY INITIATIVES
7.3
CERTIFICATIONS, LABELING, AND ECO-STANDARDS
7.4
REIMBURSEMENT SCENARIO
7.5
COMBINATION THERAPIES
8
CUSTOMER LANDSCAPE & BUYER BEHAVIOR
Unlocking buyer insights: Navigate stakeholder dynamics and unmet needs to boost market profitability.
86
8.1
DECISION-MAKING PROCESS
8.2
BUYER STAKEHOLDERS AND BUYING EVALUATION CRITERIA
8.2.1
KEY STAKEHOLDERS IN BUYING PROCESS
8.2.2
KEY BUYING CRITERIA, BY END USER
8.3
ADOPTION BARRIERS & INTERNAL CHALLENGES
8.4
UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
9
CAR T-CELL THERAPY MARKET, BY PRODUCT
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 51 Data Tables
90
9.2.1
FOCUS ON INCREASED RESEARCH ACTIVITIES FOR INDICATION EXPANSIONS TO DRIVE MARKET
9.3.1
NEED FOR TREATMENT OF MULTIPLE INDICATIONS TO PROPEL MARKET GROWTH
9.4.1
GLOBAL REACH AND SIGNIFICANT REVENUE GROWTH TO SUPPORT MARKET
9.5.1
FOCUS ON REGULATORY APPROVALS ACROSS MAJOR REGIONS TO BOOST MARKET GROWTH
9.6.1
EFFORTS TO SECURE REIMBURSEMENT AND IMPROVE MARKET ACCESS FACILITATE PATIENT TO AID MARKET ADOPTION
9.7.1
APPROVAL FOR PEDIATRIC USE TO AUGMENT MARKET GROWTH
10
CAR T-CELL THERAPY MARKET, BY TARGET
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 38 Data Tables
117
10.2.1
RISING INDICATION EXPANSIONS AND NEW APPROVALS TO DRIVE MARKET
10.3.1
GROWING PIPELINE PRODUCTS TO PROPEL MARKET GROWTH
10.4.1
DUAL TARGETING TO OFFER COMPETITIVE ADVANTAGE IN ANTIGEN ESCAPE AND TUMOR HETEROGENEITY ISSUES
10.5.1
ALLOGENEIC CELL SOURCE OF CD7 THERAPIES IN DEVELOPMENT TO SUPPORT GROWTH
11
CAR T-CELL THERAPY MARKET, BY INDICATION
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 29 Data Tables
136
11.2
B-CELL LYMPHOMA (BCL)
11.2.1
RISING PREVALENCE OF B-CELL LYMPHOMAS TO PROPEL MARKET GROWTH
11.3.1
GROWING PRODUCT REVENUE AND RISING GLOBAL INCIDENCE OF MYELOMA TO DRIVE MARKET
11.4
ACUTE LYMPHOBLASTIC LEUKEMIA (ALL)
11.4.1
EXPANDING R&D PIPELINE TO AUGMENT MARKET GROWTH
12
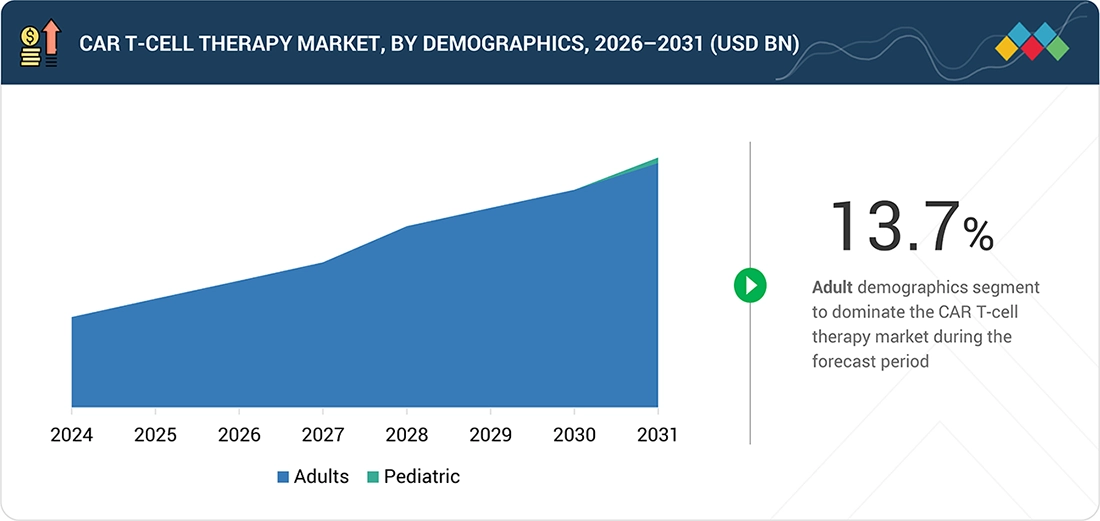
CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 15 Data Tables
152
12.2.1
GROWING PREVALENCE OF HEMATOLOGIC CANCERS TO DRIVE MARKET
12.3.1
HIGH UNMET NEEDS TO LIMIT SEGMENT GROWTH
13
CAR T-CELL THERAPY MARKET, BY END USER
Market Size & Growth Rate Forecast Analysis to 2031 in USD Million | 22 Data Tables
160
13.2.1
GROWING NEED FOR STANDARD ONCOLOGY TREATMENT PROTOCOLS TO DRIVE MARKET
13.3.1
NEED FOR OPTIMAL PATIENT CARE AND SPECIALIZED TREATMENT OPTIONS TO SPUR MARKET GROWTH
13.4
LONG-TERM CARE FACILITIES
13.4.1
INCREASING INCIDENCE OF CHRONIC DISORDERS AMONG GERIATRIC POPULATION TO PROPEL MARKET GROWTH
14
CAR T-CELL THERAPY MARKET, BY REGION
Comprehensive coverage of 7 Regions with country-level deep-dive of 15 Countries | 137 Data Tables.
