
North America Capnography Equipment Market Size, Growth, Share & Trends Analysis
North America Capnography Equipment Market, by Product (Multiparameter, Standalone, Software, Accessories), Technology (Mainstream, Sidestream, Microstream), Application (Cardiac Care, Respiratory Monitoring), End User (Hospitals, ASC), & Country - Forecast to 2031




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
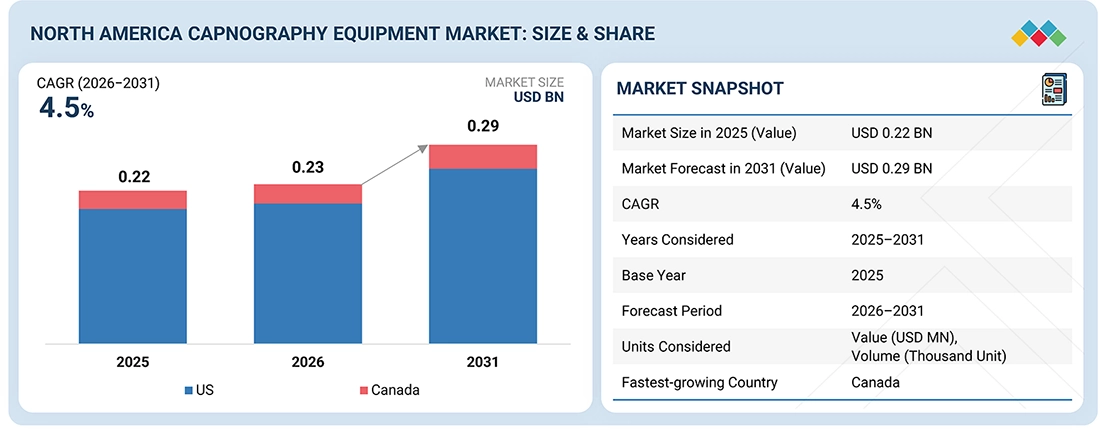
The North America Capnography Equipment Market, valued at USD 0.22 billion in 2025, stood at USD 0.23 billion in 2026 and is projected to advance at a resilient CAGR of 4.5% from 2026 to 2031, culminating in a forecasted valuation of USD 0.29 billion by the end of the period. The primary driver of the North America capnography equipment market is the increasing number of surgical and diagnostic procedures that require anesthesia and procedural sedation. This trend necessitates continuous respiratory monitoring for patient safety. Additionally, the rising rates of chronic respiratory and cardiovascular diseases, along with a rapidly aging population, have led to a greater demand for real-time carbon dioxide monitoring in hospitals, emergency rooms, and intensive care units. Furthermore, stringent regulatory and clinical safety standards, particularly the requirement for capnography during sedation and mechanical ventilation, have significantly affected the use of this technology in healthcare facilities. The region's advanced healthcare systems, high healthcare spending, and quick adoption of innovative patient monitoring solutions also contribute to market expansion. Moreover, the increasing use of capnography in non-intubated patients, ambulatory surgical centers, and emergency medical services is expanding its clinical applications. Innovations such as portable and handheld devices, integration with multi-parameter monitoring systems, and more reliable sensors are driving demand for both replacement products and new installations, thus fostering continuous growth in the North America capnography market.
KEY TAKEAWAYS
-
By CountryThe US accounted for the largest share of 88.0% of the North America capnography equipment market in 2025.
-
By OfferingBased on product, the capnography accessories and disposables segment accounted for the largest share of 54.5% of the North America capnography equipment market in 2025.
-
By ApplicationBased on technology, the mainstream capnography segment accounted for the largest share of the North America capnography equipment market in 2025.
-
By TechnologyBased on application, the cardiac care segment accounted for the largest share of the North America capnography equipment market in 2025.
-
By End UserBased on end user, the hospitals segment is projected to register the highest growth rate in the North America capnography equipment market during the forecast period.
-
COMPETITIVE LANDSCAPEMedtronic and Koninklijke Philips N.V. were recognized as star players due to their established strong product portfolios.
-
COMPETITIVE LANDSCAPEInfinium Medical has positioned itself as a leading startup and SME, thanks to its strong product portfolio and effective business strategy.
