
Europe Medical Simulation Market Size, Growth, Share & Trends Analysis
Europe Medical Simulation Market by Offering (Anatomical Models [Patient (High Fidelity), Surgical (Laparoscopic, Ortho, Gynae, Spine), Trainers, Ultrasound, Eye]), Technology (3D Printing, Virtual Patient, Procedural Rehearsal), End User - Forecast 2030




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
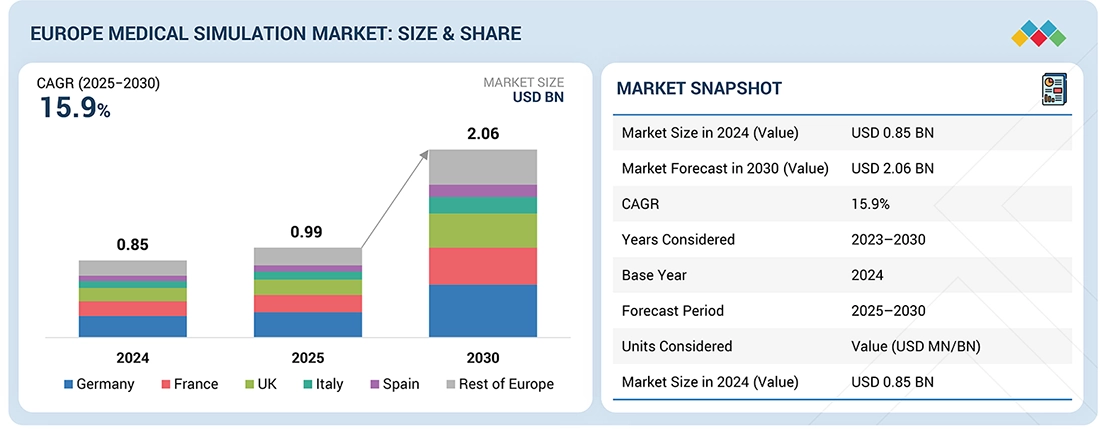
The Europe Medical Simulation market, valued at USD 0.85 billion in 2024, stood at USD 0.99 billion in 2025 and is projected to advance at a resilient CAGR of 15.9% from 2025 to 2030, culminating in a forecasted valuation of USD 2.06 billion by the end of the period. The Europe healthcare simulation market is growing steadily, driven by a strong emphasis on patient safety, standardized medical training, and widespread adoption of simulation-based education across hospitals and academic institutions (Karolinska Institutet, Imperial College London, etc.).
KEY TAKEAWAYS
-
By CountryGermany is expected to dominate the market with a share of 28.0% in 2024.
-
By Product & ServiceBy product & service, the web-based simulation segment is expected to register the highest CAGR of 17.5%.
-
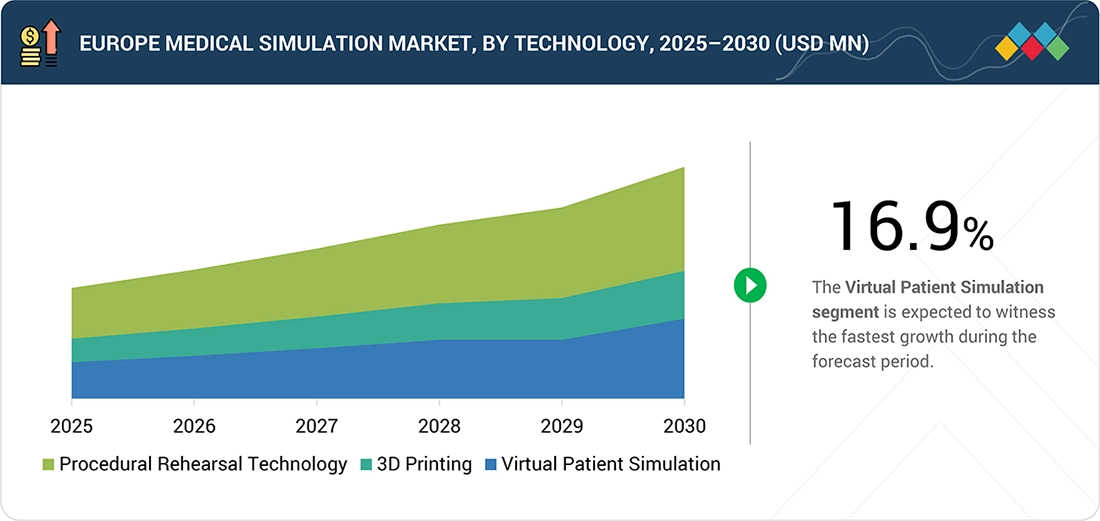
By TechnologyBy technology, the virtual patient simulation segment is expected to register the highest CAGR of 16.9%.