172
14.2.1.1
US TO DOMINATE CAR T-CELL THERAPY MARKET DURING STUDY PERIOD
14.2.2.1
REGULATORY APPROVALS AND GOVERNMENT INITIATIVES FOR REGENERATIVE MEDICINE RESEARCH TO DRIVE MARKET
14.3.1.1
KEY PRODUCT APPROVALS AND FOCUS ON CLINICAL RESEARCH TO AID MARKET GROWTH
14.3.2.1
TECHNOLOGICAL ADVANCEMENTS IN AUTOMATION AND PRESENCE OF ROBUST RESEARCH INFRASTRUCTURE TO SPUR MARKET GROWTH
14.3.3.1
GROWING FOCUS ON CELL & GENE THERAPY INITIATIVES TO BOOST MARKET GROWTH
14.3.4.1
GROWTH IN BIOTECH SECTOR TO AUGMENT MARKET GROWTH
14.3.5.1
RISING FOCUS ON CELL THERAPIES AND GROWING FOCUS ON STRINGENT REGULATIONS TO AUGMENT MARKET GROWTH
14.4.1.1
RISING NUMBER OF CAR T-CELL CLINICAL TRIALS TO FUEL MARKET GROWTH
14.4.2.1
INCREASING NUMBER OF PRODUCT APPROVALS AND RISING GERIATRIC POPULATION TO PROPEL MARKET GROWTH
14.4.3.1
GROWING FOCUS ON PRODUCT COMMERCIALIZATION AND RISING CANCER INCIDENCE TO BOOST MARKET DEMAND
14.4.4.1
ROBUST INFRASTRUCTURE FOR CLINICAL TRIALS AND WELL-DEVELOPED HEALTHCARE SECTOR TO AID MARKET GROWTH
14.4.5.1
GROWTH IN BIOPHARMACEUTICAL INDUSTRY TO FACILITATE MARKET GROWTH
14.4.6
REST OF ASIA PACIFIC
14.5.1.1
PRODUCT APPROVALS AND HIGH HEALTHCARE EXPENDITURE TO SUPPORT MARKET GROWTH
14.5.2
REST OF LATIN AMERICA
14.6.2
KINGDOM OF SAUDI ARABIA
14.6.2.1
INCREASED NUMBER OF PRODUCT APPROVALS AND HIGH HEALTHCARE EXPENDITURE TO BOOST MARKET GROWTH
14.6.3.1
UAE MARKET GROWTH TO BE DRIVEN BY LOCALIZED MANUFACTURING, EXPANDED CLINICAL APPLICATIONS, AND STRATEGIC COLLABORATIONS
14.6.4
REST OF MIDDLE EAST
14.7.1
SIGNIFICANT BURDEN OF HEMATOLOGIC MALIGNANCIES AND INFECTIOUS DISEASES TO PROPEL MARKET GROWTH
15
COMPETITIVE LANDSCAPE
Discover strategic insights and market positioning of key players in CAR T-cell therapy.
