
Molecular Infectious Disease Testing Market
Molecular Infectious Disease Testing Market by Product & Service (Kits, Instruments), Type (Singleplex, Multiplex), Test Type (Lab, PoC), Disease (STD, Flu, GI, HAI), Technology (PCR, NGS), End User (Diagnostic Labs, Hospitals) - Global Forecast to 2031




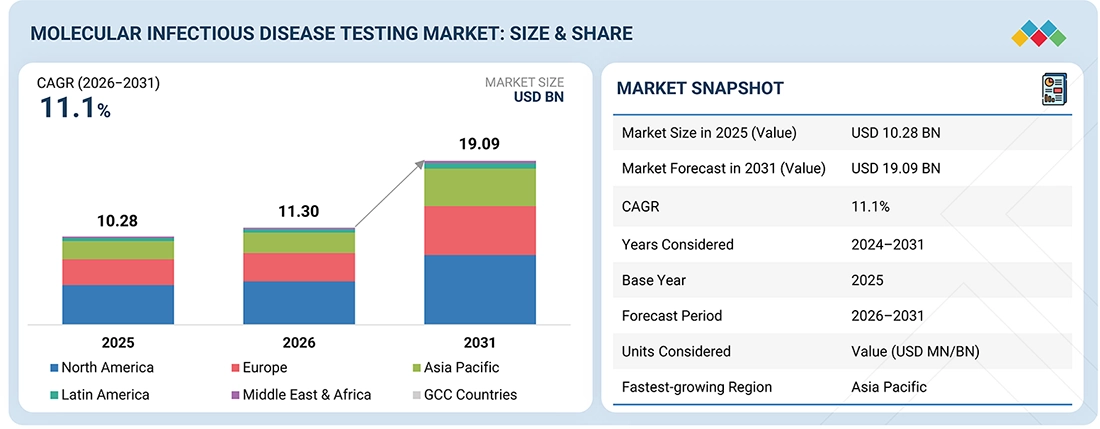
MOLECULAR INFECTIOUS DISEASE TESTING MARKET SIZE, SHARE & GROWTH SNAPSHOT
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
The global molecular infectious disease testing market is projected to grow from USD 11.30 billion in 2026 to USD 19.09 billion by 2031, at a CAGR of 11.1% during the forecast period. The market was valued at USD 10.28 billion in 2025. This growth is primarily driven by the increasing incidence of infectious diseases such as HIV, HPV, hepatitis, and chlamydia/gonorrhea, as well as the emergence of new pathogens that require timely and accurate diagnosis. Additionally, rising investments in research and development (R&D) have led to significant advancements in molecular diagnostics, including PCR (polymerase chain reaction) and NGS (next-generation sequencing). Other important factors contributing to this growth include both public and private financing, as well as government policies aimed at enhancing diagnostic capabilities.
KEY TAKEAWAYS
-
BY REGIONBy region, North America held the largest market share in 2025.
-
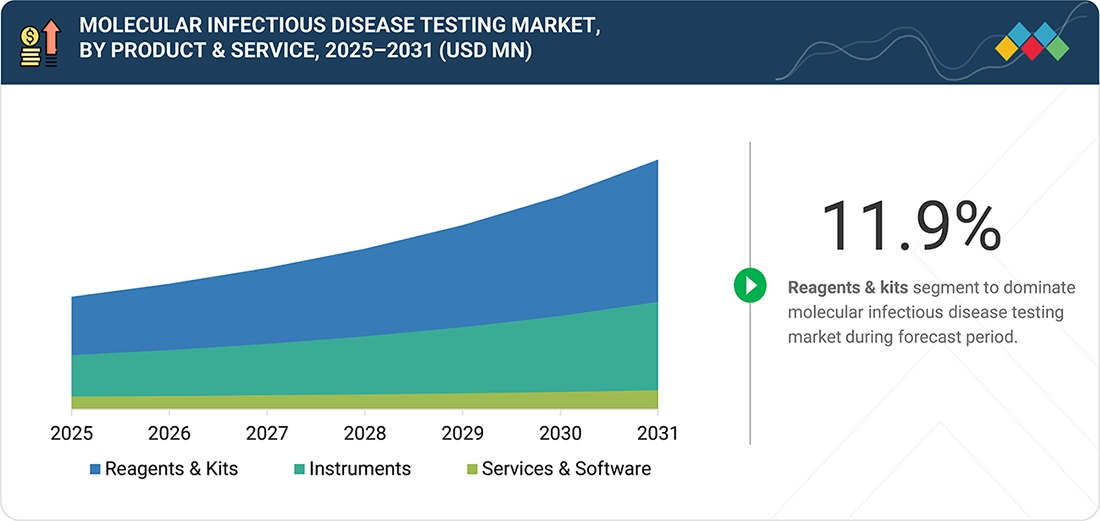
BY PRODUCT & SERVICEBy product & service, the reagents & kits segment is expected to register the highest growth rate of 11.9% during the forecast period.
