
Middle East & Africa Pharmaceutical Drug Delivery Market Size, Growth, Share & Trends Analysis
Middle East & Africa Pharmaceutical Drug Delivery Market by Route of Administration (Injectable [Syringe, Auto injector], Oral [Tablet, Capsule, Syrup], Topical, Nasal, Ocular, Rectal), Application (Cancer, Diabetes, CVD, Ophthalmic) - Forecast to 2031




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
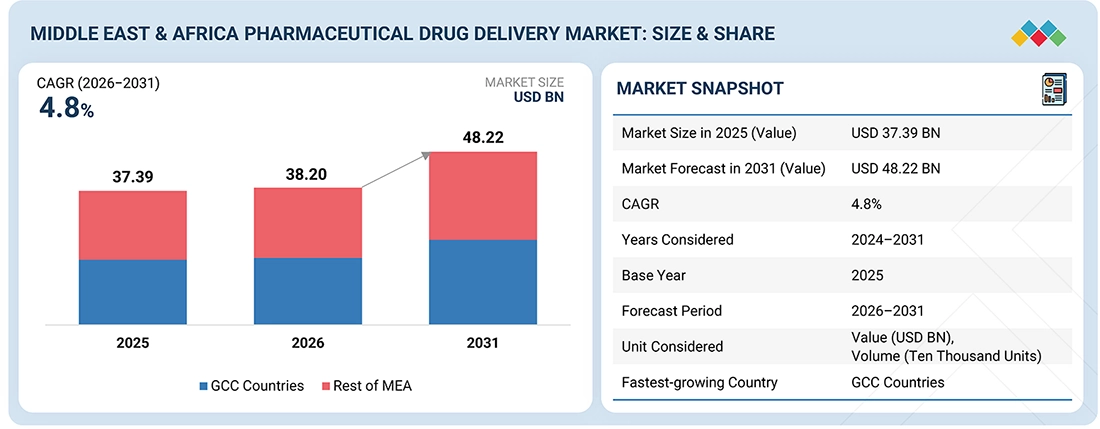
The Middle East & Africa Pharmaceutical Drug Delivery market, valued at USD 37.39 billion in 2025, stood at USD 38.20 billion in 2026 and is projected to advance at a resilient CAGR of 4.8% from 2026 to 2031, culminating in a forecasted valuation of USD 48.22 billion by the end of the period. The Middle East & Africa pharmaceutical drug delivery market is expanding steadily due to the modernization of healthcare infrastructure and the growing adoption of patient-friendly treatment modalities. The major growth driver is the increasing incidence of cancer, diabetes, respiratory disorders, and autoimmune disorders, which is driving demand for convenient and efficient drug delivery systems such as prefilled syringes, auto-injectors, inhalers, long-acting injectors, and transdermal systems. Saudi Arabia, the UAE, South Africa, Egypt, Kenya, and Nigeria, among others, are experiencing an increasing trend toward home-based treatment instead of hospital care to reduce the burden on public hospitals and facilitate patient adherence. Manufacturing capacity, packaging technology, and regulatory frameworks continue to improve in the major Middle East & Africa markets, thereby widening availability, improving treatment accuracy, and increasing the adoption of modern drug delivery solutions throughout the region.
KEY TAKEAWAYS
-
By Route of AdministrationBy route of administration, the injectable drug delivery segment dominated the market in 2025.