235
15.2
KEY PLAYER STRATEGY/RIGHT TO WIN
15.2.1
OVERVIEW OF MAJOR STRATEGIES ADOPTED BY PLAYERS IN CAR T-CELL THERAPY MARKET
15.3
REVENUE ANALYSIS, 2023–2025
15.4
MARKET SHARE ANALYSIS, 2025
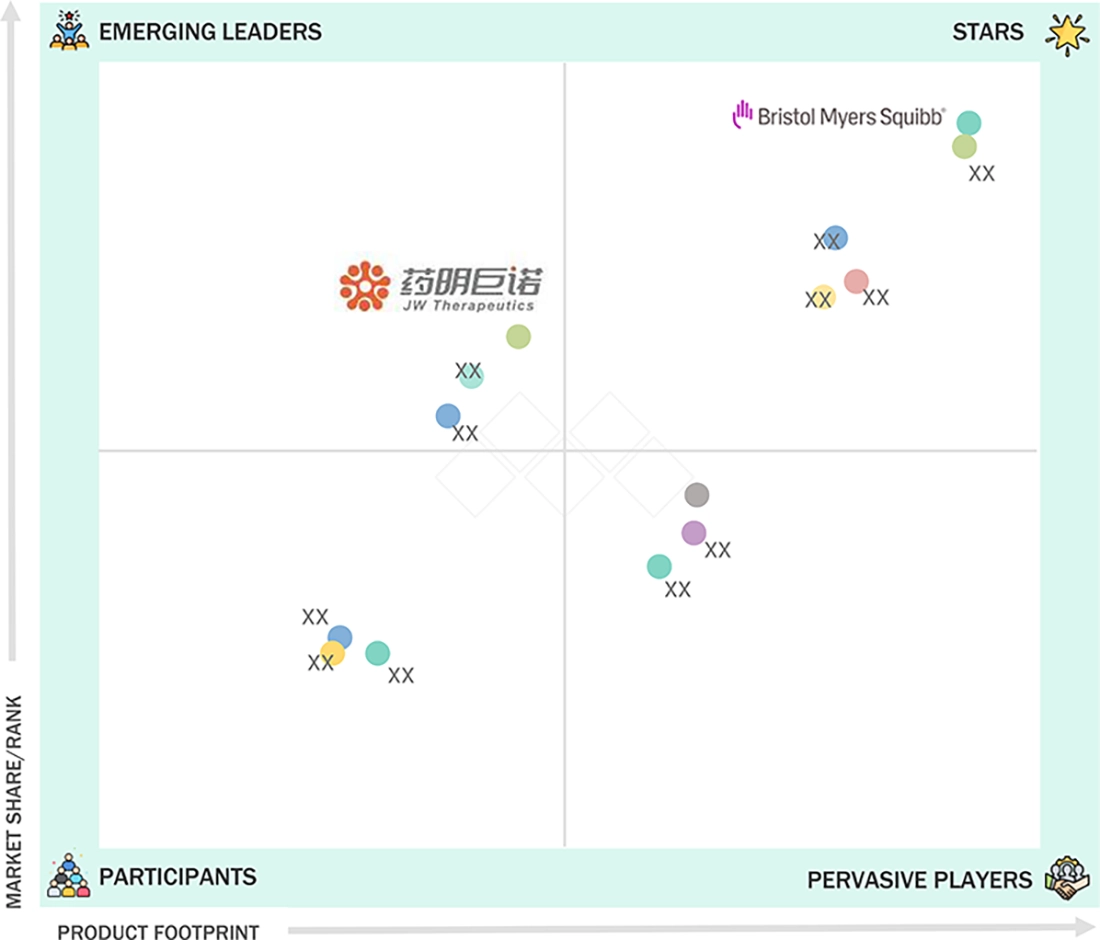
15.5
COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
15.5.5
COMPANY FOOTPRINT: KEY PLAYERS, 2025
15.5.5.1
COMPANY FOOTPRINT
15.5.5.2
REGION FOOTPRINT
15.5.5.3
PRODUCT FOOTPRINT
15.5.5.4
TARGET FOOTPRINT
15.5.5.5
INDICATION FOOTPRINT
15.6
COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
15.6.1
PROGRESSIVE COMPANIES
15.6.2
RESPONSIVE COMPANIES
15.6.5
COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
15.6.5.1
DETAILED LIST OF KEY STARTUPS/SMES
15.6.5.2
COMPETITIVE BENCHMARKING OF STARTUPS/SMES
15.7
COMPANY VALUATION & FINANCIAL METRICS
15.8
BRAND/PRODUCT COMPARISON
15.9
COMPETITIVE SCENARIO
15.9.4
OTHER DEVELOPMENTS
16
COMPANY PROFILES
In-depth Company Profiles of Leading Market Players with detailed Business Overview, Product and Service Portfolio, Recent Developments, and Unique Analyst Perspective (MnM View)
255
16.1.1.1
BUSINESS OVERVIEW
16.1.1.2
PRODUCTS OFFERED
16.1.1.3
RECENT DEVELOPMENTS
16.1.1.3.1
PRODUCT APPROVALS
16.1.1.4.2
STRATEGIC CHOICES
16.1.1.4.3
WEAKNESSES & COMPETITIVE THREATS
16.1.2
GILEAD SCIENCES, INC.
16.1.3
BRISTOL-MYERS SQUIBB COMPANY
16.1.5
AUTOLUS THERAPEUTICS
16.1.6
JW (CAYMAN) THERAPEUTICS CO., LTD.
16.1.7
IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT)
16.1.8
CARSGEN THERAPEUTICS HOLDINGS LIMITED
16.1.9
IASO BIOTHERAPEUTICS
16.1.10
IMMUNEEL THERAPEUTICS
16.2.2
CARTESIAN THERAPEUTICS, INC.
16.2.3
ALLOGENE THERAPEUTICS
16.2.4
LYELL IMMUNOPHARMA, INC.
16.2.5
KYVERNA THERAPEUTICS, INC.
16.2.8
ATARA BIOTHERAPEUTICS, INC.