-
By End UserBy end user, the academic institutes segment held the largest market share.
-
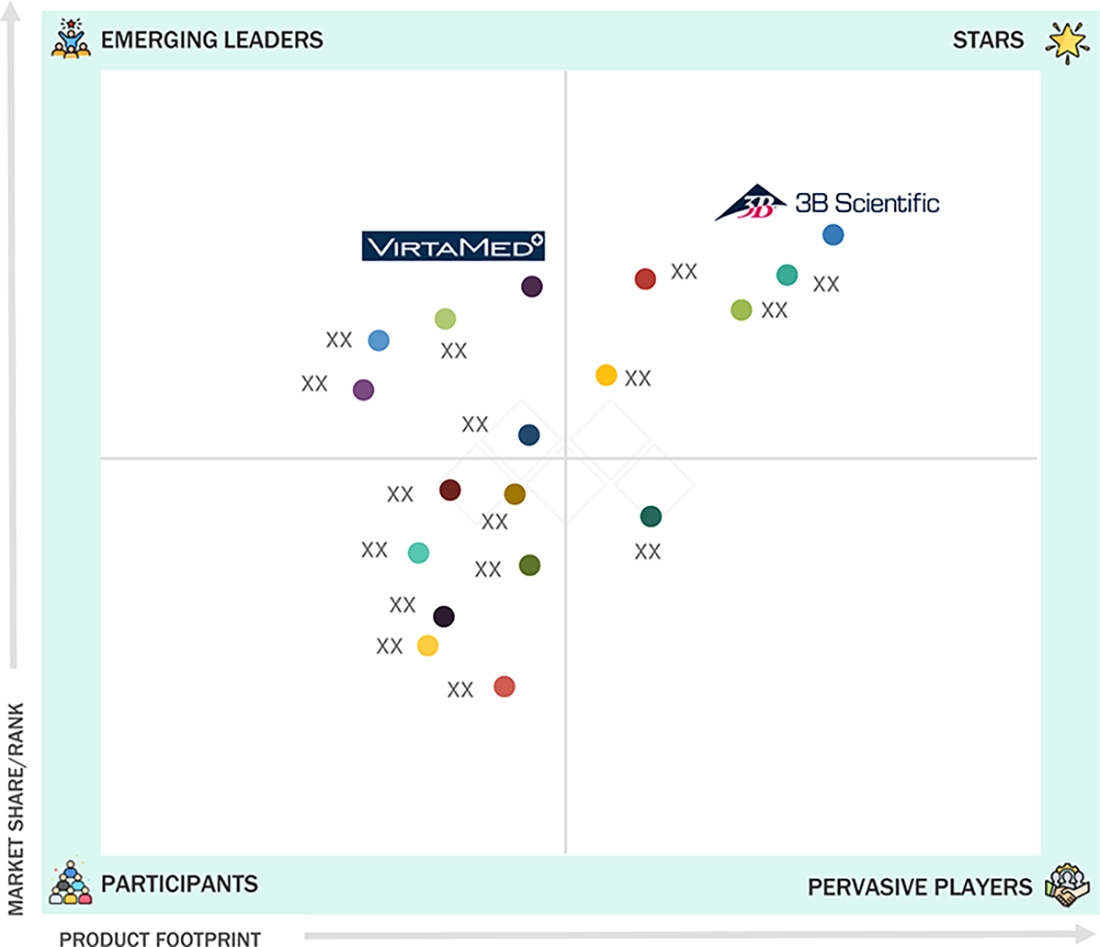
Competitive LandscapeSurgical Science Sweden AB, Laerdal Medical, and 3B Scientific were identified as some of the star players in the market, given their strong market share and product footprint.