-
BY TESTBy test, the singleplex testing segment accounted for the largest market share in 2025.
-
BY TEST TYPEBy test type, the laboratory tests segment is expected to register the highest CAGR during the forecast period.
-
BY DISEASEBy disease, the sexually transmitted diseases segment accounted for the largest share of 41.0% in 2025.
-
BY TECHNOLOGYBy technology, the polymerase chain reaction segment held the largest market share of 44.1% in 2025.
-
BY END USERBy end user, the diagnostic laboratories segment is expected to grow at the highest rate of 12.1% during the forecast period.
-
COMPETITIVE LANDSCAPE - KEY PLAYERSDanaher (US), F. Hoffmann-La Roche Ltd. (Switzerland), bioMérieux (France), Hologic, Inc. (US), and Abbott (US) were identified as star players because of their strong market presence and extensive product portfolios.
-
COMPETITIVE LANDSCAPE - STARTUPSVela Diagnostics (Singapore), Molbio Diagnostics Pvt. Ltd. (India), Uniogen OY (Finland), geneOmbio Technologies (India), and Advanced Molecular Diagnostics (UK) have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas.
The market for molecular infectious disease testing is expected to grow rapidly due to the increasing prevalence of infectious diseases worldwide, such as HIV, hepatitis, HPV, and respiratory infections. Molecular tests, including PCR and next-generation sequencing (NGS) techniques, have seen significant advancements, contributing to greater efficiency and accuracy in pathogen diagnosis. Additionally, funding from the healthcare sector and steady investments have improved research and development, leading to faster market expansion.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
Due to the high incidence of infectious diseases and advances in molecular diagnostics, there is a growing demand for molecular testing in this area. Molecular diagnostic tests are vital for identifying pathogens, controlling epidemics, and monitoring infectious diseases. The increased use of PCR, multiplex methods, and sequencing, along with funding from both public and private sectors, is expected to lead to a rise in the number of molecular diagnostic tests available. By investing in quality reagents, advanced laboratory equipment, and qualified personnel, market players can expect to achieve improved results in this field.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Growing global infectious disease burden

-
Advancements in molecular diagnostic technologies
Level
-
Lack of standardized reimbursement structures
-
High cost of instruments and consumables
Level
-
Expansion potential across emerging economies
-
Rising demand for point-of-care (POC) testing
Level
-
Stringent and varying regulatory requirements
-
Operational barriers and labor shortage challenges
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Growing funding for molecular testing
Significant investments in molecular testing are a key factor driving the market for molecular infectious disease testing. Funding from governments, public health organizations, and the private sector has improved testing technologies and increased the availability of molecular tests. Notable advancements are occurring in technologies such as real-time PCR and next-generation sequencing. Additionally, government initiatives aimed at controlling infectious diseases have led to greater investments in molecular testing technologies, ensuring their integration into the healthcare framework.
Restraint: Lack of standardized reimbursement structures
Differences in reimbursement processes are considered significant obstacles for the molecular diagnostics market, particularly in the area of infectious diseases. Varying reimbursement standards across different locations can create challenges for both providers and patients. This variability may hinder the widespread adoption of molecular testing, limit access to essential diagnostic tests, and raise costs for healthcare facilities. Additionally, the lack of standardized reimbursement practices could discourage laboratories from implementing these molecular testing tools.
Opportunity: Expansion potential across emerging economies
There is significant potential in emerging markets due to growth opportunities in the molecular infectious disease testing industry. The development of the healthcare sector, an increase in disposable income, and greater awareness of infectious diseases are driving demand for advanced molecular testing. Additionally, government policies that support the use of molecular diagnostic tools and foreign investments in healthcare are facilitating the adoption of these technologies by businesses.
Challenge: Operational barriers and labor shortage challenges
The implementation of molecular infectious disease diagnostics is expected to encounter significant challenges due to staffing and logistical issues. A shortage of well-trained personnel may hinder the adoption of new molecular diagnostic techniques, particularly in less developed countries. Additionally, logistical challenges, including insufficient infrastructure, regulatory concerns, and disruptions in the supply chain, could complicate the integration of these innovative technologies.