16.2.9
CARIBOU BIOSCIENCES
16.2.11
CRISPR THERAPEUTICS
16.2.12
POSEIDA THERAPEUTICS
17
RESEARCH METHODOLOGY
299
17.1.1.1
OBJECTIVES OF SECONDARY RESEARCH
17.1.2.1
BREAKDOWN OF PRIMARIES
17.1.2.2
OBJECTIVES OF PRIMARY RESEARCH
17.2
MARKET SIZE ESTIMATION
17.2.1
BOTTOM-UP APPROACH
17.2.2
INSIGHTS FROM PRIMARY EXPERTS
17.2.3
SEGMENTAL MARKET SIZE ESTIMATION
17.3
GROWTH RATE ASSUMPTIONS
17.3.2
IMPACT OF SUPPLY- AND DEMAND-SIDE FACTORS
17.5
RESEARCH LIMITATIONS
18.2
KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL
18.3
CUSTOMIZATION OPTIONS
TABLE 1
CAR T-CELL THERAPY MARKET: INCLUSIONS & EXCLUSIONS
TABLE 2
KEY PIPELINE PRODUCTS IN PHASE-2 AND PHASE-3 CAR T-CELL THERAPY MARKET (AS OF 2025)
TABLE 3
IMPACT OF PORTER’S FIVE FORCES ON CAR T-CELL THERAPY MARKET
TABLE 4
ROLE OF MAJOR COMPANIES IN CAR T-CELL THERAPY MARKET ECOSYSTEM (SUPPLY AND DEMAND SIDES)
TABLE 5
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY TYPE, 2025 (USD)
TABLE 6
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY REGION, 2025
TABLE 7
KEY CONFERENCES & EVENTS IN CAR T-CELL THERAPY MARKET, JANUARY 2026–DECEMBER 2027
TABLE 8
US-ADJUSTED RECIPROCAL TARIFF RATES
TABLE 9
NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 10
EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 11
ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 12
REST OF THE WORLD: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
TABLE 13
COUNTRY-WISE REGULATORY SCENARIO FOR CAR T-CELL THERAPY PRODUCT MANUFACTURERS
TABLE 14
CAR T-CELL THERAPY MARKET: REIMBURSEMENT STATUS AND FUNDING CHANNEL, BY COUNTRY/REGION (AS OF 2026)
TABLE 15
CAR T-CELL THERAPY MARKET: HCPCS CODES OF COMMERCIALIZED THERAPIES
TABLE 16
COMBINATION THERAPY TRIALS IN CAR T-CELL THERAPY MARKET
TABLE 17
INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS, BY END USER (%)
TABLE 18
KEY BUYING CRITERIA, BY END USER
TABLE 19
CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 20
CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (NUMBER OF THERAPIES)
TABLE 21
CAR T-CELL THERAPY MARKET FOR ABECMA, BY REGION, 2024–2031 (USD MILLION)
TABLE 22
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 23
EUROPE: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 24
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 25
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 26
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 27
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 28
CAR T-CELL THERAPY MARKET FOR BREYANZI, BY REGION, 2024–2031 (USD MILLION)
TABLE 29
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 30
EUROPE: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 31
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 32
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 33
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 34
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 35
CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY REGION, 2024–2031 (USD MILLION)
TABLE 36
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 37
EUROPE: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 38
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 39
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 40
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 41
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 42
CAR T-CELL THERAPY MARKET FOR YESCARTA, BY REGION, 2024–2031 (USD MILLION)
TABLE 43
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 44
EUROPE: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 45
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 46
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 47
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 48
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 49
CAR T-CELL THERAPY MARKET FOR TECARTUS, BY REGION, 2024–2031 (USD MILLION)
TABLE 50
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 51
EUROPE: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 52
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 53
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 54
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 55
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 56
CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY REGION, 2024–2031 (USD MILLION)
TABLE 57
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 58
EUROPE: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 59
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 60
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 61
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 62
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 63
CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY REGION, 2024–2031 (USD MILLION)
TABLE 64
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 65
EUROPE: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 66
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 67
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 68
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 69
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 70
CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 71
COMMERCIALLY AVAILABLE CD19 ANTIGEN TARGETING CAR T-CELL THERAPIES
TABLE 72
CD19-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 73
NORTH AMERICA: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 74
EUROPE: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 75
ASIA PACIFIC: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 76
LATIN AMERICA: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 77
MIDDLE EAST: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 78
GCC COUNTRIES: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 79
COMMERCIALLY AVAILABLE BCMA ANTIGEN TARGETING CAR T-CELL THERAPIES
TABLE 80
BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 81
NORTH AMERICA: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 82
EUROPE: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 83
ASIA PACIFIC: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 84
LATIN AMERICA: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 85
MIDDLE EAST: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 86
GCC COUNTRIES: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 87
CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 88
NORTH AMERICA: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 89
EUROPE: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 90
ASIA PACIFIC: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 91
LATIN AMERICA: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 92
MIDDLE EAST: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 93
GCC COUNTRIES: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 94
CD7-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 95
NORTH AMERICA: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 96
EUROPE: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 97
ASIA PACIFIC: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 98
LATIN AMERICA: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 99
MIDDLE EAST: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 100
GCC COUNTRIES: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 101
OTHER CAR T-CELL THERAPY TARGETS MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 102
NORTH AMERICA: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 103
EUROPE: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 104
ASIA PACIFIC: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 105
LATIN AMERICA: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 106
MIDDLE EAST: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 107
GCC COUNTRIES: OTHER CAR T-CELL THERAPY TARGETS MARKETA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 108
CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 109
CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY REGION, 2024–2031 (USD MILLION)
TABLE 110
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 111
EUROPE: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 112
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 113
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 114
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 115
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 116
CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY REGION, 2024–2031(USD MILLION)
TABLE 117
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 118
EUROPE: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 119
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 120
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 121
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 122
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELLOMA, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 123
CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY REGION, 2024–2031(USD MILLION)
TABLE 124
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031(USD MILLION)
TABLE 125
EUROPE: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031(USD MILLION)
TABLE 126
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031(USD MILLION)
TABLE 127
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 128
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 129
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 130
CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY REGION, 2024–2031 (USD MILLION)
TABLE 131
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 132
EUROPE: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 133
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 134
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 135
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 136
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 137
CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 138
CAR T-CELL THERAPY MARKET FOR ADULTS, BY REGION, 2024–2031 (USD MILLION)
TABLE 139
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 140
EUROPE: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 141
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 142
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 143
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 144
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 145
CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY REGION, 2024–2031 (USD MILLION)
TABLE 146
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 147
EUROPE: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 148
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 149
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 150
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 151
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 152
CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 153
CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY REGION, 2024–2031 (USD MILLION)
TABLE 154
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 155
EUROPE: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 156
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 157
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 158
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 159
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 160
CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY REGION, 2024–2031 (USD MILLION)
TABLE 161
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 162
EUROPE: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 163
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 164
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 165