-
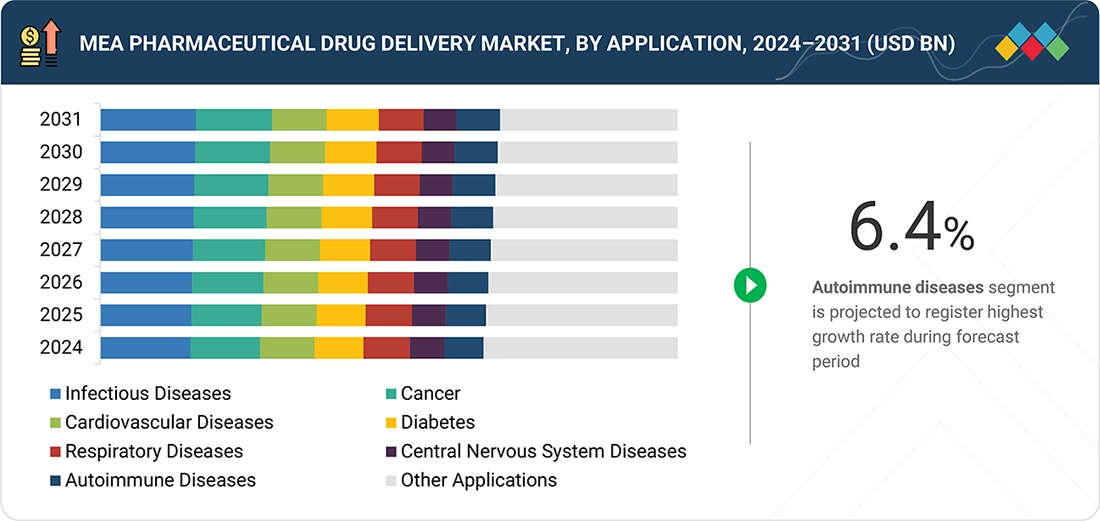
By ApplicationBy application, the autoimmune diseases segment is expected to register the highest CAGR of 6.4%, during the forecst period.
-
By Facility of UseBy facility of use, the hospitals segment dominated the market, with a share of 66.5% in 2025.
-
Competitive Landscape - Key PlayersSanofi (France), Pfizer Inc. (US), Novartis AG (Switzerland), AstraZeneca (UK), and GSK plc (UK) were identified as the star players in the Middle East & Africa pharmaceutical drug delivery market, given their extensive reach and comprehensive product portfolios.
-
Competitive Landscape - StartupsEitan Medical (Israel), Tabuk Pharmaceuticals (KSA), Pharco (KSA) have distinguished themselves among startups and SMEs due to their innovations in drug delivery systems, novel delivery technologies, and specialized formulation capabilities focused on improving patient convenience and therapeutic outcomes.
The pharmaceutical drug delivery market in Middle East & Africa is on a gradual rise. There has been an increasing demand for advanced injectable, inhalation, and transdermal systems in Saudi Arabia, the UAE, South Africa, Qatar, which is the primary reason behind the growth. The trend toward biologics and biosimilars usage in the region is the primary cause of the growing requirement for reliable, easy-to-use drug delivery formats, and it is also the biggest factor that supports the further shift of self-administration for chronic diseases. Wearable injectors, prefilled syringes, smart devices, and improved inhalers based on the latest technology give patients more freedom to use the products and hence are increasing their compliance with the treatment. However, pharmaceutical companies are still heavily investing in long- acting injectables, controlled, release formulations, and other advanced delivery solutions. Besides that, the regional regulatory authorities such as the Saudi Food and Drug Authority (SFDA), UAE Ministry of Health and Prevention (MOHAP), Egyptian Drug Authority (EDA), and South African Health Products Regulatory Authority (SAHPRA) are putting more and more emphasis on safety, accuracy, and device, drug integration. Thus, they are helping the rapid adoption of advanced and user, friendly drug delivery technologies in the Middle East & Africa.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The massive trends and changes, as well as the shift from traditional small-molecule drugs to biologics, are driving a paradigm shift in the Middle East & Africa pharmaceutical drug delivery market. The shift is marked by increased investment in next-generation injectables, wearable drug-delivery systems, and biologics-compatible technologies across key markets in the Middle East & Africa, including Saudi Arabia, the UAE, Egypt, and South Africa. The shift is also marked by the high adoption of connected and sensing-enabled devices, which can be used for digital therapy, remote monitoring, and home use, as the region increasingly seeks to reduce hospitalizations and increase patient compliance. Additionally, innovative medical device technologies, including on-body injectors, microneedle patches, nanoparticles, and smart inhalers, are increasingly transforming the drug delivery market in the Middle East & Africa, offering more precise and patient-friendly delivery. Nevertheless, persistent supply-chain constraints, cold-chain fragility in several African markets, sterility and quality control requirements, and stricter regulatory expectations for drug–device combination products from agencies such as SFDA, MOHAP, SAHPRA, and the EDA are compelling pharmaceutical manufacturers to adopt more automated, user-centric, and technology-integrated delivery platforms tailored to the MEA region.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Government investment in healthcare infrastructure

-
Rising burden of chronic diseases, such as cancer, diabetes, and cardiovascular disorders, across Middle East & Africa
Level
-
Limited local manufacturing & heavy dependence on imports
-
Shortage of skilled personnel for handling complex delivery systems
Level
-
High demand for wearable, smart, & connected delivery platforms
-
Local manufacturing & fill–finish expansion
Level
-
Supply chain constraints & cold chain fragility
-
Infrastructure gaps in rural Africa
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Government investment in healthcare infrastructure
Currently, the investments that the government is sponsoring for healthcare infrastructure development in the Middle East & Africa region are encouraging the use of modern pharmaceutical drug delivery systems. For example, some countries that invest significantly in healthcare infrastructure development include Saudi Arabia and the UAE. These investments include building new healthcare facilities, cancer treatment centers, vaccine production centers, biologics research centers, and so forth. As the Middle East & Africa region continues to improve the quality of healthcare, the use of modern drug delivery systems is likely to increase in the region.