MOLECULAR INFECTIOUS DISEASE TESTING MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Provides molecular diagnostic platforms and assay kits for infectious disease detection in clinical laboratories. | High accuracy and reproducibility, supports multiplex testing, enables rapid pathogen identification, and improves workflow efficiency. |
 |
Offers molecular testing solutions, primarily PCR-based, for detecting infectious diseases in clinical labs. | Fast turnaround, sensitive and specific results, supports early detection, and integrates with laboratory workflows. |
 |
Supplies molecular infectious disease diagnostic kits and instruments for hospitals and reference labs. | Reliable and robust testing, supports high-throughput diagnostics, enhances pathogen detection capabilities, and improves patient outcomes. |
 |
Provides molecular assays and diagnostic platforms for sexually transmitted and other infectious diseases. | Accurate and sensitive testing, scalable for large populations, enables timely intervention, and facilitates automated lab workflows. |
 |
Offers molecular infectious disease testing solutions for clinical laboratories and hospital settings. | Rapid, precise results, user-friendly platforms, supports broad infectious disease panels, and enhances operational efficiency. |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
In the molecular infectious disease testing industry, manufacturers, laboratories, hospitals, and healthcare providers work together to ensure accurate diagnosis of infections. Manufacturers design and supply molecular tests and systems, such as PCR tests and multiplex kits, which laboratories and clinics use for efficient testing. Laboratory personnel, physicians, and healthcare institutions depend on the innovations provided by manufacturers for diagnosing infectious diseases. The industry is experiencing growth due to regulatory support, technological advancements, and collaborative partnerships within the sector.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Molecular Infectious Disease Testing Market, by Product & Service
The molecular infectious disease testing market can be divided into several categories based on products & services, including reagents & kits, instruments, and services & software. Reagents & kits are essential components of molecular diagnostics because they facilitate the precise and efficient detection of infectious agents using primers, probes, enzymes, and testing kits. The demand for these items remains steady due to the increasing number of infectious disease cases and the need for repeated testing in diagnostic procedures. Additionally, the ongoing advancements in molecular technologies and the expansion of testing programs across various healthcare settings further underscore the importance of high-quality reagents and kits for obtaining reliable results.
Molecular Infectious Disease Testing Market, by Type
Molecular testing for infectious diseases can be categorized into two types: singleplex testing and multiplex testing. Multiplex testing has gained significant popularity because it allows for the simultaneous identification and differentiation of multiple pathogens, which saves both time and effort. The rising prevalence of co-infections and the demand for rapid, accurate, and efficient testing have further contributed to the increased use of multiplex testing. Additionally, there have been innovations in assay design and the development of user-friendly systems.
Molecular Infectious Disease Testing Market, by Test Type
The market for molecular infectious disease testing can be divided into two main categories: laboratory testing and point-of-care testing. Laboratory testing is widely accepted due to its high precision, scalability, and ability to diagnose a wide range of pathogens. Techniques such as PCR and sequencing-based diagnostics are commonly used in hospitals and diagnostic laboratories because of their accuracy and reliability. With advancements in laboratory automation and high-throughput methods, the turnaround time for test results has significantly decreased, making these approaches not only efficient but also suitable for diagnosing more complex cases.
Molecular Infectious Disease Testing Market, by Disease
Testing for infectious diseases through molecular methods is categorized based on the type of infections, which include respiratory infections, gastrointestinal infections, sexually transmitted diseases (STDs), vaginitis, meningitis, vector-borne infections, hospital-acquired infections, and other infectious diseases. The demand for STD tests is rising due to the increasing prevalence of these diseases and growing awareness about the importance of early diagnosis and treatment. Advanced molecular techniques, such as polymerase chain reaction (PCR) and nucleic acid amplification tests, provide rapid and sensitive results that are essential for effective disease management. Additionally, the expansion of public health programs aimed at the prevention and treatment of these diseases is contributing to the growth of molecular STD testing.
Molecular Infectious Disease Testing Market, by Technology
The market for molecular infectious disease testing is categorized by technology. This includes several methods such as polymerase chain reaction (PCR), isothermal nucleic acid amplification, DNA sequencing, next-generation sequencing (NGS), in situ hybridization, DNA microarrays, and other advanced tests. PCR technology is one of the most commonly used methods due to its high reliability and accuracy in detecting a wide range of pathogens. Its popularity can be attributed to the availability of standardized protocols, ready-to-use test kits, and increased automation, which enhance the speed and efficiency of the testing process. Moreover, technological advancements have further solidified PCR's role as a key technology in delivering precise molecular diagnostics.