The growth of the capnography equipment market in North American is driven by several factors, including the rising prevalence of respiratory and chronic diseases, the implementation of stringent clinical guidelines that require continuous monitoring during anesthesia and sedation, and a strong focus on patient safety. The region benefits from a well-established healthcare infrastructure, high healthcare spending, and favorable reimbursement policies, all of which support the widespread adoption of this technology. Additionally, advancements in portable and combined capnography devices are expanding their use beyond hospitals to include emergency and outpatient settings. New product launches and partnerships among key industry players also contribute to market expansion.
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
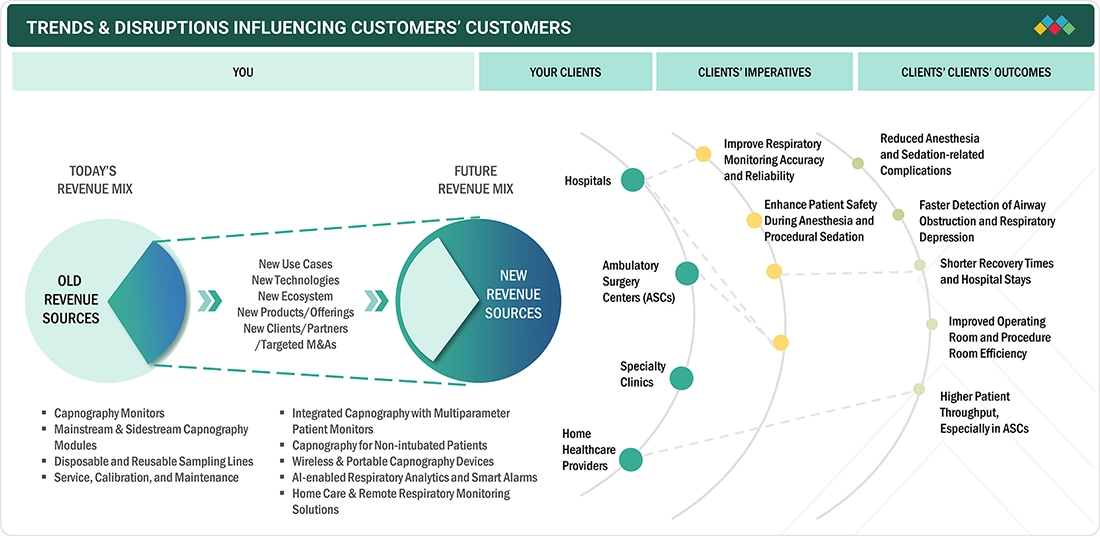
The North America capnography equipment market is currently experiencing significant changes driven by evolving healthcare needs and technological advancements. The market is shifting away from its traditional revenue sources, which have primarily come from standard capnography monitors, modules, and disposable sampling lines. Moving forward, the focus is expected to be on new technologies, innovative use cases, and integrated solutions. Emerging innovations, such as wireless and portable capnography devices, AI-enabled respiratory analytics, multiparameter patient monitoring, and remote or home monitoring solutions, are predicted to account for a substantial portion of future revenues. These advancements aim to provide more accurate respiratory monitoring, enhance patient safety during anesthesia and procedural sedation, facilitate early detection of respiratory issues, and improve workflows in hospitals, ambulatory surgery centers, specialty clinics, and home healthcare settings. Ultimately, these improvements are expected to lead to faster recoveries, increased patient throughput, and better clinical outcomes.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Rising prevalence of respiratory and cardiovascular diseases

-
Growing adoption of advanced patient monitoring systems in hospitals and ICUs
Level
-
High cost of capnography monitors and accessories
Level
-
Integration with telemedicine and remote patient monitoring platforms
-
Development of cost-effective disposable accessories and sensors
Level
-
Stringent regulatory requirements for medical device approval
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Rising prevalence of respiratory and cardiovascular diseases
The North America capnography equipment market is experiencing significant growth, primarily driven by the increasing incidence of respiratory and cardiovascular diseases. Continuous, real-time monitoring of ventilation and gas exchange is becoming essential in clinical settings. Chronic respiratory conditions such as COPD, asthma, and sleep apnea are affecting a growing number of adults in the region, leading healthcare providers to adopt capnography systems more widely across hospitals, emergency departments, and critical care settings. These systems help detect respiratory compromise early, enabling more effective ventilation for patients. Additionally, the rising prevalence of cardiovascular diseases often results in problems with pulmonary function, necessitating complex surgical and intensive care procedures. This prompts more cardiothoracic and anesthesiology teams to employ capnography to ensure patient hemodynamics and safety during cardiac and perioperative interventions. As a result, these trends are driving physicians to adopt modern capnography solutions to enhance treatment outcomes and address the increasing healthcare demands associated with these chronic conditions. This, in turn, is stimulating growth in the North America capnography market.
