
North America Blood Glucose Monitoring Devices Market Size, Growth, Share & Trends Analysis
North America Blood Glucose Monitoring Devices Market by Product Type (Self-monitoring, Professional Point of Care), Application (Diabetes Management, Health & Wellness Monitoring), Test Site, End User (Self/Homecare) - Forecast to 2030




OVERVIEW
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
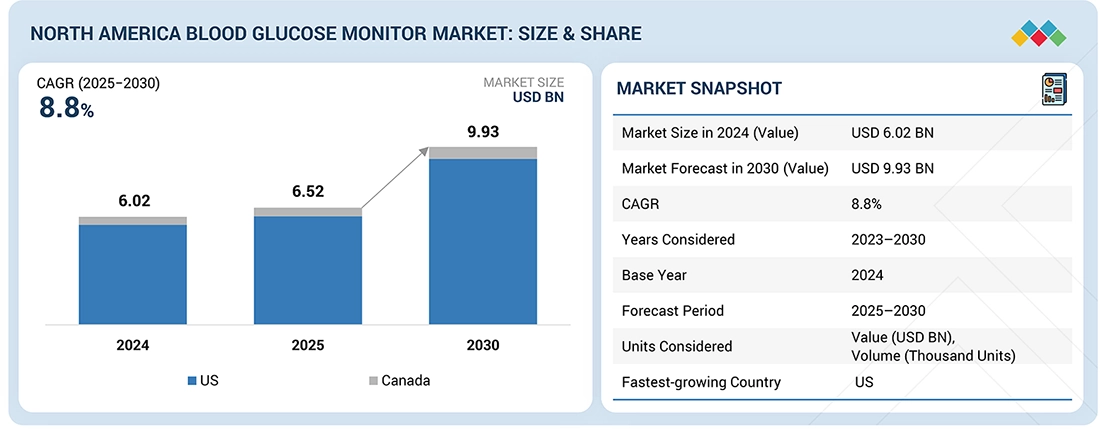
The North America Blood Glucose Monitoring Devices market, valued at USD 6.02 billion in 2024, stood at USD 6.52 billion in 2025 and is projected to advance at a resilient CAGR of 8.8% from 2025 to 2030, culminating in a forecasted valuation of USD 9.93 billion by the end of the period. Advances in glucose monitoring technologies, including continuous glucose monitors (CGMs) and smart self-monitoring devices, offer greater accuracy, real-time data, and improved patient convenience compared with traditional methods. These innovations support better glycemic management, reduce complications, and enhance overall diabetes care outcomes. Rising diabetes prevalence, increasing health awareness, and growing adoption of digital health solutions are driving demand for blood glucose monitoring devices.
KEY TAKEAWAYS
-
BY COUNTRYBy country, the US accounted for the largest share (92.5%) of the North American blood glucose monitoring devices market in 2024.
-
BY PRODUCT TYPEBy product type, the self-monitoring blood glucose systems segment held the largest share of 41.8% in the North America blood glucose monitoring devices market in 2024.
-
BY APPLICATIONBased on application, the diabetes management segment is expected to grow at the highest CAGR of 8.9% during the forecast period.
-
BY TEST SITEBased on test site, the fingertip segment contributed the largest share because of its convenience, fast results, and extensive usage among patients and healthcare professionals.
-
BY END USERBased on end user, the self/home care segment accounted for the largest share in the market.
-
COMPETITIVE LANDSCAPE- KEY PLAYERSAbbott Laboratories (US), DexCom, Inc. (US), and Medtronic (Ireland) were identified as some of the key players in the North America blood glucose monitoring devices market, given their substantial market share and product/service footprint.
-
COMPETITIVE LANDSCAPE- STARTUPSCompanies such as LifePlus and Biolinq Incorporated, among others, have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas, underscoring their potential as emerging market leaders.
The North America blood glucose monitoring devices market is experiencing steady growth, driven by several key factors. Rising diabetes prevalence, fueled by aging populations, urbanization, and lifestyle changes, is increasing demand for effective self-monitoring solutions. In addition, growing adoption of advanced continuous and non-invasive glucose monitoring technologies, along with innovations in connectivity, mobile health integration, and smart analytics, is broadening their clinical and home-based use.