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 166
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 167
CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY REGION, 2024–2031(USD MILLION)
TABLE 168
NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 169
EUROPE: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 170
ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 171
LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 172
MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 173
GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024–2031(USD MILLION)
TABLE 174
CAR T-CELL THERAPY MARKET, BY REGION, 2024–2031 (USD MILLION)
TABLE 175
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 176
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 177
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 178
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 179
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 180
NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 181
US: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 182
US: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 183
US: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 184
US: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 185
US: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 186
CANADA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 187
CANADA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 188
CANADA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 189
CANADA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 190
CANADA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 191
EUROPE: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 192
EUROPE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 193
EUROPE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 194
EUROPE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 195
EUROPE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 196
EUROPE: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 197
GERMANY: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 198
GERMANY: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 199
GERMANY: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 200
GERMANY: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 201
GERMANY: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 202
UK: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 203
UK: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 204
UK: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 205
UK: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 206
UK: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 207
FRANCE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 208
FRANCE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 209
FRANCE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 210
FRANCE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 211
FRANCE: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 212
ITALY: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 213
ITALY: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 214
ITALY: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 215
ITALY: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 216
ITALY: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 217
SPAIN: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 218
SPAIN: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 219
SPAIN: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 220
SPAIN: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 221
SPAIN: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 222
REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 223
REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 224
REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 225
REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 226
REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 227
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 228
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 229
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 230
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 231
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 232
ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 233
CHINA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 234
CHINA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 235
CHINA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 236
CHINA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 237
CHINA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 238
JAPAN: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 239
JAPAN: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 240
JAPAN: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 241
JAPAN: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 242
JAPAN: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 243
INDIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 244
INDIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 245
INDIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 246
INDIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 247
INDIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 248
AUSTRALIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 249
AUSTRALIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 250
AUSTRALIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 251
AUSTRALIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 252
AUSTRALIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 253
SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 254
SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 255
SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 256
SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 257
SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 258
REST OF ASIA PACIFC: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 259
REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 260
REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 261
REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 262
REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 263
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 264
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 265
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 266
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 267
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 268
LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 269
BRAZIL: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 270
BRAZIL: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 271
BRAZIL: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 272
BRAZIL: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 273
BRAZIL: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 274
REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 275
REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 276
REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 277
REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 278
REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 279
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 280
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 281
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 282
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 283
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 284
MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 285
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024–2031 (USD MILLION)
TABLE 286
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 287
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 288
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 289
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 290
GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 291
KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 292
KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 293
KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 294
KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 295
KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 296
UAE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 297
UAE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 298
UAE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 299
UAE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 300
UAE: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 301
REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 302
REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 303
REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 304
REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 305
REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 306
AFRICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024–2031 (USD MILLION)
TABLE 307
AFRICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024–2031 (USD MILLION)
TABLE 308
AFRICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024–2031 (USD MILLION)
TABLE 309
AFRICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024–2031 (USD MILLION)
TABLE 310
AFRICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024–2031 (USD MILLION)
TABLE 311
OVERVIEW OF MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CAR T-CELL THERAPY MARKET
TABLE 312
CAR T-CELL THERAPY MARKET: DEGREE OF COMPETITION
TABLE 313
CAR T-CELL THERAPY MARKET: REGION FOOTPRINT
TABLE 314
CAR T-CELL THERAPY MARKET: PRODUCT FOOTPRINT
TABLE 315
CAR T-CELL THERAPY MARKET: TARGET FOOTPRINT
TABLE 316
CAR T-CELL THERAPY MARKET: INDICATION FOOTPRINT
TABLE 317
CAR T-CELL THERAPY MARKET: DETAILED LIST OF KEY STARTUPS/SME PLAYERS
TABLE 318
CAR T-CELL THERAPY MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SME PLAYERS, BY DEMOGRAPHY AND REGION, 2025
TABLE 319
CAR T-CELL THERAPY MARKET: PRODUCT APPROVALS, JANUARY 2022–JANUARY 2026
TABLE 320
CAR T-CELL THERAPY MARKET: DEALS, JANUARY 2022–JANUARY 2026
TABLE 321
CAR T-CELL THERAPY MARKET: EXPANSIONS, JANUARY 2022–JANUARY 2026
TABLE 322
CAR T-CELL THERAPY MARKET: OTHER DEVELOPMENTS, JANUARY 2022–JANUARY 2026
TABLE 323
JOHNSON & JOHNSON: COMPANY OVERVIEW
TABLE 324
JOHNSON & JOHNSON: PRODUCTS OFFERED
TABLE 325
JOHNSON & JOHNSON: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 326
JOHNSON & JOHNSON: DEALS, JANUARY 2022−JANUARY 2026
TABLE 327
GILEAD SCIENCES, INC.: COMPANY OVERVIEW
TABLE 328
GILEAD SCIENCES, INC.: PRODUCTS OFFERED
TABLE 329
GILEAD SCIENCES, INC.: PRODUCT APPROVALS, JANUARY 2022–JANUARY 2026
TABLE 330
GILEAD SCIENCES, INC.: DEALS, JANUARY 2022–JANUARY 2026
TABLE 331
GILEAD SCIENCES, INC.: OTHER DEVELOPMENTS, JANUARY 2022–JANUARY 2026
TABLE 332
BRISTOL-MYERS SQUIBB COMPANY: COMPANY OVERVIEW
TABLE 333
BRISTOL-MYERS SQUIBB COMPANY: PRODUCTS OFFERED
TABLE 334
BRISTOL-MYERS SQUIBB COMPANY: PRODUCT APPROVALS, JANUARY 2022–JANUARY 2026
TABLE 335
BRISTOL-MYERS SQUIBB COMPANY: DEALS, JANUARY 2022–JANUARY 2026
TABLE 336
NOVARTIS AG: COMPANY OVERVIEW
TABLE 337
NOVARTIS AG: PRODUCTS OFFERED
TABLE 338
NOVARTIS AG: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 339
NOVARTIS AG: OTHER DEVELOPMENTS, JANUARY 2022−JANUARY 2026
TABLE 340
AUTOLUS THERAPEUTICS: COMPANY OVERVIEW
TABLE 341
AUTOLUS THERAPEUTICS: PRODUCTS OFFERED
TABLE 342
AUTOLUS THERAPEUTICS: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 343
AUTOLUS THERAPEUTICS: DEALS, JANUARY 2022−JANUARY 2026
TABLE 344
JW (CAYMAN) THERAPEUTICS CO., LTD.: COMPANY OVERVIEW
TABLE 345
JW (CAYMAN) THERAPEUTICS CO., LTD.: PRODUCTS OFFERED
TABLE 346