-
Competitive LandscapeHaag-Streit AG and HRV Simulation, among others, have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas, underscoring their potential as emerging market leaders
Rising digital health investments and increased use of high-fidelity simulators and VR-based training in Germany, the UK, France, and the Nordics are driving Europe’s healthcare simulation market. For example, medical universities and teaching hospitals in the UK (King’s College London) and Germany (Charité – Universitätsmedizin Berlin) are integrating VR and surgical simulators into clinical training, supported by strong public healthcare systems and cross-border education initiatives.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The medical simulation market in Europe is being driven by the growing adoption of high-fidelity simulators, portable platforms, and immersive VR/AR to improve training efficiency, patient safety, and workforce readiness. Hospitals (Hospital Virtual Valdecilla, Spain), academic institutions, and military organizations are shifting from traditional training models to scalable, technology-enabled simulations that support faster skill development, standardized education, and risk reduction across healthcare systems.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Strong focus on patient safety, quality improvement, and risk reduction across European health systems

-
Rapid adoption of digital and VR-based medical education
Level
-
Limited public funding for simulation centers
-
Uneven adoption across European regions
Level
-
Scale VR and AR simulation to address clinician shortages in Europe
-
Healthcare workforce shortages driving simulation adoption
Level
-
Fragmented healthcare and education systems across countries create inconsistent adoption
-
Operational challenges
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Strong focus on patient safety, quality improvement, and risk reduction across European health systems
There is a growing emphasis across Europe on patient safety and quality improvement that is driving medical simulation toward reducing clinical risk and improving outcomes. Simulation is being incorporated into training, competency assessment, and quality programs by regulatory bodies and national health systems to minimize medical errors and standardize the delivery of care. For example, the UK NHS uses simulation in high-risk areas such as emergency care and surgery to improve team coordination, while France’s Haute Autorité de Santé promotes simulation for continuous professional development and crisis management.
Restraint: Limited public funding for simulation centers
The lack of governmental spending in simulation centers is an important factor hampering the medical simulation market in Europe, and this is especially true in public health systems where government spending is strictly governed. There are instances where the healthcare and academic community is presented with mixed situations, such as resource allocation and patient needs, which leaves less spending on expensive simulation tools and facilities. There are possibilities in such cases where equitable distribution in regions might not occur, and there is a lack of advancement in high-end simulators related to VR and AI technology.
Opportunity: Scale VR and AR simulation to address clinician shortages in Europe
Europe's growing clinician shortage, driven by aging populations, burnout, and limited training capacity, is accelerating demand for VR- and AR-based medical simulation. Immersive, location-independent simulation allows for quicker acquisition of skills and standardized training with minimal reliance on scarce clinical placements or senior staff time. For instance, France's Assistance Publique-Hôpitaux de Paris uses simulation, including VR-based training, to upskill residents and nursing staff en masse while ensuring patient safety. Consequently, VR and AR simulation offer a very good market opportunity to increase workforce capacity and enhance care quality within European healthcare systems.
Challenge: Fragmented healthcare and education systems across countries create inconsistent adoption
Europe’s fragmented healthcare and medical education systems create uneven adoption of medical simulation, driven by differences in regulation, funding, and training standards across countries. For example, the UK’s NHS has more widely embedded simulation into clinical training, while adoption in parts of Southern and Eastern Europe remains limited. This lack of harmonization makes cross-border scaling difficult for vendors and slows the broader growth of the Europe medical simulation market.