Restraint: High cost of capnography monitors and accessories
In the North America capnography equipment market, the high cost of capnography monitors and their peripherals poses a significant challenge to the broader adoption of these products, particularly among healthcare facilities with limited budgets. Advanced capnography systems, whether standalone monitors or integrated multi-parameter devices, are quite expensive. Standalone monitors typically cost several thousand dollars, with recurring expenses for consumables such as sampling lines, sensors, and filters. As a result of these steep initial and ongoing costs, small hospitals, clinics, and ambulatory care settings may be hesitant to invest in or upgrade to the latest capnography technologies, despite their clinical benefits. Furthermore, inconsistent reimbursement policies for capnography procedures in outpatient and non-hospital settings exacerbate the cost issue, leading some providers to refrain from purchasing these devices and accessories unless required by guidelines or supported by strong reimbursement. Consequently, the high price remains a significant barrier to market entry, resulting in slow growth in segments sensitive to capital expenditures and ongoing operational costs.
Opportunity: Integration with telemedicine and remote patient monitoring platforms
In the North America capnography equipment market, a significant opportunity exists in integrating this technology with telemedicine and remote patient monitoring platforms. As the healthcare system increasingly focuses on patient-centric and connected models, digital health solutions and remote care are becoming more prevalent. Capnography instruments, particularly portable and wearable devices, are now equipped with wireless communication and cloud-based data sharing. This innovation allows clinicians to access patients' real-time respiratory data even when they are not physically present in a hospital. This capability enables continuous monitoring for patients with chronic respiratory diseases, those recovering from surgery, or individuals receiving care at home. Early detection of deterioration in their condition enables timely medical intervention. By integrating capnography outputs with telemedicine platforms, healthcare providers can better coordinate care, conduct virtual consultations, and manage chronic conditions more effectively. Moreover, this integration diversifies the use of capnography equipment beyond acute care, presenting new opportunities for market expansion in North America.
Challenge: Stringent regulatory requirements for medical device approval
A significant challenge in the North America capnography equipment market is the strict regulatory requirements for medical device approval, particularly in the US. Capnography devices must comply with rigorous safety, efficacy, and quality standards set by regulatory authorities such as the US Food and Drug Administration (FDA). The approval process typically requires extensive clinical test data, thorough documentation, and lengthy review periods, which can significantly delay product launches and increase development costs for manufacturers. Additionally, even minor changes to a product already on the market may require a complete reassessment, further postponing time to market. These complex, lengthy regulatory procedures not only create barriers to new entrants and smaller businesses but also hinder the pace of innovation and the broader adoption of advanced capnography technologies in healthcare facilities across North America.