JW (CAYMAN) THERAPEUTICS CO., LTD.: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 347
JW (CAYMAN) THERAPEUTICS CO., LTD.: DEALS, JANUARY 2022−JANUARY 2026
TABLE 348
IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): COMPANY OVERVIEW
TABLE 349
IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): PRODUCTS OFFERED
TABLE 350
IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): DEALS, JANUARY 2022−JANUARY 2026
TABLE 351
IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): OTHER DEVELOPMENTS, JANUARY 2022–JANUARY 2026
TABLE 352
CARSGEN THERAPEUTICS HOLDINGS LIMITED: COMPANY OVERVIEW
TABLE 353
CARSGEN THERAPEUTICS HOLDINGS LIMITED: PRODUCTS OFFERED
TABLE 354
CARSGEN THERAPEUTICS HOLDINGS LIMITED: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 355
CARSGEN THERAPEUTICS HOLDINGS LIMITED: DEALS, JANUARY 2022−JANUARY 2026
TABLE 356
CARSGEN THERAPEUTICS HOLDINGS LIMITED: EXPANSIONS, JANUARY 2022−JANUARY 2026
TABLE 357
IASO BIOTHERAPEUTICS: COMPANY OVERVIEW
TABLE 358
IASO BIOTHERAPEUTICS: PRODUCTS OFFERED
TABLE 359
IASO BIOTHERAPEUTICS: PRODUCT APPROVALS, JANUARY 2022−JANUARY 2026
TABLE 360
IASO BIOTHERAPEUTICS: DEALS, JANUARY 2022−JANUARY 2026
TABLE 361
IMMUNEEL THERAPEUTICS: COMPANY OVERVIEW
TABLE 362
IMMUNEEL THERAPEUTICS: PRODUCTS OFFERED
TABLE 363
IMMUNEEL THERAPEUTICS: PRODUCT LAUNCHES, JANUARY 2022−JANUARY 2026
TABLE 364
WUGEN: COMPANY OVERVIEW
TABLE 365
CARTESIAN THERAPEUTICS, INC.: COMPANY OVERVIEW
TABLE 366
ALLOGENE THERAPEUTICS: COMPANY OVERVIEW
TABLE 367
LYELL IMMUNOPHARMA, INC.: COMPANY OVERVIEW
TABLE 368
KYVERNA THERAPEUTICS, INC.: COMPANY OVERVIEW
TABLE 369
CELLICTIS SA: COMPANY OVERVIEW
TABLE 370
BRAINCHILD BIO: COMPANY OVERVIEW
TABLE 371
ATARA BIOTHERAPEUTICS, INC.: COMPANY OVERVIEW
TABLE 372
CARIBOU BIOSCIENCES: COMPANY OVERVIEW
TABLE 373
ARCELLX: COMPANY OVERVIEW
TABLE 374
CRISPR THERAPEUTICS: COMPANY OVERVIEW
TABLE 375
POSEIDA THERAPEUTICS: COMPANY OVERVIEW
TABLE 376
CABALETTA BIO: COMPANY OVERVIEW
TABLE 377
IMPACT ANALYSIS OF SUPPLY- AND DEMAND-SIDE FACTORS ON CAR T-CELL THERAPY MARKET
TABLE 378
CAR T-CELL THERAPY MARKET: RISK ANALYSIS
FIGURE 1
CAR T-CELL THERAPY MARKET SEGMENTATION & REGIONAL SCOPE
FIGURE 2
CAR T-CELL THERAPY MARKET: YEARS CONSIDERED
FIGURE 3
CAR T-CELL THERAPY MARKET SCENARIO
FIGURE 4
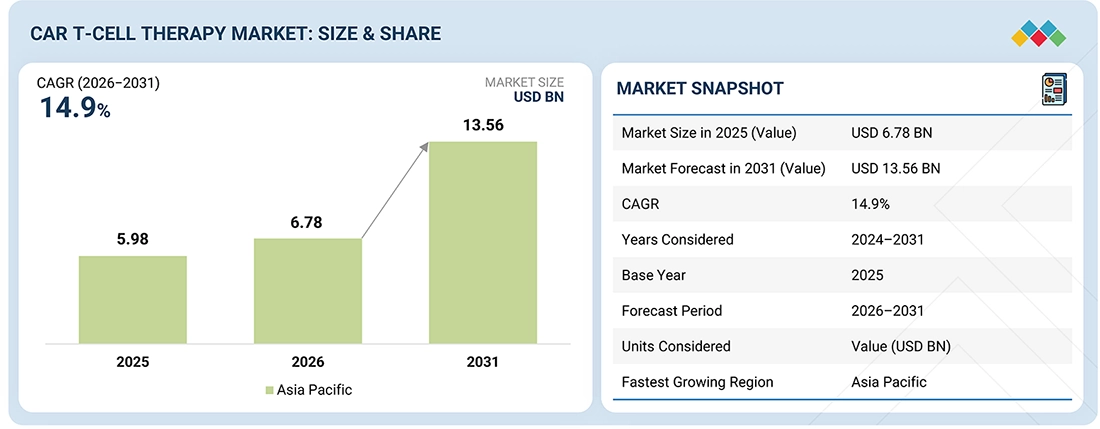
GLOBAL CAR T-CELL THERAPY MARKET SIZE, 2024–2031 (USD MILLION)
FIGURE 5
MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CAR T-CELL THERAPY MARKET, 2023–2025
FIGURE 6
DISRUPTIONS INFLUENCING GROWTH OF CAR T-CELL THERAPY MARKET
FIGURE 7
HIGH-GROWTH SEGMENTS IN CAR T-CELL THERAPY MARKET, BY PRODUCT, TARGET, END USER, AND REGION, 2026–2031
FIGURE 8
ASIA PACIFIC TO REGISTER HIGHEST CAGR IN CAR T-CELL THERAPY MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD
FIGURE 9
TECHNOLOGICAL ADVANCEMENTS IN CAR T-CELL THERAPIES TO DRIVE GROWTH
FIGURE 10
US AND ADULTS DOMINATED NORTH AMERICAN CAR T-CELL THERAPY MARKET IN 2025
FIGURE 11
INDIA TO REGISTER FASTEST GROWTH RATE DURING FORECAST PERIOD
FIGURE 12
CAR T-CELL THERAPY MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
FIGURE 13
GLOBAL CAR T-CELL THERAPY TRIALS DISTRIBUTION, BY TARGET (ACROSS ALL PHASES OF DEVELOPMENT), 2025
FIGURE 14
GLOBAL CAR T-CELL THERAPY TRIALS DISTRIBUTION OF TARGET BY DEVELOPMENT PHASE, 2025
FIGURE 15
CAR T-CELL THERAPY MARKET: PORTER’S FIVE FORCES ANALYSIS
FIGURE 16
CAR T-CELL THERAPY MARKET: VALUE CHAIN ANALYSIS
FIGURE 17
CAR T-CELL THERAPY MARKET: ECOSYSTEM ANALYSIS
FIGURE 18
AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY KEY PLAYER, 2025 (USD)
FIGURE 19
TRENDS/DISRUPTIONS IMPACTING CUSTOMER’S BUSINESS IN CAR T-CELL THERAPY MARKET
FIGURE 20
FUNDING AND NUMBER OF DEALS IN CAR T-CELL THERAPY MARKET (USD MILLION)
FIGURE 21
IMPACT OF AI/GEN AI ON CAR T-CELL THERAPY MARKET
FIGURE 22
INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS, BY END USER
FIGURE 23
KEY BUYING CRITERIA FOR MAJOR END USERS
FIGURE 24
NORTH AMERICA: CAR T-CELL THERAPY MARKET SNAPSHOT
FIGURE 25
ASIA PACIFIC: CAR T-CELL THERAPY MARKET SNAPSHOT
FIGURE 26
REVENUE ANALYSIS OF KEY PLAYERS IN CAR T-CELL THERAPY MARKET, 2023–2025 (USD MILLION)
FIGURE 27
MARKET SHARE ANALYSIS OF KEY PLAYERS IN CAR T-CELL THERAPY MARKET (2025)
FIGURE 28
CAR T-CELL THERAPY MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
FIGURE 29
CAR T-CELL MARKET: COMPANY FOOTPRINT
FIGURE 30
CAR T-CELL THERAPY MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
FIGURE 31
EV/EBITDA OF KEY VENDORS
FIGURE 32
YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
FIGURE 33
CAR T-CELL THERAPY MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS
FIGURE 34
JOHNSON & JOHNSON: COMPANY SNAPSHOT
FIGURE 35
GILEAD SCIENCES, INC.: COMPANY SNAPSHOT
FIGURE 36
BRISTOL-MYERS SQUIBB COMPANY: COMPANY SNAPSHOT
FIGURE 37
NOVARTIS AG: COMPANY SNAPSHOT
FIGURE 38
JW (CAYMAN) THERAPEUTICS CO., LTD.: COMPANY SNAPSHOT
FIGURE 39
CARSGEN THERAPEUTICS HOLDINGS LIMITED: COMPANY SNAPSHOT
FIGURE 40
CAR T-CELL THERAPY MARKET: RESEARCH DESIGN
FIGURE 41
CAR T-CELL THERAPY MARKET: BREAKDOWN OF DEMAND– AND SUPPLY-SIDE PRIMARIES
FIGURE 42
CAR T-CELL THERAPY MARKET SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2025
FIGURE 43
CAR T-CELL THERAPY MARKET SIZE ESTIMATION: COMPANY REVENUE ANALYSIS-BASED ESTIMATION (PRODUCTS), 2025
FIGURE 44
ILLUSTRATIVE EXAMPLE OF GILEAD SCIENCES, INC.: REVENUE SHARE ANALYSIS (2025)
FIGURE 45
CAR T-CELL THERAPY MARKET VALIDATION FROM PRIMARY SOURCES
FIGURE 46
CAR T-CELL THERAPY MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
FIGURE 47
CAR T-CELL THERAPY MARKET: CAGR PROJECTIONS, 2026–2031
FIGURE 48
CAR T-CELL THERAPY MARKET: DATA TRIANGULATION METHODOLOGY






.svg)




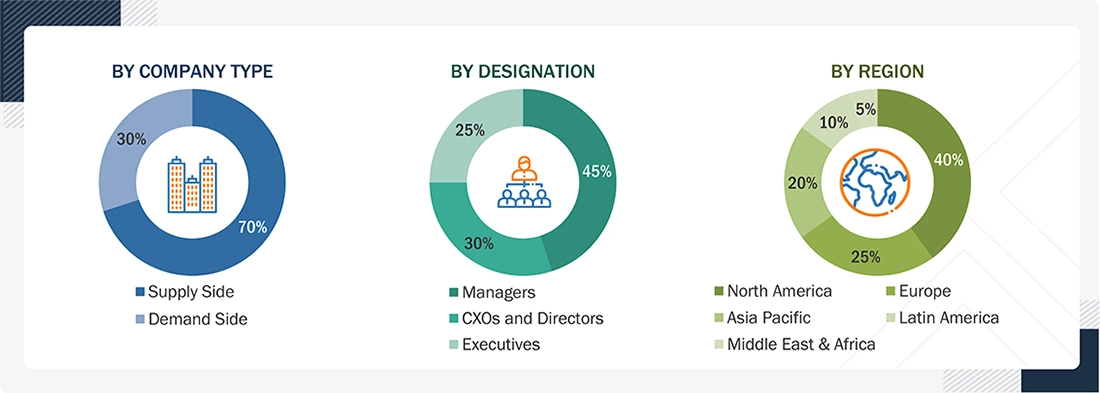



















Growth opportunities and latent adjacency in CAR T-Cell Therapy Market