
Latin America Point of Care Diagnostics Market Size, Growth, Share & Trends Analysis
Latin America Point of Care Diagnostics Market by Product (Glucose, Infectious Disease [TB, HAI, STD], Pregnancy), Purchase Mode (Rx, OTC), Technology (Biochemistry, MDx [RT-PCR, INAAT]), Sample (Blood, Urine), End User (Home Care, Hospitals) - Forecast to 2031




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
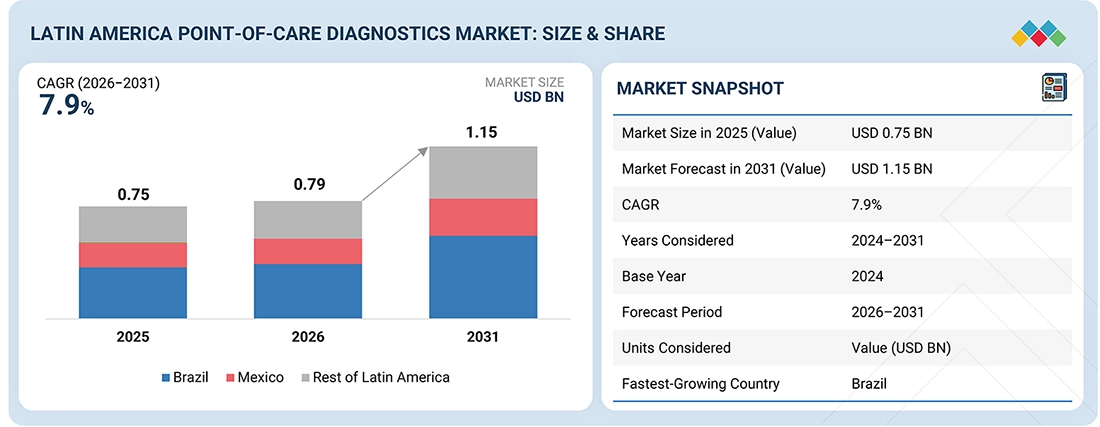
The Latin America point-of-care diagnostics market, valued at US$0.75 billion in 2025, stood at US$0.79 billion in 2026 and is projected to advance at a resilient CAGR of 7.9% from 2026 to 2031, culminating in a forecasted valuation of US$1.15 billion by the end of the period. This growth is driven by the increasing demand for diagnostic solutions to address the rising burden of chronic and infectious diseases. Key factors contributing to this expansion include improvements in healthcare infrastructure, increased public and private investments in decentralized testing, and greater access to near-patient diagnostic technologies. Additionally, a heightened focus on early disease detection, the wider adoption of self-testing and home-based diagnostics, and the growing availability of cost-effective point-of-care solutions are further fueling the adoption of these diagnostics in hospitals, clinics, community healthcare centers, and remote areas. As a result, the market is expected to grow from USD 0.79 billion in 2026 to USD 1.15 billion by 2031, at a CAGR of 7.9% during the forecast period.
KEY TAKEAWAYS
-
By ProductBy product, the infectious disease diagnostics segment is expected to register the highest CAGR of 9.5% during the forecast period.
-
By Mode of PurchaseThe prescription-based segment is expected to grow at the highest CAGR of 9.1% during the forecast period.
-
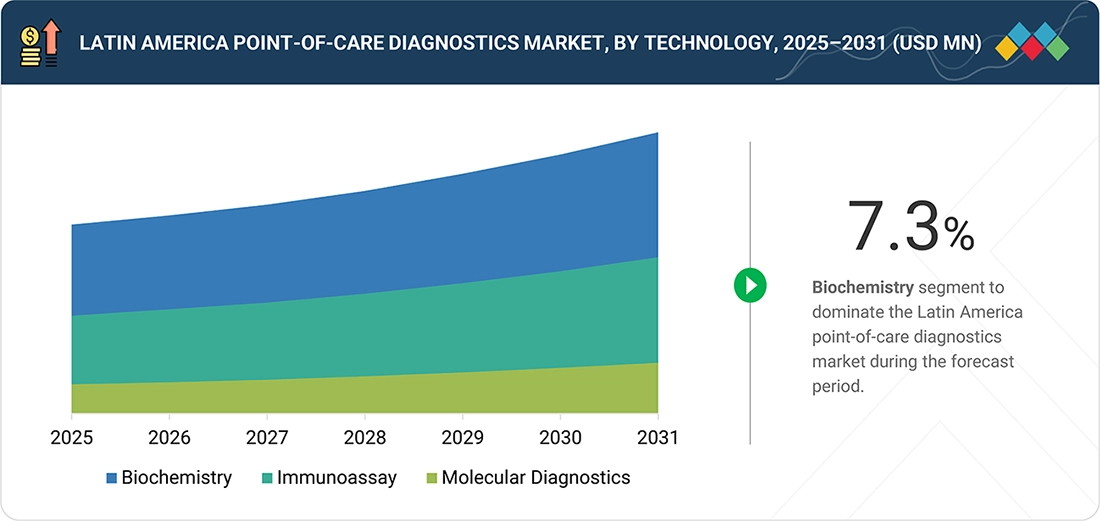
By TechnologyBy technology, the biochemistry segment held the largest share of 59.7% in 2025 due to extensive use in point-of-care devices.