NORTH AMERICA CAPNOGRAPHY EQUIPMENT MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Continuous Patient Monitors are advanced monitoring systems designed to provide real-time measurement of multiple vital signs, including CO2 levels, heart rate, SpO2, blood pressure, and respiratory rate across critical, surgical, and general care settings | Enable continuous assessment of patient status, support early detection of respiratory or cardiac distress, and improve patient safety and clinical decision-making |
 |
Capnography Systems are specialized devices that measure and display the concentration or partial pressure of exhaled CO2 to assess ventilation and airway integrity during anesthesia, sedation, and critical care | Ensure accurate monitoring of respiratory function, enable early detection of hypoventilation or airway obstruction, and enhance patient safety during surgical and emergency procedures |
 |
Airway Adapter which are disposable or reusable connectors that link the patient’s breathing circuit to the capnography sensor for CO2 measurement during ventilation | Provide a secure and accurate interface for gas sampling, minimize leakage, and ensure reliable CO2 readings for both intubated and non-intubated patients |
 |
Capnography Sensor are core component of capnography systems that detects and analyzes CO2 levels from exhaled breath using infrared spectroscopy or similar technologies | Deliver precise, real-time CO2 readings, improve diagnostic accuracy, and enhance the effectiveness of patient monitoring in both mainstream and sidestream configurations |
 |
Integrated software that processes and visualizes CO2 waveform data, enabling clinicians to monitor trends, set alerts, and analyze ventilation performance remotely or locally | Enhance clinical workflow efficiency, enable data-driven decisions through trend analysis, and support interoperability with hospital information systems for better patient management |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The North America capnography equipment market is composed of a closely integrated network of manufacturers, distributors, end users, and regulatory authorities, all working together to promote market growth and acceptance. Leading manufacturers, such as Philips, Medtronic, GE Healthcare, Masimo, Dräger, BD, Nihon Kohden, ResMed, and ICU Medical, are innovating in capnography devices and accessories. Major distributors, including US Med, Cardinal Health, McKesson, and Medline, offer these products in their portfolios. The end users are diverse, ranging from large hospital systems and specialty clinics to pediatric and diabetes care centers. Notable institutions include Advent Health, Mayo Clinic, Jackson Health System, Johns Hopkins Medicine, and Mary Bridge Children's Hospital. Regulatory oversight by agencies such as the US FDA and Health Canada ensures safety, compliance, and quality standards, fostering a trustworthy, efficient market ecosystem that supports improved patient monitoring, safety, and clinical outcomes across North America.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
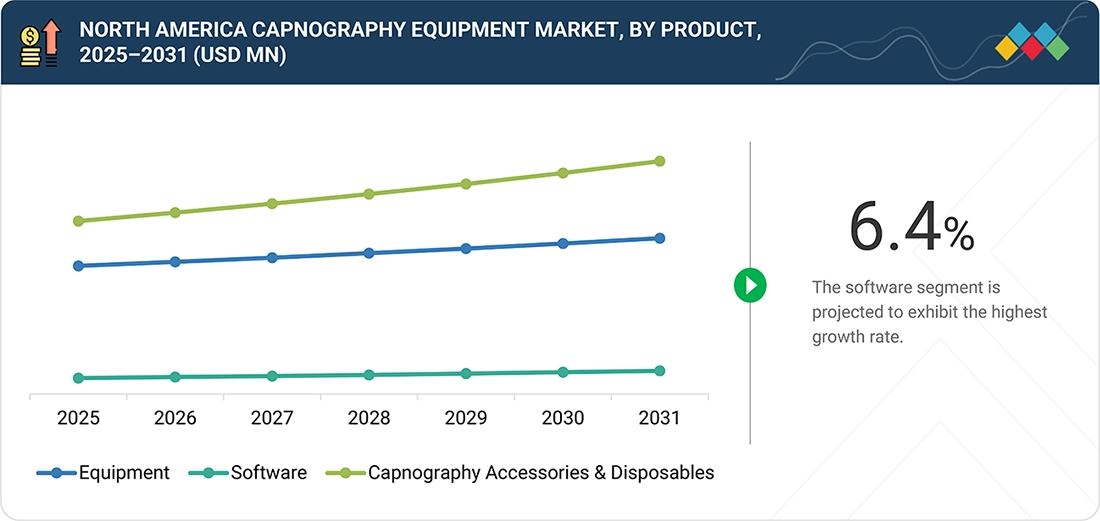
North America Capnography Equipment Market, by Product
In the product category, capnography accessories and disposables represent the largest share of the North America capnography equipment market. This is mainly due to their frequent and repeated use in hospitals, ambulatory surgical centers, and emergency care settings. Products such as nasal cannulas, sampling lines, adapters, and airway connectors are essential for most capnography applications and must be changed regularly to maintain hygiene standards, ensure measurement accuracy, and comply with clinical protocols. This creates a steady and predictable demand for these products. Additionally, the increasing number of surgeries, the rising use of procedural sedation, and the growing application of capnography for both intubated and non-intubated patients have led to higher consumption of these accessories. Furthermore, stringent infection control measures and a strong preference for single-use medical devices in North America healthcare facilities have favored disposable accessories over reusable ones, solidifying their leading position in this product segment.
