
Digital Biomarkers Market
Digital Biomarkers Market by Type (Physiological, Vocal, Idiosyncratic), Therapy [Cardio, Onco (Lung, Breast Cancer), Mental Health, Neuro], Application [Clinical Research (Phase IV)], End User (Pharma & Biotech Companies, CROs) � Global Forecast to 2031




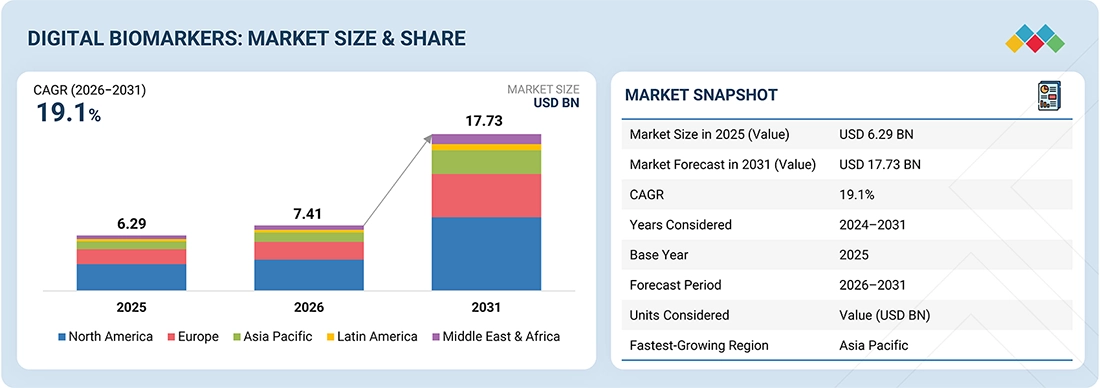
DIGITAL BIOMARKERS MARKET SIZE, SHARE & GROWTH SNAPSHOT
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
The global digital biomarkers market is projected to grow from USD 7.41 billion in 2026 to USD 17.73 billion by 2031, at a CAGR of 19.1% during the forecast period. The market was valued at USD 6.29 billion in 2025. The trend is largely driven by the increasing use of digital biomarkers in clinical trials and remote patient monitoring, as well as the development of real-world evidence in the pharmaceutical and healthcare sectors. Additionally, advancements in AI and machine learning-based analytics, multimodal data fusion, and smartphone and wearable sensing technologies are enhancing the precision and clinical relevance of digital biomarkers. Furthermore, the shift toward decentralized clinical trials and a more open regulatory environment regarding digital endpoints is contributing to their growing adoption in the pharmaceutical industry.
KEY TAKEAWAYS
-
By RegionNorth America accounted for the largest share of 48.1% of the global digital biomarkers market in 2025.
-
By TypeBy type, the physiological biomarkers segment accounted for the largest share of 40.6% of the digital biomarkers market in 2025.
-
By Therapeutic AreaBy therapeutic area, the cardiovascular segment accounted for the largest share of the digital biomarkers market in 2025.