-
By Sample TypeThe blood samples segment accounts for the largest market share in 2025.
-
By End userBy end user, home care settings and self-testing are expected to be the fastest-growing segment. This can be attributed to the increasing adoption of decentralized and patient-centric solutions.
-
Competitive LandscapeAbbott (US), F. Hoffmann La-Roche Ltd. (Switzerland), Siemens (Germany), QuidelOrtho (US), and Danaher Corporation (US) emerged as potential key players in the Latin America point-of-care diagnostics market.
-
Competitive LandscapeEmerging companies like ModiTech Med (Korea) and Response Biotech (Canada), with strong presence in niche markets, have already made a name for themselves among new and small-to-medium-sized enterprises.
The Latin America point-of-care diagnostics market is experiencing significant growth, as the healthcare industry increasingly adopts point-of-care testing to enhance diagnostic efficiency. Healthcare providers face mounting pressure to make informed clinical decisions regarding infections and chronic diseases, driving the demand for point-of-care diagnostics in the region. This market includes hospitals, clinics, and healthcare organizations. Changes in regulatory alignment, along with advancements that improve test reliability, usability, and integration, are making point-of-care diagnostics more feasible. Additionally, effective cost management in the healthcare system, the decongestion of hospitals, and the ability to reach underserved areas are all contributing to the expansion of the point-of-care diagnostics market in Latin America.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The Latin America point-of-care diagnostics market is experiencing steady growth as various health infrastructures in the region focus on improving access and accelerating clinical decision-making. Point-of-care tests are being embraced and integrated into various health facilities, including hospitals, primary care centers, community health facilities, pharmacies, and home care settings. This integration aims to address access gaps and reduce diagnostic turnaround times. Additionally, the increasing adoption of decentralized testing approaches, along with the introduction of new and rapid point-of-care diagnostic solutions, is transforming and enhancing diagnostic practices in the region. The advancement of technology, particularly the development of small, portable diagnostic tools, is further improving point-of-care diagnostics across Latin America.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Growing incidence of infectious diseases

-
Favorable government initiatives for point-of-care testing
Level
-
Pricing pressure on manufacturers
-
Stringent regulatory approval process for product commercialization
Level
-
Increasing growth potential in emerging markets
-
Rising inclination toward decentralized healthcare system
Level
-
Inadequate standardization with centralized lab methods
-
Premium pricing of novel platforms
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Growing incidence of infectious diseases
One of the most important factors driving this market's growth is the rise in infectious diseases, which is also a key issue for the Latin America market. The increasing cases of infectious diseases, such as respiratory tract infections, sexually transmitted diseases, and other viral and bacterial infections, has propelled the demand for point-of-care diagnostics. This technology enables timely decision-making without relying on laboratory facilities that may be limited in certain areas. Consequently, point-of-care diagnostics play a significant role in the detection and control of these diseases in Latin America.
Restraint: Pricing pressure on manufacturers
Cost constraints and reimbursement challenges present significant obstacles for the point-of-care diagnostics market in Latin America. This situation arises from limited healthcare budgets, which are further strained by a rigid reimbursement environment and strict cost limitations within these countries. Additionally, there are affordability issues with the prices charged for diagnostic services in public healthcare facilities. These factors hinder the profitability of point-of-care diagnostic products in the region, as it is difficult to offer premium prices for these services.