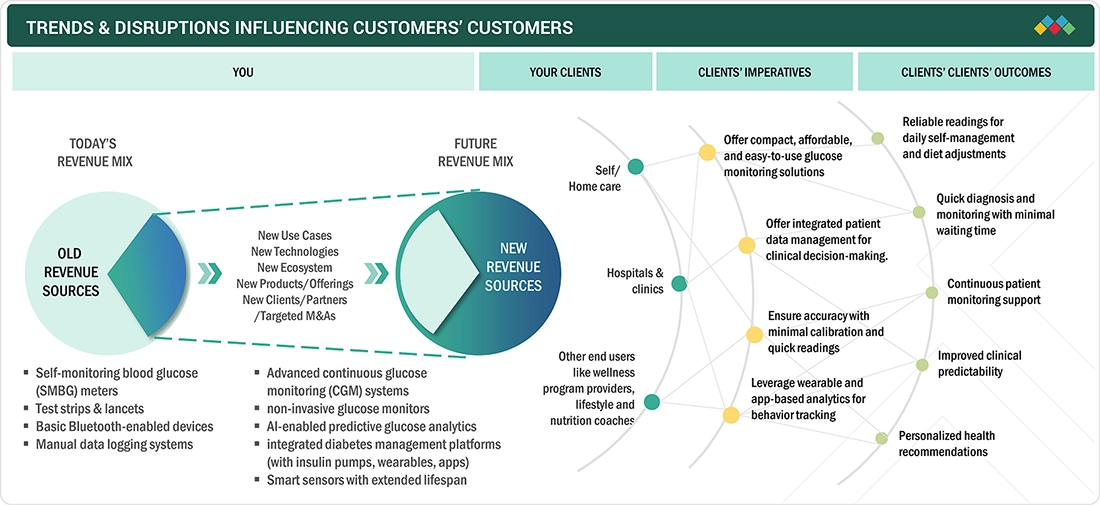
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The blood glucose monitoring devices market is shifting from traditional glucose meters and strip-based testing toward advanced, technology-driven solutions. Emerging trends include continuous glucose monitoring (CGM) systems, non-invasive and minimally invasive sensors, smart connected devices, and AI-powered analytics. These innovations are creating high-growth opportunities, driven by demand for real-time monitoring, improved accuracy, and enhanced diabetes management. End users, including hospitals, clinics, and home-care patients, are increasingly adopting these disruptive technologies, reshaping the future of diabetes care.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Increasing prevalence of diabetes

-
Growing elderly population and increasing life expectancy
Level
-
Short sensor lifespan and frequent replacements
Level
-
Integration with digital health platforms and smart devices
-
Expansion of coverage and reimbursement policies for CGMs
Level
-
Regulatory compliance challenges for non-invasive glucose monitoring technologies
-
Data overload & alarm fatigue
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Increasing prevalence of diabetes
The prevalence of diabetes in North America continues to rise, significantly driving demand for blood glucose monitoring devices. According to the International Diabetes Federation (IDF) 11th edition, released in 2025, approximately 38.5 million individuals in the US were living with diabetes in 2024, creating substantial demand for accurate and convenient glucose monitoring solutions. The IDF further indicates that diabetes prevalence across North America remains among the highest globally, reflecting a large and steadily expanding patient population requiring long-term disease management. A notable proportion of affected individuals includes patients with type 1 diabetes, including children and adolescents under 20 years of age, reinforcing the need for continuous, lifelong glucose monitoring. With diabetes representing a significant public health burden across the region, the condition continues to place sustained pressure on healthcare systems. The escalating prevalence creates sustained demand for reliable, user-friendly, and technologically advanced blood glucose monitoring solutions to enable early detection, continuous management, and improved patient outcomes.
Restraint: Short sensor lifespan and frequent replacements
The short lifespan of blood glucose monitoring sensors and the need for frequent replacements have been identified as a major limitation of the North America blood glucose monitoring devices market. Most continuous glucose monitoring (CGM) sensors are intended for short-term use, such as days or weeks, which leads to higher costs for patients and payers over time. Despite strong insurance coverage in the region, repeated sensor replacements have led to out-of-pocket expenses for users of these monitoring devices, particularly for uninsured or underinsured populations. Besides, frequent sensor changes can cause inconvenience and challenge adherence to the treatment plan for users, particularly for elderly patients and those with long-term diabetes. In addition, supply continuity, prescription renewals, and reimbursement limitations on replacement frequency can hinder consistent device use. Altogether, these factors limit the market penetration of blood glucose monitoring systems and hamper their overall growth potential in North America.