EUROPE MEDICAL SIMULATION MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
VR surgical simulators for European hospitals and medical schools | Improves surgical accuracy and speeds surgeon training |
 |
High-fidelity manikins for emergency and critical care training | Enhances patient safety and standardized care |
 |
Patient simulators for European medical education | Improves clinical decision-making, reduces training risk |
 |
Endovascular and procedural simulators for teaching hospitals | Strengthens hands-on procedural skills |
 |
Task trainers for residency and skills-based training | Accelerates competency-based education |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The Europe medical simulation ecosystem includes established vendors such as Surgical Science Sweden, Gaumard Medical, and Laerdal Medical, alongside startups like UpSurgeOn and Inovus Limited. Supported by the EU Medical Device Regulation and ASPiH, these players serve academic institutes, hospitals, and military organizations for training, patient safety, emergency preparedness, and workforce upskilling.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Europe Medical Simulation Market, By Product & Service
In 2024, healthcare simulation anatomical models held the largest share of the Europe medical simulation market due to their extensive use in foundational medical education and procedural training. Medical schools and teaching hospitals across Europe rely on anatomical models for hands-on learning and core clinical skill development. For example, institutions such as Karolinska Institute (Sweden) and University of Heidelberg Medical Faculty (Germany) widely use anatomical models in medical and nursing education, as they offer cost-effective, durable, and standardized training aligned with Europe’s academically driven healthcare education system.
Europe Medical Simulation Market, By Technology
In 2024, virtual patient simulation held the largest share of the Europe medical simulation market, driven by Europe’s strong emphasis on standardized, evidence-based medical education. Many medical institutions across Germany, the UK, France, and the Nordic region are adopting virtual patients to train physicians in multilingual and diverse clinical scenarios. For example, institutions such as University College London Medical School (UK) and University of Copenhagen Faculty of Health and Medical Sciences (Denmark) use virtual patient platforms to support clinical reasoning and communication skills training. These solutions are well-suited to Europe’s publicly funded healthcare systems, as they provide scalable and cost-efficient training without reliance on physical simulation infrastructure.
Europe Medical Simulation Market, By End User
In 2024, academic institutes held the largest share of the Europe medical simulation market, driven by the focus on structured medical training in Europe. The medical schools, nursing colleges, as well as teaching hospitals affiliated with universities, have largely embraced simulation-based training as part of building clinical skills and answering the call for patient safety requirements. For instance, universities such as Université Paris Cité in France, as well as the University of Oslo Faculty of Medicine in Norway, have incorporated simulation solutions into their curriculum, making academic institutes the main adopters in the region for healthcare simulation solutions.
EUROPE MEDICAL SIMULATION MARKET: COMPANY EVALUATION MATRIX
In the Europe Medical Simulation market matrix, 3B Scientific (Star Player) leads with a dominant presence, a comprehensive portfolio of anatomical models, patient simulators, and digital learning tools that support standardized medical education and clinical skills training. VirtaMed AG (Emerging Leader) is known for its high-precision mixed-reality surgical simulators used in orthopedics, gynecology, urology, and other procedural specialties.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Surgical Science Sweden AB (Sweden)
- Laerdal Medical (Norway)
- 3B Scientific (Germany)
- Mentice AB (Sweden)
- Limbs & Things (UK)
- Kyoto Kagaku Co., Ltd. (Japan)
- CAE Inc. (Canada)
- Simulaids (US)
- Anatomage (US)
- Gaumard Scientific Co (US)
- Nasco Healthcare (US)
- Inovus Limited (UK)