Restraint: Limited local manufacturing & heavy dependence on imports
The growth of the Middle East & Africa pharmaceutical drug delivery market is hindered by limited production capabilities and reliance on imported medical devices, parts, and drug delivery systems. Most high-precision parts, including autoinjector components, cartridges, prefilled syringe barrels, cannulas, wearable injector components, and drug delivery systems, are imported from Europe, the US, and Asian countries. Saudi Arabia, the UAE, Egypt, and South Africa have initiated localization initiatives, but local production capabilities for advanced drug-device combinations are still in the early stages. The dependency on imported parts and drug delivery systems increases the production costs, which include international freight costs, currency risks, and customs duties, and also increases the time taken for regulatory approvals, which acts as a restraint for the drug delivery market in the Middle East & Africa.
Opportunity: High demand for wearable, smart & connected delivery platforms
In the Middle East & Africa region, the demand for wearable, smart, and connected drug delivery systems is rapidly increasing. With the rise of diabetes, oncological disorders, and autoimmune conditions, the focus is on drug delivery systems that provide accurate dosing, remote patient monitoring, and reduced clinical interventions. Pharmaceutical companies operating globally and operating in the MEA region, such as Pfizer, AstraZeneca, Novo Nordisk, and Johnson & Johnson, are mentioning the increased adoption of body-worn drug injectors, smart autoinjectors, inhalers, dosage tracking systems, and connected drug delivery systems in their annual reports. The MEA region is witnessing a boost in telemedicine services and digital healthcare services from governments such as the UAE, Saudi Arabia, Egypt, and South Africa. This is a huge opportunity for drug delivery companies that provide advanced digital drug delivery technologies that are user-friendly for end users.
Challenge: Supply chain constraints & cold chain fragility
The pharmaceutical drug delivery market in the MEA is still experiencing issues concerning supply chain, especially with the cold chain. Many advanced injectables, vaccines, biologics, and temperature-sensitive formulations require robust cold chain infrastructure, which remains uneven across countries such as Kenya, Nigeria, Ethiopia, and rural South Africa. Limited availability of specialized transport, storage facilities, and validated temperature-monitoring systems increases the risk of product degradation and delays. Additionally, global shortages of device components, longer international shipping lead times, and rising logistics costs further strain MEA supply chains. The regulatory bodies, including SFDA, MOHAP, SAHPRA, and EDA, often stress the importance of an effective and efficient cold chain.