Opportunity: Integration with digital health platforms and smart devices
Integrating blood glucose monitors with digital health platforms and smart devices is emerging as a transformative opportunity in diabetes management. Advances in Bluetooth and cloud connectivity enable real-time glucose data to sync with smartphones, wearables, and electronic health records, enabling continuous monitoring and data-driven decision-making. Patients benefit from personalized alerts, automated insulin-dose recommendations, and trend analytics that improve self-management and clinical outcomes. For healthcare providers, integrated platforms enable remote monitoring, teleconsultations, and population-level analytics, enhancing care coordination and reducing hospital visits. As governments and insurers increasingly support digital health adoption, this trend is expected to accelerate, positioning connected glucose monitoring as a cornerstone of next-generation diabetes care.
Challenge: Regulatory compliance challenges for non-invasive glucose monitoring technologies
Non-invasive glucose monitoring technologies face significant regulatory challenges as authorities such as the FDA require rigorous proof of safety, accuracy, and consistency across diverse populations and conditions. Unlike conventional blood-based systems, these devices rely on optical or electromagnetic sensing, which can be affected by skin tone, hydration, and environmental factors, complicating validation. The absence of standardized testing protocols increases uncertainty, leading to lengthy approval timelines and costly clinical studies. Consequently, despite strong innovation potential, regulatory and reimbursement barriers continue to delay the large-scale commercialization of non-invasive glucose monitoring solutions.
NORTH AMERICA BLOOD GLUCOSE MONITORING DEVICES MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
The FreeStyle Libre 3 continuous glucose monitoring (CGM) system is designed for real-time glucose tracking without routine fingersticks; it transmits minute-by-minute readings to a connected smartphone app. | Eliminates fingerstick calibration, enhances patient comfort, enables proactive glucose management, and supports better glycemic control through trend-based insights |
 |
Dexcom's G7 all-in-one CGM sensor provides real-time glucose readings, customizable alerts, and integration with smartphones and insulin pumps for seamless diabetes management. | Offers superior accuracy and fast warm-up; provides predictive alerts to prevent hypo- and hyperglycemia; improves adherence through connectivity and data visualization |
 |
The Guardian 3 Sensor is used with Medtronic insulin pumps to deliver continuous glucose data every five minutes, supporting automated insulin delivery and trend-based therapy adjustments. | Enables hybrid closed-loop insulin management, improves time-in-range, reduces glycemic variability, and enhances safety for insulin-dependent users |
 |
The Omnitest self-monitoring blood glucose (SMBG) device is designed for capillary blood testing, offering reliable and quick results for daily home or point-of-care glucose monitoring. | Provides affordable, accurate, and easy-to-use glucose testing; supports frequent monitoring and improved patient engagement in self-managed care |
 |
The Accu-Chek range of SMBG systems is designed for fast and dependable glucose measurement with connectivity to digital platforms for progress tracking and data sharing. | Ensures consistent accuracy, simplifies diabetes management, enables remote data access, and strengthens patient–clinician communication for improved outcomes |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The blood glucose monitoring devices market operates within a complex ecosystem of stakeholders that collectively drive innovation, distribution, and adoption. Device manufacturers develop accurate, reliable blood glucose monitoring systems, while distributors ensure these products reach hospitals, clinics, pharmacies, and retail channels. Healthcare providers, including hospitals and specialized diabetes care centers, integrate these devices into patient management, with doctors, nurses, and other professionals guiding use and monitoring outcomes. Patients and caregivers form the end-user base, relying on these devices for daily diabetes management. Health advocacy organizations promote awareness, best practices, and policy support, while government and regulatory bodies ensure safety, efficacy, and compliance, shaping market access and product standards. Together, this interconnected network defines the operational and innovation landscape of the global blood glucose monitoring devices market.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