Opportunity: Increasing growth potential in emerging markets
The Latin America point-of-care diagnostics market has significant growth potential, driven by increased access to healthcare services, rising incidence of infectious and chronic diseases, and heightened awareness of the importance of early disease detection. Many countries in the region have limited centralized laboratory infrastructure, making point-of-care testing an efficient solution for disease detection. Additionally, growing investments in healthcare, the development of primary care facilities, and the availability of portable, affordable, and easy-to-use devices present considerable opportunities for market expansion. As there is a rising emphasis on preventing and treating conditions cost-effectively, this environment offers an ideal platform for companies to enter and operate in emerging and underserved markets across Latin America.
Challenge: Inadequate standardization with centralized lab methods
The lack of standardization in the Latin America point-of-care diagnostics market, especially when compared to centralized laboratory-based tests, poses a significant challenge. Issues arise from differences in test protocols, inconsistent execution of these protocols, and varying levels of personnel training, all of which can lead to unreliable test results. In Latin America, healthcare personnel conduct these tests in decentralized settings that differ greatly from the highly controlled environments of laboratory-based protocols. As a result, there are valid concerns about the reliability and quality of the results obtained from point-of-care diagnostics.
LATIN AMERICA POINT OF CARE DIAGNOSTICS MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
The deployment of quick point-of-care products like i-STAT and ID NOW in infectious disease testing, cardiac markers, and metabolism in hospitals, emergency rooms, primary care sites, retail clinics, and community healthcare settings in Latin America countries | It supports rapid clinical decision-making, aids in reducing turnaround time, promotes decentralized diagnostics, and enhances patient throughput in resource-constrained care settings |
 |
The application of Waived CLIA and near-patient immunoassays/blood gas analysis equipment that is utilized in emergency departments, intensive care units, and outpatient sites within the Latin America region | Expands access to rapid diagnostic testing, enhances the delivery of care in both acute and outpatient settings, and decreases dependency on centralized laboratory infrastructure |
 |
Adoption of cobas Liat point-of-care molecular systems for the diagnosis of infectious diseases, such as respiratory pathogens and hospital-acquired infections, in Latin America countries | Enables early disease detection, offers improved infection control activities, and assures the reliable delivery of molecular diagnostic information for treatment decisions |
 |
Application of quick antigen, immunoassay, and molecular point-of-care tests in the diagnosis of respiratory pathogens, women's health, and cardiac markers in physicians' offices, retail clinics, and decentralized healthcare settings in the Latin America region | Increases patient access to diagnostics, enhances decentralized testing strategies, and aids quicker diagnostic decisions in primary and ambulatory patient settings |
 |
Distribution of point-of-care diagnostic instrumentation and infectious disease consumables for acute care testing via subsidiary operations throughout Latin America | Ensures the scalable deployment of point-of-care solutions, test performance, and the achievement of efficient diagnostics in hospitals, emergency departments, and urgent care centers |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The Latin America point-of-care diagnostics market includes key players throughout the entire value chain, from the manufacturing of point-of-care devices and test kits to regional distributors and retail and pharmacy channels that enhance product availability and access. Research organizations and local partners also play a crucial role in product adaptation and technology transfer. Moreover, end users are increasingly relying on point-of-care solutions that offer rapid, decentralized testing. This capability enables faster clinical decision-making and improves access to diagnostics in both urban and underserved areas across Latin America.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Latin America Point-of-care Diagnostics Market, by Product
The Latin America point-of-care diagnostics market comprises a range of products, including glucose monitoring, cardiometabolic testing, infectious disease testing, coagulation testing, pregnancy and fertility testing, cancer marker testing, urinalysis, cholesterol testing, hematology, drug abuse testing, thyroid-stimulating hormone testing, and fecal occult blood testing. During the forecast period, the glucose monitoring segment is expected to contribute significantly to overall market growth. This is primarily due to the rising prevalence of diabetes, improved access to testing devices, and an increasing number of individuals opting for self-testing and home-based care solutions. Additionally, heightened awareness of disease management and gradual advances in portable glucose testing devices are key factors driving the growth of the glucose monitoring market in Latin America.
Latin America Point-of-care Diagnostics Market, by Mode of Purchase
The Latin America point-of-care diagnostics market is categorized based on the mode of purchase into two segments: over-the-counter testing products and prescription-based testing products. Prescription-based testing products hold a larger market share primarily due to regulations established by health organizations and ministries in the region. These regulations ensure the quality, accuracy, and reliability of testing kits and products. Prescription-based testing products are predominantly used in hospitals, medical offices, and laboratories in Latin America, as these environments require greater supervision. The growing confidence among physicians in point-of-care (POC) testing kits, particularly those approved by health organizations, is driving increased demand for prescription-based testing products in the Latin America market.