MIDDLE EAST & AFRICA PHARMACEUTICAL DRUG DELIVERY MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Sanofi partnered with Biovac in South Africa to locally manufacture and supply inactivated polio vaccines for African countries. | Strengthens regional vaccine security | Reduces import dependency | Improves supply timelines for public immunization programs across Africa |
 |
Pfizer and Thermo Fisher expanded localized next-generation sequencing (NGS) testing across the Middle East & Africa to support oncology and precision-medicine treatment decisions. | Enables faster diagnosis | Supports advanced biologics use | Reduces sample exports | Improves access to targeted therapies across MEA |
 |
Novartis runs regional access programs across APMA (including MEA) that support wider adoption of cardiovascular, oncology, and biologic therapies that rely on injectable and long-acting delivery systems. | Expands access to advanced injectable therapies | Improves treatment continuity | Strengthens physician/patient adoption in MEA markets |
 |
AstraZeneca’s Gulf Region operations support regional rollout and medical-affairs programs for inhalation therapies and biologics used across MEA. | Improves regional launch speed for respiratory and injectable biologics | Enhances clinician training | Strengthens product availability |
 |
GSK partnered with the Abu Dhabi Department of Health to establish a regional vaccine distribution hub in the UAE serving the MEA region. | Enhances cold chain capacity | Speeds vaccine delivery | Improves supply stability for MEA public-health programs |
 |
Hikma expanded its partnership with Celltrion to commercialize multiple biosimilars across MENA, strengthening access to affordable injectable biologics. | Increases availability of specialty injectables in MEA | Spports hospital access to cost-effective biologics | Reinforces Hikma’s leadership in the region |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The pharmaceutical drug delivery industry in the Middle East & Africa is intertwined with global giants, local players, device vendors, healthcare facilities, and regulatory bodies spread over Saudi Arabia, the UAE, South Africa, and Rest of Middle East & Africa. As biologics, vaccines, and treatments for chronic diseases become more widely adopted, the region is heading toward safer and more handy delivery methods. Hence, there has been an upsurge in the use of injectables, inhalers, autoinjectors, prefilled syringes, and transdermal formats. The regulatory bodies such as the Saudi Food & Drug Authority (SFDA), UAE Ministry of Health and Prevention (MOHAP), Egyptian Drug Authority (EDA), and South African Health Products Regulatory Authority (SAHPRA) are raising the bar for the combination products and patient safety to help the market evolution. Meanwhile, the ever increasing chronic disease load and the limited capacity of public hospitals have resulted in a higher demand for home based and self administered therapies. Therefore, the adoption of wearable injectors, long, acting formulations, and drug, delivery devices with high user, friendliness is gradually becoming the norm throughout the region.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Middle East & Africa Pharmaceutical Drug Delivery Market, By Route of Administration
In 2025, the Middle East & Africa pharmaceutical drug delivery market is dominated by the injectable drug delivery segment, in terms of market share. An extensive part of the regions demand derives from biologics, monoclonal antibodies, vaccines, biosimilars, and other large molecule drugs that are mostly administered through parenteral routes. One main reason for the growing requisition of delivery solutions that are reliable, precise, and effective is a surge in the number of chronic disease cases such as cancer, diabetes, autoimmune disorders, and cardiovascular diseases in countries such as Saudi Arabia, the UAE, Qatar and South Africa. Patients and healthcare providers are starting to use more self-administration devices such as autoinjectors, prefilled syringes, pen injectors, and wearable injectors that not only help to reduce the number of clinic visits but also increase ease of use and treatment adherence. Moreover, the launch of needle-free injectors, rapid SC infusion systems, and on-body delivery devices are contributing to the rise in acceptance, usability, and trust in healthcare settings across the MEA region.
Middle East & Africa Pharmaceutical Drug Delivery Market, By Application
The autoimmune application segment in the Middle East & Africa pharmaceutical drug delivery market is expected to record the fastest growth during the forecast period. The rising number of people affected by immune-related diseases drives this trend. Additionally, the autoimmune disorders of the Middle East & Africa region are increasingly using targeted biologics, biosimilars, and immunotherapies. Due to the improved infrastructure of specialty care in countries such as Saudi Arabia, the UAE, South Africa, and Egypt, the patients of the Middle East & Africa region are increasingly using autoimmune drugs. As a result, the initiation rate of long-term autoimmune treatment, which requires a drug delivery system, is increasing. Therefore, the Middle East & Africa pharmaceutical drug delivery market, which includes autoinjectors, pen injectors, prefilled syringes, and wearable injectors, makes the autoimmune application segment the fastest-growing application segment.
Middle East & Africa Pharmaceutical Drug Delivery Market, By Facility of Use
Within the Middle East & Africa (MEA) region, in 2025 hospitals held the majority of share in the pharmaceutical drug delivery market. This is primarily driven by the rise in chronic and acute patient loads who require complex therapies administered through controlled injectable, infusion, and device-assisted systems. The increased use of biologics, oncology drugs, radiopharmaceuticals, and high-precision infusion therapies is leading to a higher number of patients seeking hospital-based treatment. Hospitals in Saudi Arabia, the UAE, South Africa are being equipped with enhanced cold chain storage, emergency services, and advanced drug, delivery technologies which are some of the reasons why hospitals continue to be the first drug delivery points, in particular for fast-acting and high risk medicines. Furthermore, using smart injectors, implantable and combination product platforms has improved treatment safety, and precision. As hospital networks in MEA are being expanded and modernized, the demand for advanced drug, delivery solutions in this segment will continue to grow.