Molecular Infectious Disease Testing Market, by End User
The molecular infectious disease diagnostics market includes various end users such as diagnostic laboratories, hospitals & clinics, and other end users. Diagnostic laboratories play a crucial role in this market due to the rising demand for rapid and accurate testing methods. As the need for precise pathogen detection grows, these laboratories are increasingly adopting advanced testing techniques, including PCR (polymerase chain reaction) and multiplexing technology. Furthermore, with a heightened focus on disease prevention, there is a growing necessity for faster test results to aid in the prevention and management of diseases.
REGION
North America accounted for largest share of global molecular infectious disease testing market during forecast period.
In North America, several factors have contributed to the growth of the molecular infectious disease diagnostics market. These factors include the presence of advanced healthcare facilities, the adoption of modern diagnostic techniques, and substantial investments in research and development. Additionally, government interventions, health initiatives, and the presence of key industry players play significant roles. The high prevalence of infectious diseases and the increasing demand for accurate testing are also important contributors to this market's expansion.
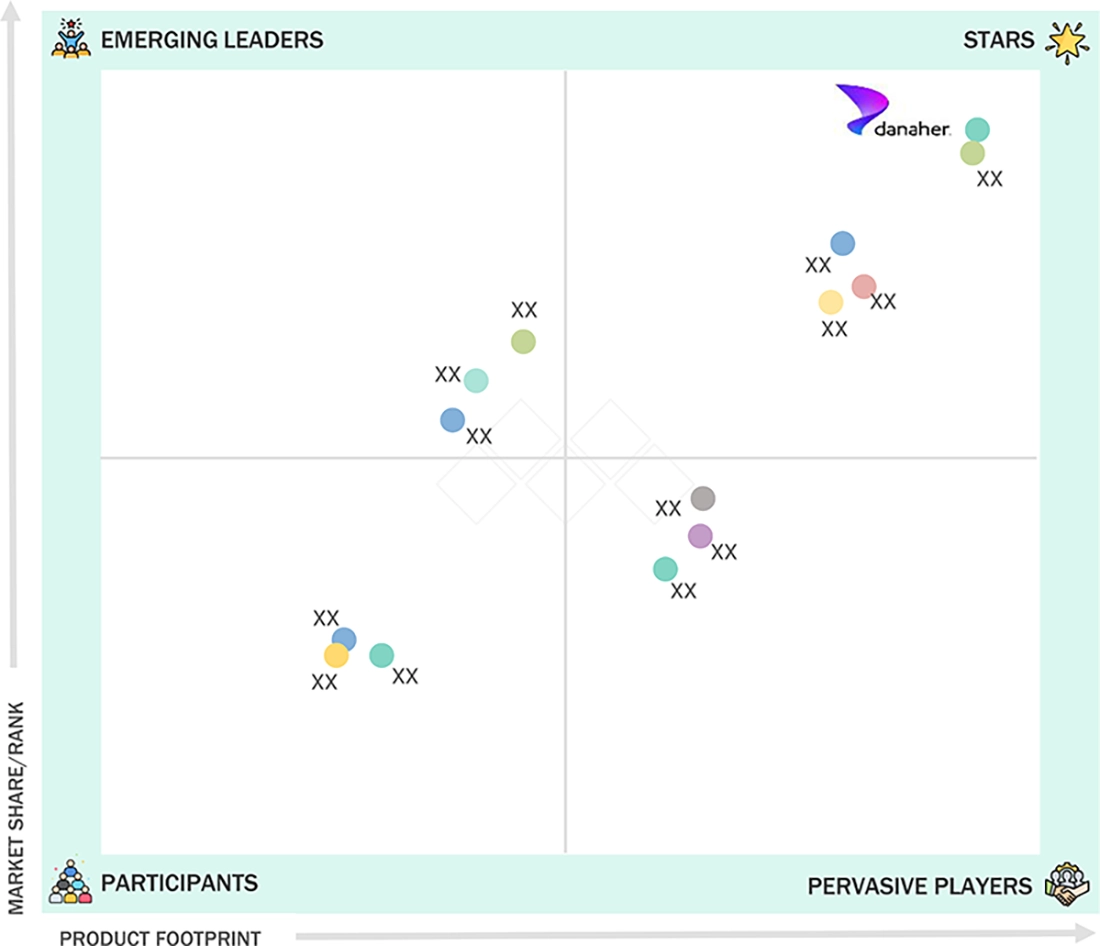
MOLECULAR INFECTIOUS DISEASE TESTING MARKET: COMPANY EVALUATION MATRIX
Danaher is a major player in the molecular infectious disease testing industry, offering a wide array of diagnostic test kits designed to detect infectious diseases through PCR-based methods and other reagents. The test kits produced by the company provide fast, accurate, and reliable results. Danaher has a strong presence in North America, Europe, and the Asia Pacific region, where it implements its molecular infectious disease testing solutions in clinics, diagnostic laboratories, and public health facilities. The company strategically collaborates with medical organizations to ensure that its tests and test kits are used effectively and efficiently. These partnerships enhance access to Danaher's tests in diagnostic laboratories and facilitate training programs to ensure that qualified personnel operate the tests properly. Additionally, Danaher consistently updates its range of tests to address emerging pathogens and evolving infection trends.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 10.28 Billion |
| Market Forecast in 2031 (Value) | USD 19.01 Billion |
| CAGR | CAGR of 11.1% from 2026–2031 |
| Years Considered | 2024–2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Million/Billion) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa, and the GCC Countries |
WHAT IS IN IT FOR YOU: MOLECULAR INFECTIOUS DISEASE TESTING MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Product Analysis | Product matrix, which provides a detailed comparison of the product portfolio of each company in the molecular infectious disease testing market. | Enables easy comparison of competitors’ offerings, helping identify gaps, overlaps, and differentiation opportunities. |