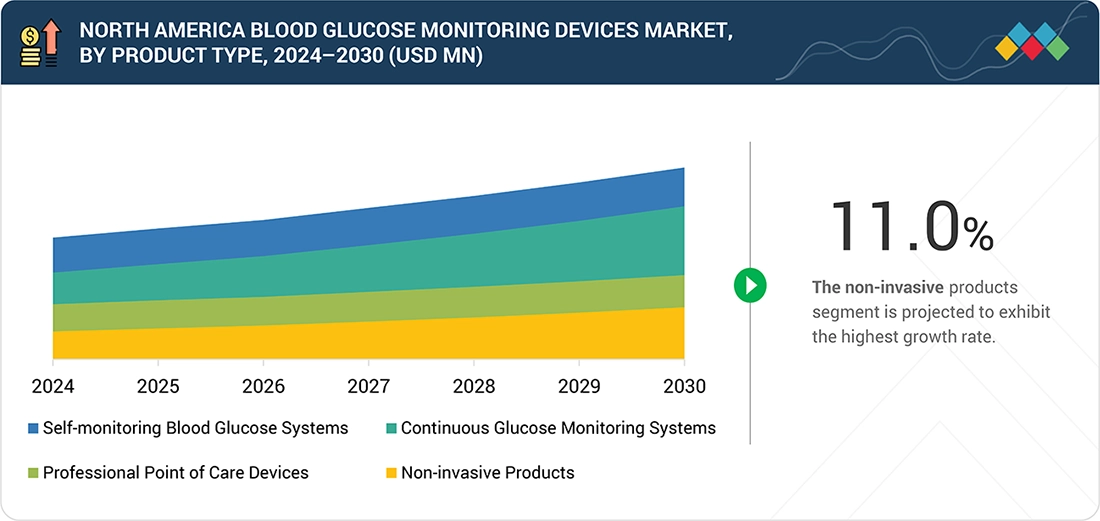
North America Blood Glucose Monitoring Devices Market, By Product Type
By product, the North America blood glucose monitoring devices market is categorized into four primary segments: self-monitoring blood glucose systems, continuous glucose monitoring (CGM) systems, professional point-of-care devices, and noninvasive products. Among these, CGM systems are expected to grow fastest during the forecast period, driven by rapid technological advancements and rising demand for real-time glucose tracking. CGM devices provide continuous, automated glucose readings without multiple finger-prick tests, enabling better glycemic control and early detection of fluctuations. Their integration with digital health platforms, insulin pumps, and smartphone applications further enhances patient convenience and treatment personalization. Additionally, regulatory approvals of next-generation sensors, such as Dexcom G7 and Abbott FreeStyle Libre 3, coupled with expanding reimbursement support and increasing adoption among people with type 1 and type 2 diabetes, are accelerating market expansion for CGM systems.
North America Blood Glucose Monitoring Devices Market, By Application
The North America blood glucose monitoring devices market is segmented by application into diabetes management, health and wellness monitoring, and other uses, with the diabetes management segment projected to grow fastest during the forecast period. Rising prevalence of type 1 and type 2 diabetes has driven demand for continuous, reliable glucose monitoring, with continuous glucose monitoring (CGM) systems providing real-time data to support optimized insulin therapy, dietary adjustments, and lifestyle modifications. Integration with digital health platforms enables seamless tracking, automated reminders, and enhanced collaboration with healthcare professionals, enabling personalized care and tailored treatment plans. Consistent monitoring facilitates early detection of glucose fluctuations, helping patients maintain optimal glycemic control and reduce the risk of severe complications. Furthermore, supportive government initiatives, public health programs, and insurance coverage that promote early diagnosis and effective diabetes management are expanding access, while user-friendly home monitoring devices empower patients to self-manage their condition, improving adherence and long-term health outcomes. The convergence of technology, policy support, and patient-centric solutions is expected to further accelerate adoption in this high-growth segment.
North America Blood Glucose Monitoring Devices Market, By End User
Based on end users, the North America blood glucose monitoring devices market is segmented into three categories: self/home care, hospitals and clinics, and other end users. Among these, the self/home care segment holds the largest share, driven by a growing preference for convenient in-home diabetes management, rising awareness of the importance of frequent glucose monitoring, and an increasing number of individuals managing diabetes independently at home. The widespread availability of user-friendly devices, coupled with easy access and affordability, has further accelerated adoption. Technological advancements, including smart meters, continuous glucose monitoring systems, and seamless smartphone integration, have enhanced device usability and engagement, positioning home monitoring solutions as the primary growth engine for the market. Additionally, the shift toward proactive health management and telemedicine adoption has reinforced the dominance of the self-care segment, underscoring its critical role in shaping market expansion.