REGION
GCC countries to exhibit fastest growth in Middle East & Africa pharmaceutical drug delivery market during forecast period
GCC countries are expected to record the highest growth rate in the the Middle East & Africa pharmaceutical drug delivery market, and this is attributed to a number of factors, which include the rapid growth in the use of biologics and biosimilars, the growth in cancer and chronic disease, and the availability of advanced treatment options in the region, which includes Saudi Arabia, the UAE, Qatar, and Kuwait. There is a significant trend toward innovative drug delivery systems in the GCC region, driven by large investments in healthcare and specialty care, and by the high growth in healthcare reform initiatives undertaken by governments in these regions. Additionally, the use of self-administration, long-acting injectables, and patient-friendly drug delivery systems has helped GCC countries drive growth in the pharmaceutical drug delivery systems market. The use of digital health solutions, remote monitoring technologies, and home healthcare has helped ensure that advanced drug delivery systems are made available in the region, enabling GCC countries to achieve the fastest growth in the Middle East & Africa region.

MIDDLE EAST & AFRICA PHARMACEUTICAL DRUG DELIVERY MARKET: COMPANY EVALUATION MATRIX
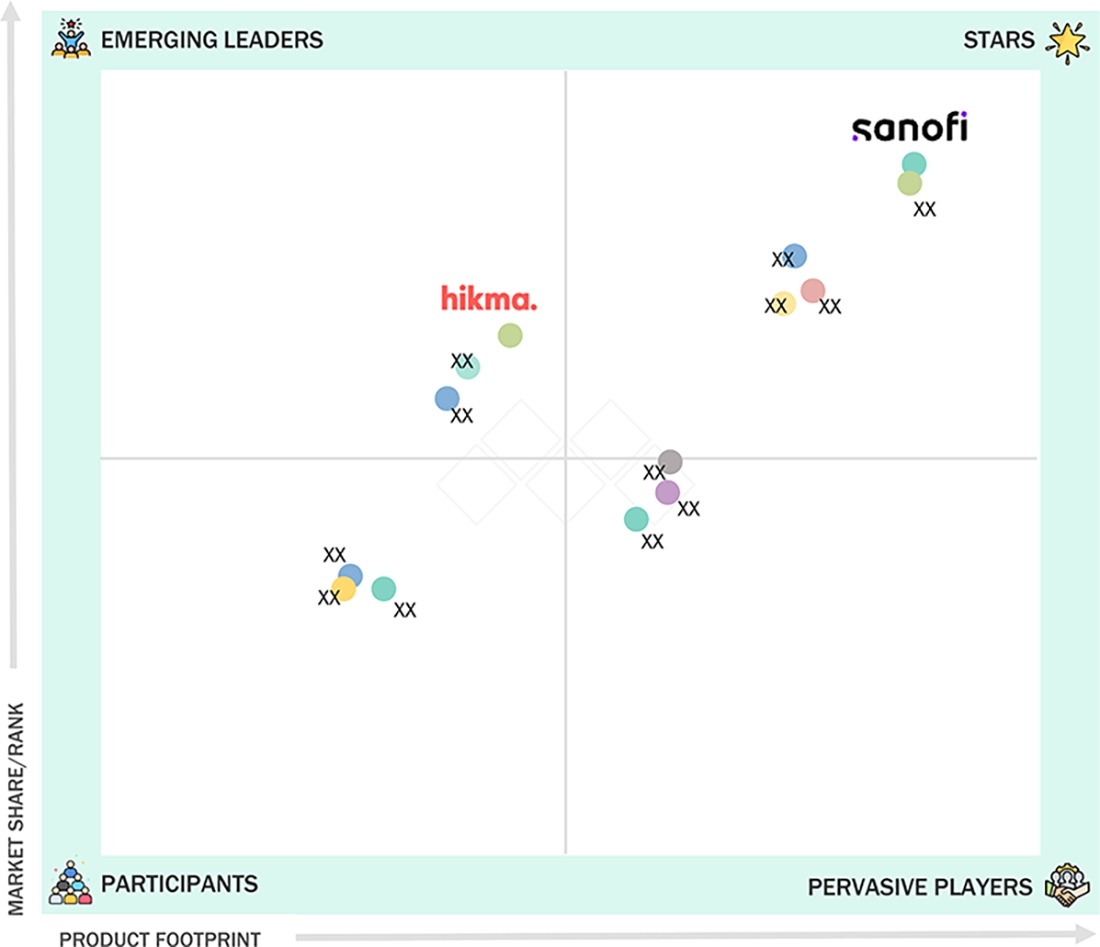
Among the major pharmaceutical companies in the Middle East & Africa drug delivery market matrix, the star players are Pfizer, Sanofi, Novartis, and AstraZeneca. These players have been able to capitalize on their robust R&D, extensive biologics and vaccines, and established presence in the major MEA markets. Moreover, their presence is further amplified by their state-of-the-art drug delivery methods, such as prefilled syringes, autoinjectors, inhalers, controlled release devices, and lipid nanoparticle formulations, supported by strong manufacturing capabilities and well-established regional supply chains. In contrast, Hikma Pharmaceuticals and Viatris are considered as the emerging leaders in the Middle East & Africa drug delivery market. Hikma Pharmaceuticals is growing its footprint by biosimilar licensing agreements, sterile injectable product development, and strong MENA sales and marketing. Viatris is leveraging its broad generics range, extensive distribution networks, and robust tender participation to scale up the availability of injectables and chronic disease therapies in the MEA.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Sanofi (France)
- Pfizer Inc. (US)
- Novartis AG (Switzerland)
- AstraZeneca (UK)
- GSK plc (UK)
- Hikma Pharmaceuticals plc (UK)
- Bayer AG (Germany)
- F. Hoffmann-La Roche (Switzerland)
- Novo Nordisk A/S (Denmark)
- BD (US)
- B. Braun (Germany)
- Abbott (US)
- Amgen (US)
- Johnson & Johnson (US)
- Viatris Inc. (US)
- Aspen Holdings (South Africa)
- EVA Pharma (Egypt)
- Cipla (India)
- West Pharmaceutical Services Inc. (US)
- Julphar (UAE)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 37.39 Billion |
| Market Forecast in 2031 (Value) | USD 48.22 Billion |