| Company Information | Additional five company profiles of players operating in the molecular infectious disease testing market. | Provides insights into competitors’ strategies, innovation focus, and partnerships, supporting strategic planning. |
RECENT DEVELOPMENTS
- December 2024 : bioMérieux (France) announced that its BIOFIRE FILMARRAY Tropical Fever (TF) Panel has received Special 510(k) clearance from the US Food and Drug Administration. This novel PCR testing solution is designed to identify pathogens quickly and accurately for patients with unexplained fever, enabling the overall optimization of treatment.
- November 2024 : QIAGEN N.V. (Netherlands) announced that the US FDA cleared the QIAstat-Dx Meningitis/Encephalitis Panel for clinical use.
- September 2024 : F. Hoffmann-La Roche Ltd. (Switzerland) launched the cobas Respiratory flex test, the first test that uses Roche's novel and proprietary TAGS (Temperature-activated Generation of Signal) technology. This technology uses multiplex polymerase chain reaction (PCR) testing, combined with color, temperature, and data processing, to identify as many as 15 pathogens in a single PCR test.
Table of Contents

Methodology
The objective of this study is to analyze key market dynamics, including drivers, opportunities, restraints, and challenges, as well as the strategies employed by key players. It aims to track developments within companies, such as acquisitions, product launches, expansions, agreements, and partnerships, to assess the competitive landscape of the molecular infectious disease testing market. This analysis will evaluate market players across various parameters, including business and product strategies.
To estimate the market size, both top-down and bottom-up approaches were employed. The estimation process for segments and subsegments utilized market breakdown and data triangulation methods.
The four steps involved in estimating the market size are:
Secondary Research
In the secondary research process, a variety of sources were utilized to gather information for this study. These included annual reports, press releases, and investor presentations from companies, as well as white papers, certified publications, and articles by recognized authors. Additionally, gold-standard and silver-standard websites, regulatory bodies, and databases such as D&B Hoovers, Bloomberg Business, and Factiva were referenced.
Primary Research
During the primary research process, we interviewed various sources from both the supply and demand sides to gather qualitative and quantitative information for this report. Our primary sources included industry experts from core and related fields, as well as preferred suppliers, manufacturers, distributors, service providers, technology developers, researchers, and organizations involved in all segments of the industry’s value chain.
We conducted in-depth interviews with a range of primary respondents, including key industry participants, subject-matter experts, C-level executives from major market players, and industry consultants. This approach allowed us to obtain and verify essential qualitative and quantitative information while also assessing future prospects.