North America Capnography Equipment Market, by Technology
In the technology segment, mainstream capnography accounted for the largest share of the North America capnography equipment market. This is primarily due to its superior accuracy in measuring, quick response time, and reliable detection of end-tidal carbon dioxide at the airway. It is most commonly used in operating rooms and intensive care units where intubated patients require continuous, real-time respiratory monitoring for effective anesthesia delivery and ventilator management. The widespread use of mainstream capnography in high-acuity clinical settings across the US and Canada is supported by clinical standards and hospital protocols that emphasize the critical need for precise ventilation monitoring. Additionally, the region benefits from an advanced healthcare infrastructure, alongside extensive installations of modern anesthesia systems and multiparameter patient monitors that are compatible with mainstream capnography, which helps this technology maintain its leading position. Ongoing technological advancements, such as lighter sensor designs, reduced dead space, and improved moisture handling, have increased usability while enhancing patient safety. Together with a growing number of surgical and critical care procedures, these advantages have established mainstream capnography as the dominant technology segment in the North America market.
North America Capnography Equipment Market, by Application
Cardiac care is the largest revenue contributor in the North America capnography equipment industry among various application segments. This is primarily due to the high volume of cardiac surgeries, interventional cardiology procedures, and critical care cases that require continuous respiratory and hemodynamic monitoring. Capnography is a valuable tool in cardiac care because it can detect hypoventilation, airway obstruction, and changes in cardiac output at an early stage during anesthesia, sedation, and mechanical ventilation. This makes it an essential monitoring tool in catheterization labs, operating rooms, and cardiac intensive care units. The high prevalence of cardiovascular diseases in the US and Canada, along with an increasing aging population, has led to a rising demand for advanced respiratory monitoring in cardiac settings. Furthermore, there is greater clinical awareness of the relationship between end-tidal CO2 levels and circulatory status, leading to the routine use of capnography for both procedural and postoperative monitoring of cardiac patients. The availability of well-equipped cardiac centers, attractive reimbursement policies, and stringent safety measures for high-risk cardiac interventions have all contributed to a higher adoption rate of capnography. Together, these factors solidify cardiac care as the largest application segment in the North America capnography equipment market.
North America Capnography Equipment Market, by End User
Hospitals are the primary users in the North America capnography equipment market. This dominance is largely due to their high patient volumes, the diverse range of surgical and diagnostic procedures they perform, and their extensive use of anesthesia and mechanical ventilation. Hospitals are also the main sites for complex surgeries, emergency care, and intensive care treatment, where continuous respiratory monitoring is essential for patient safety. Consequently, capnography is widely used in operating rooms, intensive care units, emergency departments, and post-anesthesia care units, driving strong, consistent demand for both standalone and integrated capnography systems. Additionally, North America hospitals are technologically advanced, featuring superior healthcare facilities and a higher adoption rate of multi-parameter patient monitors that include capnography. Stringent regulatory guidelines and clinical protocols also require the use of capnography during sedation and ventilation, further supporting its widespread use in hospital settings. The availability of trained healthcare professionals, sufficient procurement budgets, and the need to care for high-risk and critically ill patients further contribute to the fact that hospitals are the dominant segment in the North America capnography equipment market.
REGION
US accounted for largest share of North America capnography equipment market in 2025
The North America capnography equipment market is primarily driven by the US, influenced by several key factors. These include a high volume of surgical interventions, extensive use of anesthesia and procedural sedation, and a strong adoption of innovative patient monitoring technologies in healthcare facilities. In US hospitals and ambulatory surgical centers, it is now standard practice to use capnography to ensure patient safety during procedures such as intubation, mechanical ventilation, and moderate to deep sedation. As a result, there is a strong market for both standalone and integrated capnography devices. Additionally, the large population of individuals in the US with chronic respiratory and cardiovascular diseases contributes to the increasing demand for continuous CO2 monitoring in critical care and emergency settings. Strict clinical guidelines and regulations from organizations such as the American Society of Anesthesiologists (ASA) and other professional societies have also played a key role in promoting the regular use of capnography during anesthesia and sedation procedures. The US continues to lead the North America capnography equipment market due to the presence of major manufacturers, well-structured reimbursement systems, high healthcare expenditure, and the rapid adoption of advanced monitoring solutions.