| Growth Rate | CAGR of 4.8% from 2026–2031 |
| Years Considered | 2024–2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Million/Billion), Volume (Thousand/Million Units) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Countries Covered | GCC Countries [Kingdom of Saudi Arabia, United Arab Emirates , Qatar, Kuwait, Other GCC Countries], Rest of GCC Countries |
| Parent & Related Segment Reports |
Pharmaceutical Drug Delivery Market Europe Pharmaceutical Drug Delivery Market Asia Pacific Pharmaceutical Drug Delivery Market Latin America Pharmaceutical Drug Delivery Market US Pharmaceutical Drug Delivery Market |
WHAT IS IN IT FOR YOU: MIDDLE EAST & AFRICA PHARMACEUTICAL DRUG DELIVERY MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Product Analysis | Analyzed major drug-delivery formats used in the Middle East & Africa, including injectables (prefilled syringes, autoinjectors, pen injectors, wearable injectors), inhalation systems, transdermal patches. Assessed , adoption patterns across GCC and African markets, and regulatory requirements from SFDA (Saudi Arabia), MOHAP (UAE), SAHPRA (South Africa), and EDA (Egypt). | Helped clients understand which delivery systems align with MEA treatment needs, chronic-disease patterns, and healthcare infrastructure. Identified high-growth areas such as long-acting injectables, connected home-use devices, and biologic-friendly delivery technologies. |
| Company Information | Profiled global and regional players active in MEA, including Pfizer, Roche, Novartis, Sanofi, AstraZeneca, GSK, Bayer, BD, Johnson & Johnson, Abbott, Amgen, Hikma, Aspen, Eva Pharma, Julphar, Cipla, and Viatris. Evaluated their portfolios, biologic-delivery competencies, local manufacturing capacity, GCC expansion, and regional partnership/licensing activities. | Provided clear positioning of global vs. regional competitors, showing strengths in biologics, biosimilars, autoinjector uptake, sterile injectables, and digital/connected delivery systems. Identified partnership, localization, and technology-transfer opportunities for MEA market expansion. |
| Geographic Analysis | Evaluated key MEA markets such as Saudi Arabia, UAE, South Africa, Egypt, Nigeria, and Kenya. Assessed demand for injectables, wearables, inhalation systems, and controlled-release formulations. Analyzed biologics penetration, reimbursement systems, immunization programs, and local manufacturing ecosystems. | Enabled clients to prioritize high-growth MEA regions and delivery technologies. Highlighted opportunities for localization, supply-chain improvement, regulatory acceleration, and partnerships with strong regional players such as Hikma, Aspen, Eva Pharma, Julphar, and Cipla. |
RECENT DEVELOPMENTS
- October 2025 : Hikma Pharmaceuticals expanded its partnership with Celltrion Inc. through exclusive licensing agreements for the commercialization of six additional biosimilars across the Middle East and North Africa (MENA). These biosimilars address key therapeutic areas including oncology, autoimmune diseases, allergic disorders, and ophthalmology.