REGION
US to be fastest-growing country in North America blood glucose monitoring devices market during forecast period
The blood glucose monitoring devices market in the US is expected to register the highest CAGR in North America during the forecast period. Market growth is being driven by factors such as the rising incidence of diabetes, greater awareness of the importance of regular blood glucose monitoring, and the growing use of continuous, connected, and digital glucose monitoring technologies. The country's large and aging population, combined with the high prevalence of lifestyle-related diseases, is increasing the need for accurate, convenient, and patient-friendly monitoring solutions. Moreover, robust health care spending, extensive insurance coverage, and the rapid adoption of digital health platforms, mobile applications, and remote patient monitoring systems are expected to boost the US blood glucose monitoring devices market.
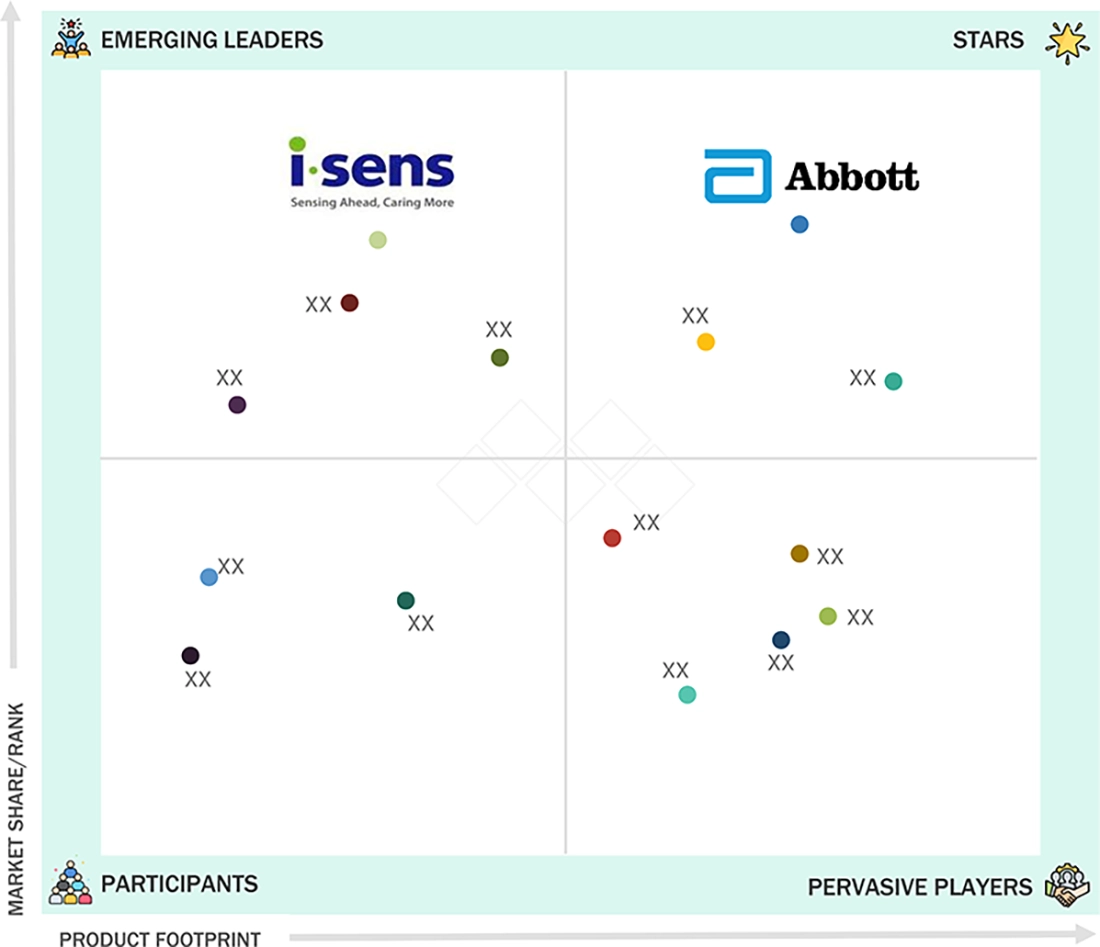
NORTH AMERICA BLOOD GLUCOSE MONITORING DEVICES MARKET: COMPANY EVALUATION MATRIX
In the North America blood glucose monitoring devices market matrix, Abbott Laboratories (Star) maintains a dominant position, driven by its strong regional footprint and comprehensive portfolio spanning continuous glucose monitoring (CGM) and self-monitoring blood glucose (SMBG) systems. Its leadership is supported by robust R&D capabilities, clinically proven accuracy, and advanced digital integration via the widely adopted FreeStyle Libre platform in homecare and clinical settings. i-SENS, Inc. (Emerging Leader) is gradually strengthening its presence in the region with accurate, cost-effective SMBG devices and early-stage expansion into CGM through its CareSens Air system. While Abbott continues to benefit from scale, innovation, and strong brand trust among providers and payers, i-SENS shows potential to progress toward the leaders’ quadrant by leveraging its manufacturing expertise, expanding distribution partnerships, and focusing on connected, value-driven glucose monitoring solutions for price-sensitive segments in North America.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- Abbott Laboratories (US)
- Medtronic (Ireland),
- Dexcom (US)
- Sinocare (China)
- F. Hoffmann-La Roche Ltd (Switzerland)
- B Braun SE (Germany)
- Nipro (Japan)
- Senseonics (US)
- i-Sens, Inc (South Korea)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size Value in 2024 (Value) | USD 6.02 Billion |
| Market Forecast in 2030 (Value) | USD 9.93 Billion |
| Growth Rate | CAGR of 8.8% from 2025–2030 |