- VirtaMed AG (Switzerland)
- SYNBONE AG (Switzerland)
- Erler-Zimmer Medical GmbH (Germany)
- Medical-X (Netherlands)
- KaVo Dental (Germany)
- Atlay Scientific (Italy)
- Trucorp (Ireland)
- Simendo (Netherlands)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2024 (Value) | USD 0.85 Billion |
| Market Forecast in 2030 (Value) | USD 2.06 Billion |
| Growth Rate | CAGR of 15.9% from 2025-2030 |
| Years Considered | 2023-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD Million/Billion) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Countries Covered | Germany, France, UK, Italy, Spain, and Rest of Europe |
| Parent & Related Segment Reports |
Healthcare Simulation Market North America Healthcare Simulation Market US Healthcare Simulation Market Asia Pacific Healthcare Simulation Market Trauma Simulation Market Patient Simulation Market Medical Immersive Simulation Market Medical Training Simulation Market Nurse Training Software Market Medical Simulation Software Market Surgical Simulation Market |
WHAT IS IN IT FOR YOU: EUROPE MEDICAL SIMULATION MARKET REPORT CONTENT GUIDE
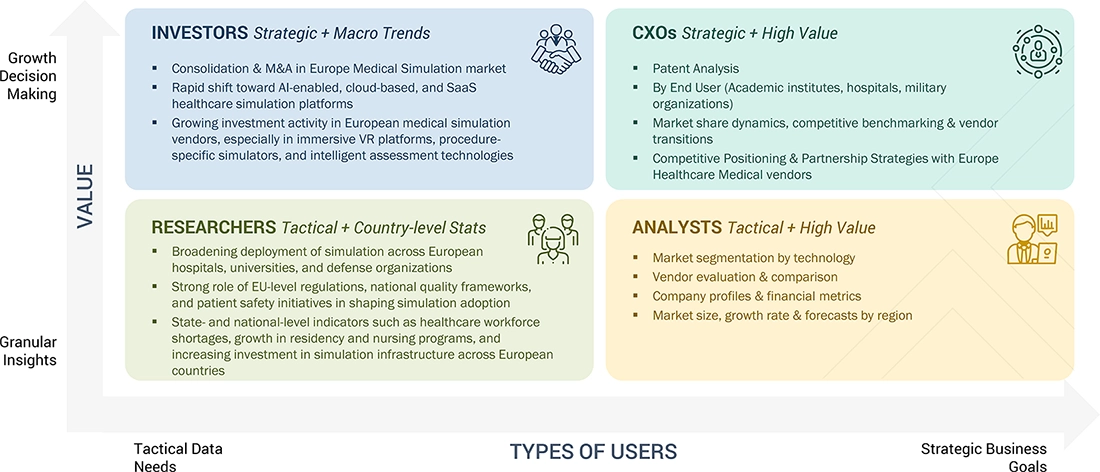
DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Local Competitive Landscape | Benchmarking of European medical simulation vendors by simulator type, technology maturity, pricing, and end users. | Supports vendor selection and competitive positioning. |
| Country Level Landscape | Analysis of country regulations, funding, and adoption across Europe. | Faster, compliant market entry. |
| Pricing Strategy | Assessment of European pricing models, costs, and demand trends. | Improved pricing fit and investment focus. |
| Technology Adoption by Region | Mapping adoption of manikins, VR/AR, and virtual patients across Europe. | Guides product and technology strategy. |
RECENT DEVELOPMENTS
- March 2025 : Surgical Science completed the acquisition of Intelligent Ultrasound Group PLC, a UK-based ultrasound simulation technology company, expanding its product portfolio in ultrasound simulation and strengthening its European presence.
- September 2024 : Gaumard Scientific partnered with Norso Medical Ltd. to distribute simulation-based healthcare training solutions, enhancing the reach of Gaumard’s products in the Irish market.
- September 2024 : Mentice entered a three-year collaboration with the German Society of Interventional Cardiology to standardize training and advance simulation-based education across Germany.
Table of Contents

Methodology
The study involved five major activities to estimate the current size of the Europe Medical Simulation Market. Exhaustive secondary research was done to collect information on the market and its different subsegments. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. Thereafter, market breakdown and data triangulation procedures were used to estimate the market size of the segments and subsegments.
Secondary Research
This research study used secondary sources, directories, and databases such as Dun & Bradstreet, Bloomberg Business, and Factiva; white papers, annual reports, and companies’ house documents; investor presentations; and the Securities and Exchange Commission (SEC) filings of companies. The market for companies providing healthcare simulation solutions is assessed using secondary data from paid and free sources. This involves analyzing the product portfolios of major players in the industry and evaluating these companies based on their performance and quality. Various resources were utilized in the secondary research process to gather information for this study. The sources include annual reports, press releases, investor presentations, white papers, academic journals, certified publications, articles by recognized authors, directories, and databases. The secondary research process involved referring to various secondary sources to identify and collect information related to the study. These sources included annual reports, press releases, investor presentations of healthcare simulation vendors, forums, certified publications, and whitepapers. The secondary research was used to obtain critical information on the industry’s value chain, the total pool of key players, market classification, and segmentation from the market and technology-oriented perspectives.