NORTH AMERICA CAPNOGRAPHY EQUIPMENT MARKET: COMPANY EVALUATION MATRIX
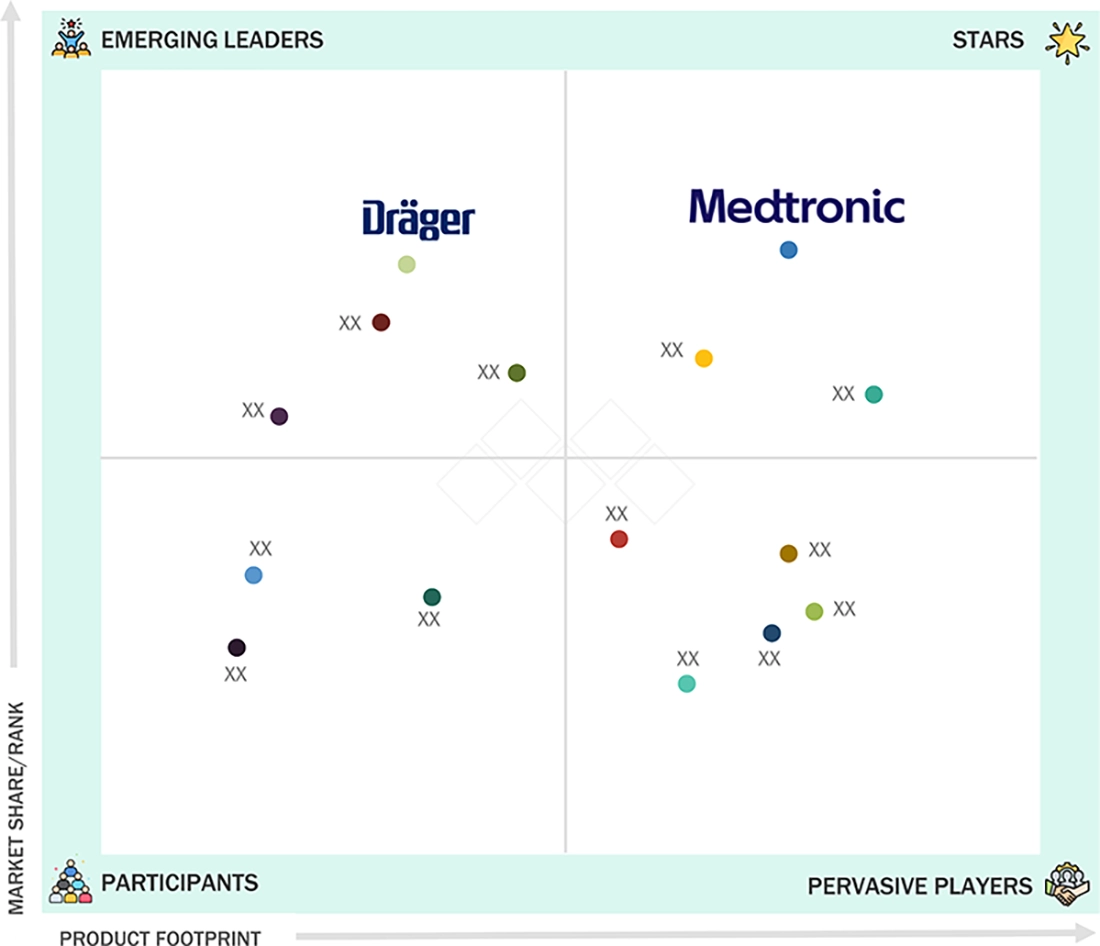
In the North America capnography equipment market matrix, Medtronic (Star) leads with scale, extensive distribution, and a comprehensive portfolio of monitoring solutions. Dräger (Emerging Leader) is gaining momentum by offering innovative capnography devices that enhance clinical accuracy, improve patient safety, and support efficient respiratory monitoring. While Medtronic dominates through its regional reach and established presence, Dräger’s focus on technological innovation positions it for rapid growth toward the leaders quadrant.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Becton, Dickinson and Company (US)
- Medtronic (Ireland)
- Koninklijke Philips N.V. (Netherlands)
- GE HealthCare (US)
- Drägerwerk AG & Co. KGaA (Germany)
- Nihon Kohden Corporation (Japan)
- Masimo (US)
- ICU Medical, Inc. (US)
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (China)
- ZOLL Medical Corporation (US)
- EDAN Instruments, Inc. (China)
- Hamilton Medical (Switzerland)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) | USD 0.22 Billion |
| Revenue Forecast in 2031 (Value) | USD 0.29 Billion |
| Growth Rate | CAGR of 4.5% from 2026–2031 |
| Years Considered | 2025–2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | Value (USD Million), Volume (Thousand Unit) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Regional Scope | North America (US and Canada) |
| Parent & Related Segment Reports |
Capnography Equipment Market Asia Pacific Capnography Equipment Market Europe Capnography Equipment Market |
WHAT IS IN IT FOR YOU: NORTH AMERICA CAPNOGRAPHY EQUIPMENT MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Volume Analysis | Market assessment by volume (units) for products |
|
| Company Information |
|
Insights on revenue shifts toward emerging innovations |
| Disease Prevalence |
|
|
RECENT DEVELOPMENTS
- September 2025 : Masimo’s renewed and expanded strategic partnership with Philips emphasizes accelerated integration of Masimo’s monitoring technologies, including NomoLine capnography sensors, into a wider range of Philips multi-parameter patient monitors and next-generation wearable platforms. The collaboration also plans market-led development of AI-enhanced monitoring tools, which could amplify innovation in capnography and other respiratory monitoring within North America hospitals and outpatient settings.