| Years Considered | 2023–2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD Million/Billion), Volume (Thousands Units) |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segments Covered | By Product Type: Self-monitoring Blood Glucose Systems, Continuous Glucose Monitoring Systems, Professional Point of Care devices, Non-invasive Products I Test Site: Fingertip, Upper Arm, Alternate Sites I By Application: Diabetes Management, Health & Wellness Monitoring, Other Applications I End User: Self/Home Care, Hospitals & Clinics, Other End Users |
| Countries Covered | US and Canada |
| Parent & Related Segment Reports |
Blood Glucose Monitoring Devices Market Europe Blood Glucose Monitor Market LATAM Blood Glucose Monitor Market APAC Blood Glucose Monitoring Devices Market |
WHAT IS IN IT FOR YOU: NORTH AMERICA BLOOD GLUCOSE MONITORING DEVICES MARKET REPORT CONTENT GUIDE

DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Volume Analysis | Market assessment by volume (units) for products such as self-monitoring blood glucose monitoring systems and continuous glucose monitoring systems |
|
| Company Information | Key players: Abbott Laboratories (US), DexCom, Inc. (US), Medtronic (Ireland), F. Hoffmann-La Roche Ltd. (Switzerland), B. Braun SE (Germany), and Ascensia Diabetes Care Holdings AG (Switzerland) - Top 3-5 players market share analysis at Asia Pacific and European country level | Insights on revenue shifts toward emerging innovations |
| Disease Prevalence |
|
|
RECENT DEVELOPMENTS
- April 2025 : DexCom, Inc. (US) announced that the US FDA has approved the DexCom G7 15-Day Continuous Glucose Monitoring System for use by people with diabetes over the age of 18.
- June 2024 : Abbott Laboratories (US) announced US FDA clearance for two new over-the-counter continuous glucose monitoring (CGM) systems: Lingo and Libre Rio, both based on Abbott's world-leading FreeStyle Libre continuous glucose monitoring technology.
- August 2024 : Medtronic received US FDA approval for the Simplera continuous glucose monitor (CGM). This is the company’s first disposable, all-in-one CGM, and is half the size of previous Medtronic CGMs.
- April 2025 : F. Hoffmann-La Roche Ltd announced the establishment of a new manufacturing facility for continuous glucose monitoring (CGM) in Indiana, US.
Table of Contents

Methodology
The study comprised four key activities to estimate the current size of the North America blood glucose monitoring devices market. First, extensive secondary research was conducted to gather information on the market, including related and parent markets. The next step involved validating these findings, assumptions, and market size estimates through primary research with industry experts across the value chain. Top-down and bottom-up approaches were used to arrive at a comprehensive estimate of the overall market size. Finally, market breakdown and data triangulation techniques were utilized to determine the sizes of segments and subsegments within the market.