Primary Research
In the primary research process, various supply and demand sources were interviewed to obtain qualitative and quantitative information for this report. Primary sources are mainly industry experts from the core and related industries and preferred suppliers, manufacturers, distributors, technology developers, researchers, and organizations related to all segments of this industry’s value chain. In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, among other experts, to obtain and verify the critical qualitative and quantitative information and assess prospects.
Primary research was conducted to identify segmentation types, industry trends, key players, and key market dynamics such as drivers, restraints, opportunities, challenges, industry trends, and strategies adopted by key players.
After completing the market engineering process, which includes calculations for market statistics, market breakdown, size estimations, forecasting, and data triangulation, extensive primary research was conducted. This research aimed to gather information and verify the critical numbers obtained during the market analysis. Additionally, primary research was conducted to identify different types of market segmentation, analyze industry trends, evaluate the competitive landscape of healthcare simulation solutions offered by various players, and understand key market dynamics such as drivers, restraints, opportunities, challenges, industry trends, and strategies employed by key market participants.
In the complete market engineering process, the top-down and bottom-up approaches and several data triangulation methods were extensively used to perform the market estimation and market forecasting for the overall market segments and subsegments listed in this report. Extensive qualitative and quantitative analysis was performed on the complete market engineering process to list the key information/insights throughout the report.
Market Size Estimation
The market size estimates and forecasts provided in this study are derived through a mix of the bottom-up approach (revenue share analysis of leading players) and the top-down approach (assessment of utilization/adoption/penetration trends by product and service, technology, end user, and region).
Data Triangulation
After arriving at the overall market size, the market size estimation processes split the market into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics of each market segment and subsegment, the data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides in the Europe Medical Simulation Market.
Market Definition
Medical/Healthcare simulation uses virtual patients, mannequins, task trainers, or computer-based environments to replicate real-life clinical situations for training, education, evaluation, or system improvement without risking patient safety. It is a strategic tool that enables safe, repeatable, and immersive learning experiences, enhancing clinical performance, reducing medical errors, and improving healthcare delivery outcomes.
Stakeholders
- Healthcare simulation vendors
- Government bodies
- Healthcare service providers
- Clinical/physician centers
- Healthcare professionals
- Health IT service providers
- Healthcare associations/institutes
- Ambulatory care centers
- Venture capitalists
- Distributors and resellers
- Maintenance and support service providers
- Integration service providers
- Healthcare payers
- Military organizations
- Advocacy groups
- Investors and financial institutions
- Industry associations and trade groups
Report Objectives
- To define, describe, and forecast the Europe Medical Simulation Market by product & service, technology, end user, and region
- To provide detailed information regarding the major factors, such as drivers, restraints, opportunities, and challenges, influencing the growth of the Europe Medical Simulation market
- To analyze micromarkets with respect to individual growth trends, prospects, and contributions to the overall Europe Medical Simulation Market
- To assess the Europe Medical Simulation Market with regard to Porter’s five forces, regulatory landscape, value chain, ecosystem map, patent protection, impact of 2025 US tariff and AI/Gen AI on the market under study, and key stakeholders’ buying criteria
- To analyze opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders in the Europe Medical Simulation market
- To profile the key players in the Europe Medical Simulation Market and comprehensively analyze their core competencies and market shares
- To track and analyze competitive developments such as agreements, partnerships, and acquisitions; expansions; product launches and enhancements; and R&D activities in the Europe Medical Simulation Market
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Europe Medical Simulation Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS





















Growth opportunities and latent adjacency in Europe Medical Simulation Market