The following is a breakdown of the primary respondents:
BREAKDOWN OF PRIMARY PARTICIPANTS:
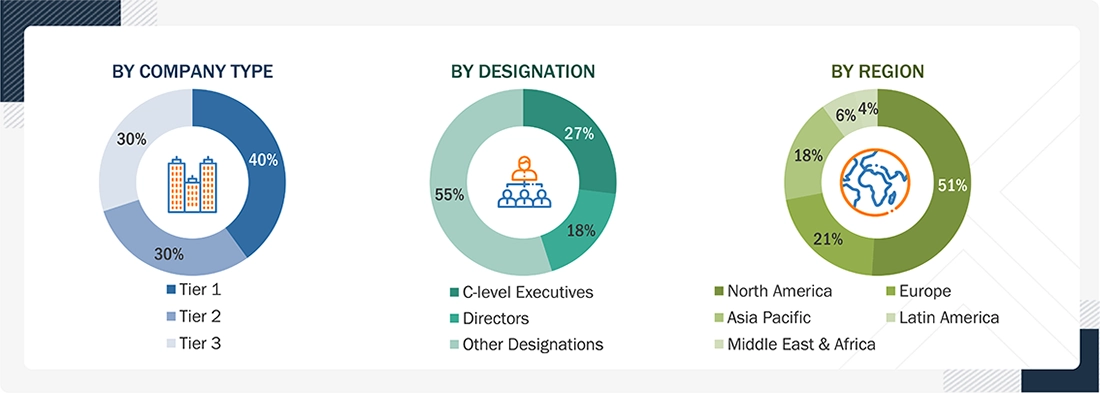
Note 1: Other designations include sales, marketing, and product managers.
Note 2: Companies are classified into tiers based on their total revenues. As of 2025, Tier 1 = >USD 100 million, Tier 2 = USD 10 million to USD 100 million, and Tier 3 = <USD 10 million.
|
COMPANY NAME |
DESIGNATION |
|
Danaher Corporation (US) |
Sales Manager |
|
F. Hoffmann-La Roche AG (Switzerland) |
Product Manager |
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
Both top-down and bottom-up approaches were used to estimate and validate the total size of the molecular infectious disease testing market. These methods were also widely used to estimate the sizes of various subsegments of the market. The research methodology used to estimate the market size includes the following:
- The key players in the industry have been identified through extensive secondary research.
- The revenues generated by leading players operating in the molecular infectious disease testing market have been determined through primary and secondary research.
- All percentage shares, splits, and breakdowns have been determined using secondary sources and verified through primary sources.
Molecular Infectious Disease Testing Market : Top-Down and Bottom-Up Approach

Data Triangulation
After arriving at the overall market size using the process described above, the total market was split into several segments and subsegments. To complete the overall market engineering process and obtain exact statistics for all segments and subsegments, data triangulation and market breakdown procedures were employed, as applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Molecular infectious disease testing is the use of molecular techniques by healthcare professionals to detect and diagnose disease-causing pathogens in human samples, such as serum, blood, urine, throat swabs, and stool, among others. Molecular diagnostics is a technique used to identify and analyze nucleic acids or proteins at a molecular level. This technique assesses an individual's genetic makeup to identify a predisposition to a particular disease or condition and diagnose it.
Key Stakeholders
- Product manufacturers, distributors, and suppliers
- Diagnostic laboratories and point-of-care testing centers
- Research laboratories and CROs
- Contract testing laboratories
- Academic universities and medical research centers
- Research and development (R&D) centers
Report Objectives
- To define, segment, and forecast the molecular infectious disease testing market, by product & service, type, test type, disease, technology, end user, and region
- To provide detailed information regarding the major factors influencing market growth (such as drivers, restraints, opportunities, and challenges)
- To analyze micromarkets with respect to individual growth trends, prospects, and contributions to the overall market
- To analyze market opportunities for stakeholders and provide details of the competitive landscape for key players
- To forecast the size of the market segments with respect to six regions: North America, Europe, the Asia Pacific, Latin America, the Middle East & Africa, and GCC countries
- To profile key players and comprehensively analyze their product portfolios, market positions, and core competencies
- To benchmark players within the market using the proprietary Company Evaluation Matrix framework, which analyzes market players on various parameters within the broad categories of business and product excellence
- To study the impact of AI on the market, along with the macroeconomic outlook for each region
Available customizations:
MarketsandMarkets offers the following customizations for this market report.
Company profiles
- An additional five company profiles of players operating in the molecular infectious disease testing market.
Country Information
- Additional country-level analysis of the molecular infectious disease testing market.
Product Analysis
- Product matrix, which provides a detailed comparison of the product portfolio of each company in the molecular infectious disease testing market.
Key Questions Addressed by the Report
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Molecular Infectious Disease Testing Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS




















Growth opportunities and latent adjacency in Molecular Infectious Disease Testing Market