- June 2025 : Philips and Medtronic have deepened their long-standing collaboration with a new multi-year agreement to expand access to advanced patient monitoring technologies, starting in the North America market. Under this deal, Philips will integrate Medtronic’s core monitoring solutions, including Microstream capnography alongside pulse oximetry and other vital sign technologies into Philips’ IntelliVue portfolio and bundle related consumables to simplify procurement for US and Canadian hospitals and care providers.
Table of Contents

Methodology
This study involved four major activities in estimating the current North America capnography equipment market size. First, extensive secondary research was conducted to gather information on the market, including related and parent markets. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. After that, market breakdown and data triangulation were used to estimate the market size of segments and subsegments.
Secondary Research
The secondary research process involved the widespread use of secondary sources, directories, databases (such as Bloomberg Businessweek, Factiva, and D&B Hoovers), white papers, annual reports, company house documents, investor presentations, and SEC filings of companies. Secondary research was used to identify and collect information useful for the extensive, technical, market-oriented, and commercial study of the North America capnography equipment market. It was also used to obtain important information about the key players and market classification and segmentation according to industry trends to the bottom-most level, and key developments related to market and technology perspectives. A database of the key industry leaders was also prepared using secondary research.
Primary Research
In the primary research process, various sources from both the supply and demand sides were interviewed to obtain qualitative and quantitative information for this report. The primary sources from the supply side include industry experts such as CEOs, vice presidents, marketing and sales directors, technology & innovation directors, and other key executives from various companies and organizations operating in the North America capnography equipment market. The primary sources from the demand side included industry experts, purchase & sales managers, doctors, and personnel from research organizations. Primary research was conducted to validate the market segmentation, identify key players in the market, and gather insights on key industry trends and key market dynamics.
A breakdown of the primary respondents for the North America capnography equipment market is provided below:
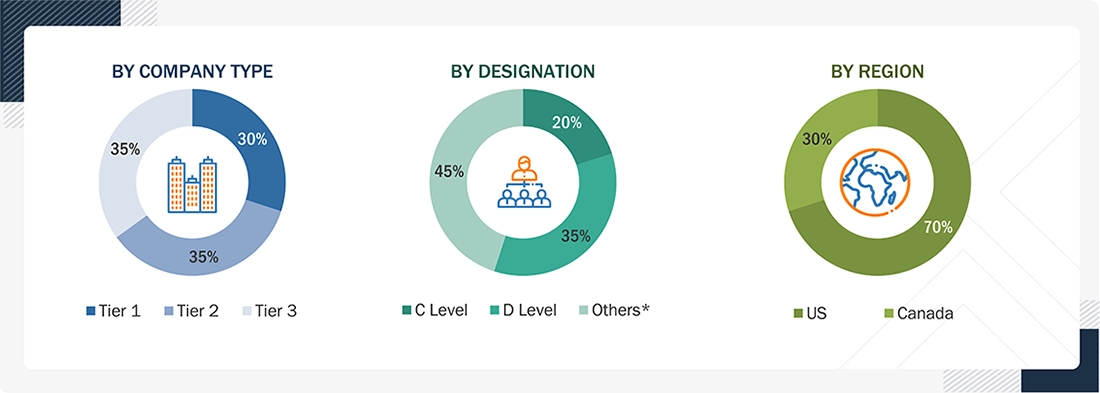
Note 1: C-level primaries include CEOs, CFOs, COOs, and VPs.