Latin America Point-of-care Diagnostics Market, by Technology
The Latin America point-of-care diagnostics market can be classified into three main technology categories: biochemistry, immunoassay technologies, and molecular diagnostics. By 2025, biochemistry-based technologies are projected to hold the largest market share in this sector. This trend is largely due to the widespread use of biochemistry-based point-of-care tests, such as glucose meters and lipid analyzers, for diagnosing chronic conditions like cardiovascular diseases, diabetes, and renal diseases, among others. The popularity of biochemistry-based point-of-care tests can be attributed to their affordability, ease of implementation, and accuracy, which have significantly contributed to the growth of this market segment in the region.
Latin America Point-of-care Diagnostics Market, by Sample
Based on sample type, the market is segmented into blood sample tests, urine sample tests, nasal/oropharyngeal swab tests, and other sample tests. In 2025, the blood sample tests segment held the largest share of the Latin America point-of-care diagnostics market. Blood samples are widely used in various point-of-care applications and are critical for monitoring chronic conditions such as heart diseases and metabolic disorders. The prevalence of these diseases is increasing in Latin America. The minimally invasive collection of blood samples, using techniques like fingerstick sampling, along with the availability of reliable blood sample point-of-care tests, has made blood sample tests a very popular choice in hospitals and home care settings across the region.
Latin America Point-of-care Diagnostics Market, by End user
The Latin American point-of-care diagnostics market can be categorized based on end users into clinical laboratories, hospitals, ambulatory care facilities, physician offices, critical and urgent care centers, home care settings, and self-testing, as well as other end users. By 2025, home care settings and self-testing are expected to dominate the Latin America point-of-care diagnostics market. This trend is largely due to consumers preferring quick, private testing options that eliminate the need to visit healthcare facilities. With the rise in chronic conditions, such as diabetes and cardiovascular diseases, the demand for point-of-care diagnostic solutions in the region has increased, especially among people living in urban and semi-urban areas.
REGION
Brazil is the fastest-growing country in region
Brazil is expected to lead the Latin American point-of-care diagnostics market, with significant growth anticipated throughout the forecast period. This growth is driven by the increasing adoption of point-of-care testing technologies and by heightened investment in healthcare facilities across the region. Additionally, Brazil is experiencing a growing patient population, an expanding primary healthcare sector, and a rising burden of infectious and chronic diseases. These factors are significantly contributing to the growth of the Latin America point-of-care diagnostics market, making Brazil the most important market in this sector.

LATIN AMERICA POINT OF CARE DIAGNOSTICS MARKET: COMPANY EVALUATION MATRIX
Abbott remains a leading player in the Latin America point-of-care diagnostics market, due to its extensive portfolio of widely adopted POC platforms and ongoing innovation in rapid testing technologies. The company offers a comprehensive menu of tests and scalable systems, supported by a large installed base that drives consistent demand for consumables across the region. Additionally, Siemens Healthineers leverages its strong presence in the Latin America POC testing market by emphasizing high clinical performance, user-friendly system design, and compliance with regional regulatory standards. This strategy helps the company maintain a competitive edge across various point-of-care diagnostics segments.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Abbott (US)
- Siemens (Germany)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- QuidelOrtho (US)
- Danaher Corporation (US)
- Becton, Dickinson and Company (US)
- Thermo Fisher Scientific Inc. (US)
- bioMérieux (France)
- EKF Diagnostics Holdings plc (UK)
- Werfen (Spain)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 0.75 Billion |
| Market Forecast in 2031 (Value) | USD 1.15 Billion |
| Growth Rate | CAGR of 7.9% from 2026–2031 |
| Years Considered | 2024–2031 |
| Base Year | 2024 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Million) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Countries Covered | Brazil, Mexico, and the Rest of Latin America |
| Parent & Related Segment Reports |
Point of Care Diagnostics Market United States Point of Care Diagnostics Market Europe Point of Care Diagnostics Market |
WHAT IS IN IT FOR YOU: LATIN AMERICA POINT OF CARE DIAGNOSTICS MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Product Analysis | Product matrix, which provides a detailed comparison of the product portfolio of each company in the Latin America point-of-care diagnostics market | Enables easy comparison of competitors’ offerings, helping identify gaps, overlaps, and differentiation opportunities |
| Company Information | Additional company profiles of players (five) operating in the Latin America point-of-care diagnostics market | Provides insights into competitors’ strategies, innovation focus, and partnerships, supporting strategic planning |
RECENT DEVELOPMENTS
- April 2024 : The i-STAT TBI cartridge (Point-of-Care Diagnostics cartridge) received clearance from the US Food and Drug Administration (FDA) for use with whole blood. This approval supports broader global commercialization efforts, including expansion opportunities in Latin America, enabling clinicians to assess patients with suspected concussion at the bedside and obtain laboratory-quality results within 15 minutes.