- March 2025 : Johnson & Johnson Services, Inc. (US) was granted US FDA approval for TREMFYA (guselkumab) for the treatment of moderately to severely active Crohn's disease in adults. TREMFYA is the first and only interleukin-23 (IL-23) inhibitor to offer both subcutaneous (SC) and intravenous (IV) induction options. This regulatory achievement has opened up the possibility for doctors and patients to have more options in starting and continuing the treatment with either IV or SC delivery, paving the way for biologic drug delivery innovations in immune-mediated inflammatory disorders.
- December 2024 : Becton Dickinson (BD) (US) expanded its injectable drug delivery portfolio with the launch of the BD EffiFlow Advanced Prefillable Syringe Platform, designed to enhance biologics compatibility, reduce extrusion force, and improve safety in high-viscosity drug administration. This portfolio expansion strengthens BD’s leadership in prefillable syringes and supports growing demand for self-injection solutions across the US biologics and specialty drug markets.
Table of Contents

Methodology
This research study extensively utilized both primary and secondary sources. It involved analyzing various factors influencing the industry to identify segmentation types, industry trends, key players, the competitive landscape, key market dynamics, and strategies employed by key players.
Secondary Research
This research study utilized a variety of comprehensive secondary sources, including directories, databases such as Bloomberg Business, Factiva, and Dun & Bradstreet, white papers, annual reports, company house documents, investor presentations, and SEC filings from various companies. Secondary research was employed to gather information crucial for an in-depth, technical, market-oriented, and commercial analysis of the veterinary imaging market. This approach also helped identify key players in the industry and allowed for classification and segmentation based on emerging trends at the most detailed level. Furthermore, significant developments pertaining to both market and technological perspectives were documented. A database of primary industry leaders was created as part of this secondary research.
Primary Research
In the primary research process, we interviewed a range of sources from both the supply and demand sides to gather qualitative and quantitative information for this report. Primary sources from the supply side include project/sales/marketing/business development managers, presidents, CEOs, vice presidents, chairpersons, chief operating officers, chief strategy officers, directors, chief information officers, and chief medical information officers related to the biochips market. Primary sources from the demand side include researchers in biotechnology and pharmaceutical companies, healthcare professionals from hospitals and diagnostic centers, academic & research institutes and contract research organizations.
The following is a breakdown of the primary respondents:
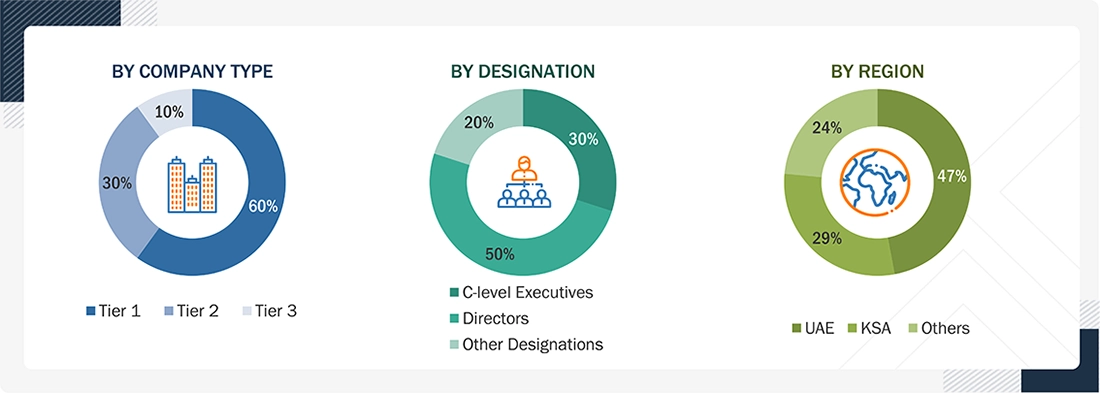
Note 1: C-level executives include CEOs, COOs, CTOs, and VPs.