Secondary Research
The secondary research process involved extensive use of various secondary sources, including directories, databases like Bloomberg Business, Factiva, and D&B Hoovers, as well as white papers, annual reports, company house documents, investor presentations, and SEC filings. This research was conducted to gather information valuable for a comprehensive, technical, market-oriented, and commercial study of the North America blood glucose monitoring devices market. It also helped obtain critical insights about key players in the industry, market classification, and segmentation based on current industry trends, down to the finest details. Additionally, a database of leading industry players was created through this secondary research.
Primary Research
In the primary research process, interviews were conducted with various sources from the supply and demand sides to gather qualitative and quantitative information for this report. On the supply side, industry experts were interviewed, including CEOs, vice presidents, marketing & sales directors, technology & innovation directors, and other key executives from prominent companies and organizations involved in the North America blood glucose monitoring devices market. For the demand side, interviews were conducted with industry experts, purchase and sales managers, doctors, and personnel from research organizations. This primary research was essential to validate market segmentation, identify key players in the industry, and gather insights on important industry trends and market dynamics.
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The research methodology used to estimate the size of the North America blood glucose monitoring devices market includes the following details. The market sizing was undertaken from the global side.
Country-level Analysis: The size of the North America blood glucose monitoring devices market was obtained from the annual presentations of leading players and secondary data available in the public domain. The share of products in the overall North America blood glucose monitoring devices market was obtained from secondary data and validated by primary participants to arrive at the total North America blood glucose monitoring devices market. Primary participants further validated the numbers.
Geographic Market Assessment (By Region and Country): The geographic assessment was done using the following approaches:
Approach 1: Geographic revenue contributions/splits of leading players in the market (wherever available) and respective growth trends
Approach 2: Geographic adoption trends for individual product segments by end users and growth prospects for each of the segments (assumptions and indicative estimates validated from primary interviews)
At each point, the assumptions and approaches were validated by industry experts who were contacted during primary research. Considering the limitations of data available from secondary research, revenue estimates for individual companies (for the overall North America blood glucose monitoring devices market and geographic market assessment) were ascertained based on a detailed analysis of their respective product offerings, geographic reach/strength (direct or through distributors or suppliers), and the shares of the leading players in a particular region or country.
North America Blood Glucose Monitoring Devices Market Size: Bottom-up Approach and Top-down Approach

Data Triangulation
After arriving at the overall size from the market size estimation process explained above, the total market was split into several segments and subsegments. The data triangulation and market breakdown procedures explained below were implemented, wherever applicable, to complete the overall market engineering process and arrive at the exact statistics for various market segments and subsegments. The data was triangulated by studying various factors and trends from the demand and supply sides. Along with this, the market size was validated using both the top-down and bottom-up approaches.
Market Definition
Blood glucose monitoring devices are medical devices used to measure and monitor North America Blood Glucose (sugar) levels in individuals, primarily for the diagnosis and management of diabetes mellitus. These monitors help patients and healthcare professionals assess glycemic control, adjust treatment plans, and prevent diabetes-related complications.
Stakeholders
- North America Blood Glucose Monitor Manufacturers
- Contract Manufacturers
- Suppliers and Distributors of North America Blood Glucose Monitors
- Senior Management
- Finance Department
- Healthcare Services Providers (Hospitals and Public & Private Clinics)
- Academic & Research Institutes
- E-commerce and Digital Platforms
- Retail Pharmacies and Supermarkets
- Trade Associations and Industry Bodies
- Regulatory Bodies and Government Agencies
- Business Research and Consulting Service Providers
- Market Research and Consulting Firms
- Venture Capitalists and Investors
Report Objectives
- To define, describe, segment, analyze, and forecast the North America blood glucose monitoring devices market by product type, application, test site, end user, and region.
- To provide detailed information about the factors influencing market growth (drivers, restraints, opportunities, and challenges) in the North America blood glucose monitoring devices market
- To analyze micromarkets concerning individual growth trends, prospects, and contributions to the overall North America blood glucose monitoring devices market
- To analyze market opportunities in the North America blood glucose monitoring devices market for stakeholders and provide details of the competitive landscape for key players
- To profile the key players in the North America blood glucose monitoring devices market and comprehensively analyze their core competencies
- To track and analyze competitive developments such as agreements, collaborations, and partnerships; acquisitions; and product approvals & launches in the North America blood glucose monitoring devices market
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the North America Blood Glucose Monitoring Devices Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS




















Growth opportunities and latent adjacency in North America Blood Glucose Monitoring Devices Market