- March 2023 : Boditech Med Inc. (South Korea) advanced to the second phase of its partnership with Raia Drogasil (RD Group) to distribute its pharmacy-based diagnostic system in Brazil. The company planned to more than double the installation of its AFIAS platform and gradually expand deployment across RD Group-owned pharmacies, enhancing point-of-care diagnostic services in Latin America.
- January 2022 : F. Hoffmann-La Roche Ltd (Switzerland) launched its cobas infinity edge, a secure and encrypted cloud-based platform designed to integrate and manage point-of-care diagnostic data. The software is intended for global use in clinical settings to streamline connectivity and data management.
Table of Contents

Methodology
The study involved major activities in estimating the current market size for the Latin America Point of Care Diagnostics Market. Exhaustive secondary research was done to collect information on the point of care diagnostics industry. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain using primary research. Different approaches, such as top-down and bottom-up, were employed to estimate the total market size. After that, the market breakup and data triangulation procedures were used to estimate the market size of the segments and subsegments of the Latin America Point of Care Diagnostics Market.
The four steps involved in estimating the market size are
Secondary Research
In the secondary research process, various secondary sources such as annual reports, press releases & investor presentations of companies, white papers, certified publications, articles by recognized authors, gold-standard & silver-standard websites, regulatory bodies, and databases (such as D&B Hoovers, Bloomberg Business, and Factiva) were referred to identify and collect information for this study.
Primary Research
In the primary research process, various sources from both the supply and demand sides were interviewed to obtain qualitative and quantitative information for this report. Primary sources were mainly industry experts from the core and related industries and preferred suppliers, manufacturers, distributors, service providers, technology developers, researchers, and organizations related to all segments of this industry’s value chain. In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts, C-level executives of key market players, and industry consultants, to obtain and verify critical qualitative and quantitative information as well as assess prospects.
To know about the assumptions considered for the study, download the pdf brochure
| COMPANY NAME | DESIGNATION |
|---|---|
| Abbott | Regional Sales Manager |
| F. Hoffmann-La Roche Ltd | Product Manager |
Market Size Estimation
Both top-down and bottom-up approaches were used to estimate and validate the Latin America Point of Care Diagnostics Market's total size. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
- The key players in the industry have been identified through extensive secondary research
- The revenues generated by leading players operating in the Latin America Point of Care Diagnostics Market have been determined through primary and secondary research.
- All percentage shares, splits, and breakdowns have been determined using secondary sources and verified through primary sources.
Data Triangulation
After arriving at the overall market size by applying the process mentioned above, the total market was split into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics for all segments and subsegments, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Point of care testing (POCT) includes screening or diagnostic tests that are designed to be used at or near the patient's site (such as at home, in an ambulance, at a physician's office, and other locations). POCT allows rapid and reliable diagnostic testing to obtain outcomes instantly, helping physicians/patients make care decisions remotely and as soon as possible.
Stakeholders
- Senior Management
- End User
- Finance/Procurement Department
- R&D Department
Report Objectives
- To define, describe, segment, and forecast the Latin America Point of Care Diagnostics Market, by product, mode of purchase, technology, sample, end user, and region
- To provide detailed information regarding the major factors influencing the market growth (such as drivers, restraints, opportunities, and challenges)
- To analyze the micromarkets1 with respect to individual growth trends, prospects, and contributions to the overall Latin America Point of Care Diagnostics Market
- To analyze market opportunities for stakeholders and provide details of the competitive landscape for key players
- To forecast the size of the market segments with respect to six regions, namely, North America, Europe, the Asia Pacific, Latin America, Middle East & Africa, and GCC countries
- To profile the key players and comprehensively analyze their product portfolios, market positions, and core competencies
- To track and analyze company developments such as product launches & approvals, partnerships, acquisitions, agreements, and other developments
- To benchmark players within the market using the proprietary Competitive Leadership Mapping framework, which analyzes market players on various parameters within the broad categories of business and product excellence
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Latin America Point of Care Diagnostics Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS





















Growth opportunities and latent adjacency in Latin America Point of Care Diagnostics Market