Note 2: Other designations include sales, marketing, and product managers.
Note 3: Tiers are defined based on a company’s total revenue. As of 2024: Tier 1 = >USD 1 billion, Tier 2 = USD 500 million to USD 1 billion, and Tier 3 = <USD 500 million.
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The total size of Middle East & Africa pharmaceutical drug delivery market was determined after data triangulation from three approaches, as mentioned below. After each approach, the weighted average of the three approaches was taken based on the level of assumptions used in each approach.

Data Triangulation
After arriving at the market size, the total market was divided into several segments and subsegments. Data triangulation and market breakdown procedures were employed wherever applicable to complete the overall market engineering process and arrive at the exact statistics for all segments and subsegments.
Market Definition
Pharmaceutical Drug delivery is a method or process of administration of a pharmaceutical drug to safely attain its desired therapeutic effect. Advancements in drug delivery offer several benefits, such as ease of use, convenience, and patient compliance. Drug delivery technologies are used for the targeted delivery and/or controlled release of therapeutic agents.
Key Stakeholders
- Pharmaceutical manufacturing companies
- Original equipment manufacturing companies
- Suppliers and distributors of pharmaceutical products and medical devices in Middle East & Africa.
- Healthcare service providers
- Teaching hospitals and academic medical centers
- Health insurance players
- Middle East & Africa Government bodies
- Medical research institutes
- Business research and consulting service providers
- Venture capitalists
- Market research and consulting firms
Report Objectives
- To define, describe, and forecast the Middle East & Africa pharmaceutical drug delivery market on the basis of pharmaceutical drug delivery product- route of administration, application, facility of use, and country
- To provide detailed information regarding the major factors influencing the market growth (such as drivers, restraints, opportunities, and challenges)
- To strategically analyze micromarkets1 with respect to individual growth trends, future prospects, and contributions to the overall market
- To analyze the opportunities in the market for key stakeholders and provide details of the competitive landscape for major market leaders
- To forecast the size of the market segments with respect to Latin America countries, namely, Brazil, Mexico, Argentina, Colombia, Peru, Chile and Rest of Latin America
- To profile the key market players and comprehensively analyze their market shares and core competencies
- To track and analyze competitive developments such as mergers and acquisitions, new product developments, partnerships, agreements, collaborations, and expansions in the Latin America pharmaceutical drug delivery market.
Available customizations:
- With the given market data, MarketsandMarkets offers customizations as per your company’s specific needs. The following customization options are available for the Latin America pharmaceutical drug delivery market report.
- PRODUCT ANALYSIS
- Product Matrix, which gives a detailed comparison of the product portfolios of the top five global players.
- COMPANY INFORMATION
- Detailed analysis and profiling of additional market players (up to 5 players)
- GEOGRAPHIC ANALYSIS
- Further breakdown of the Other GCC Countries & Rest of Middle East & Africa pharmaceutical drug delivery market into among Oman, Bahrain and other.
- COUNTRY-LEVEL VOLUME ANALYSIS BY ROUTE OD ADMINISTRATION
- Quantifies volumes by major service types for selected countries to show demand patterns. Route of Administration Market Share Analysis (Top 5 Players).
- BY ROUTE OF ADMINISTRATION MARKET SHARE ANALYSIS (TOP 5 PLAYERS)
- Route of Administration Market Share Analysis (Top 5 Players).
- ANY CONSULT/CUSTOM REQUIREMENTS AS PER CLIENT REQUESTS
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Middle East & Africa Pharmaceutical Drug Delivery Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS




















Growth opportunities and latent adjacency in Middle East & Africa Pharmaceutical Drug Delivery Market