Note 2: Other designations include sales managers, marketing managers, business development managers, product managers, distributors, and suppliers.
Note 3: Companies are classified into tiers based on their total revenue. As of 2024, Tier 1 = >USD 10.00 billion, Tier 2 = USD 1.00 billion to USD 10.00 billion, and Tier 3 = <USD 1.00 billion.
Source: MarketsandMarkets Analysis
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
For the regional market value, annual revenues were calculated based on the revenue mapping of major product manufacturers and OEMs active in the North America capnography equipment market. All the major product manufacturers were identified at the country/regional level. Revenue mapping for the respective business segments/subsegments was done for the major players. Also, the North America capnography equipment market was split into various segments and subsegments based on:
- List of major players operating in the products market at the regional and/or country level
- Product mapping of various North America capnography equipment manufacturers at the regional and/or country level
- Mapping of annual revenue generated by listed major players from North America capnography equipment (or the nearest reported business unit/product category)
- Revenue mapping of major players to be covered
- Extrapolation of the revenue mapping of the listed major players to derive the regional market value of the respective segments/subsegments
- Summation of the market value of all segments/subsegments to arrive at the regional capnography equipment market
The above-mentioned data was consolidated and added with detailed inputs and analysis from MarketsandMarkets and presented in this report.
North America Capnography Equipment Market : Top-Down and Bottom-Up Approach

Data Triangulation
After determining the overall size of the North America capnography equipment market using the above-mentioned methodology, the market was split into several segments and subsegments. The data triangulation and market breakdown procedures were employed, wherever applicable, to complete the overall market engineering process and arrive at the exact market value data for the key segments and subsegments. The extrapolated market data was triangulated by studying various macroindicators and regional trends from both demand- and supply-side participants.
Market Definition
Capnography equipment focuses on the manufacture of devices that measure the concentration of carbon dioxide in respiratory gases to accurately assess a patient’s respiratory state. These are important in emergency medicine, anesthesia, and critical care settings, as they monitor ventilation and detect potential complications related to respiratory disorders. With technological advancements and increasing awareness of the need to monitor respiration, market growth is expected to be driven by rising demand for non-invasive monitoring solutions, regulatory approvals for innovative products, and a growing patient population requiring respiratory care.
Key Stakeholders
- Manufacturers
- Clinical Research Laboratories
- Healthcare Providers
- Market Share and Consulting Firms
- Medical Device Suppliers, Distributors, Channel Partners, and Third-party Suppliers
- Clinicians and Healthcare Professionals
- Global and National Health Agencies
- Academic Medical Centers and Universities
- Business Research and Consulting Service Providers
- Venture Capitalists and Other Government Funding Organizations
Report Objectives
- To define, describe, and forecast the North America capnography equipment market by product, technology, application, end user, and country
- To provide detailed information regarding the major factors influencing the market growth (such as drivers, restraints, opportunities, and challenges)
- To strategically analyze the micromarkets with respect to individual growth trends, prospects, and contributions to the total market
- To analyze the opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders
- To profile the key market players and comprehensively analyze their market shares and core competencies
- To track and analyze competitive developments, such as product launches & approvals, agreements, partnerships, acquisitions, and collaborations, in the North America capnography equipment market
Available customizations:
- With the given market data, MarketsandMarkets offers customizations as per your company’s specific needs. The following customization options are available for the report:
Competitive Landscape Assesment
- Market share analysis, by region (North America), which provides market shares of the top 3-5 key players in the North America capnography equipment market
- Competitive leadership mapping for established players in the US
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the North America Capnog
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS


















Growth opportunities and latent adjacency in North America Capnog