-
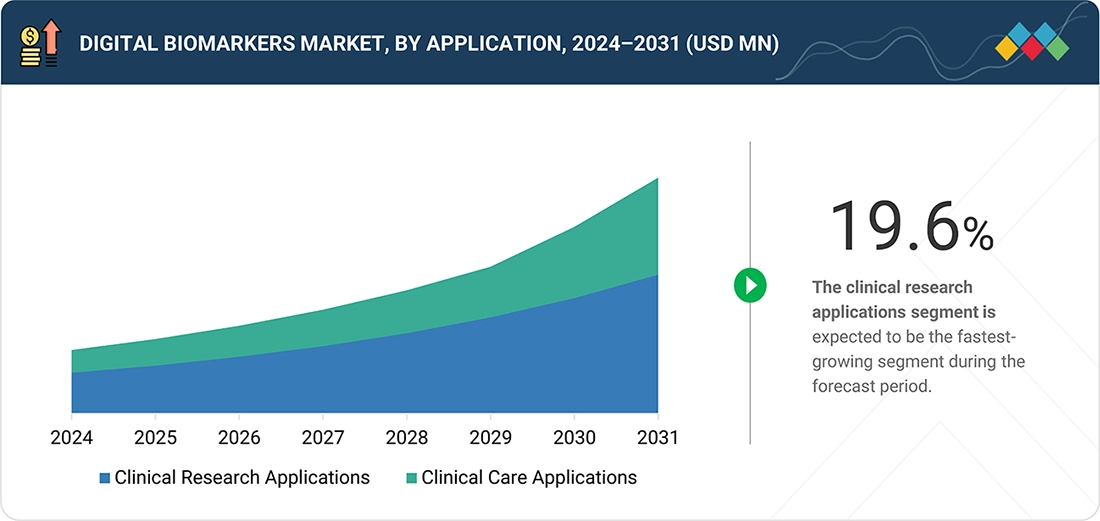
By ApplicationBy application, the clinical research applications segment is projected to register a growth rate of 19.6% in the global digital biomarkers market during the forecast period.
-
By End UserBy end user, the pharmaceutical and biotechnology companies segment accounted for the largest share of the digital biomarkers market in 2025.
-
Competitive Landscape - Key PlayersIXICO PLC, Ametris, LLC, Empatica, Inc. were identified as some of the star players in the digital biomarkers market (global), given their strong market share and product footprint.
-
Competitive Landscape - Startups/SMEsCumulus Neuroscience Limited and Delve Health have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas, underscoring their potential as emerging market leaders.
The digital biomarkers market is influenced by several factors. Key drivers include the growing trend of incorporating real-world data in clinical trials, the rise of AI-enabled digital endpoints, and the shift towards patient-centric care models. However, challenges such as the absence of standardized validation frameworks, regulatory uncertainties, and concerns regarding data privacy, interoperability, and clinical accuracy are impacting market growth.
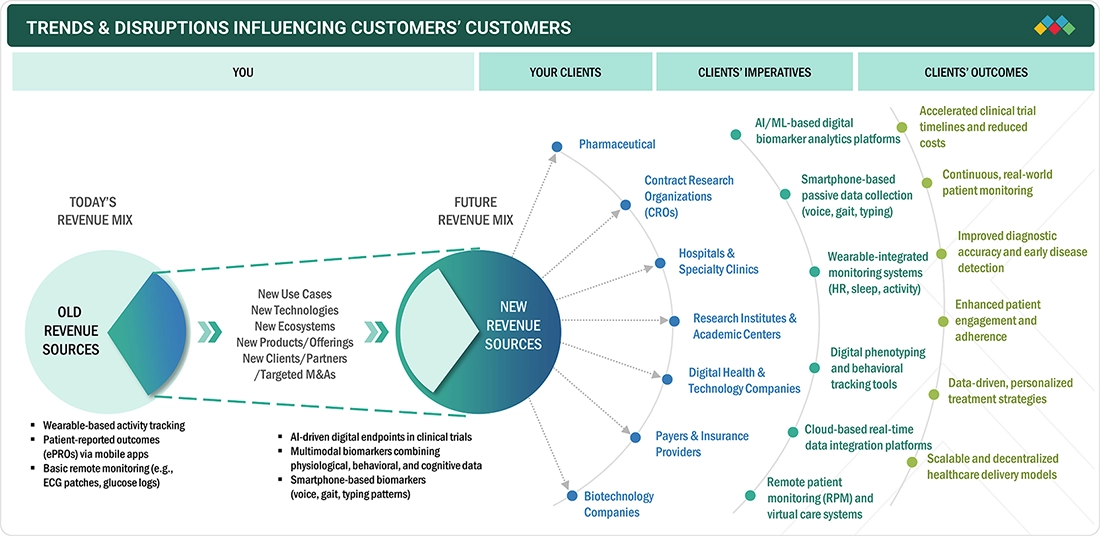
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The market for digital biomarkers is seeing a major shift with the focus on continuous and passive data collection through the use of daily devices such as smartphones. In addition, there is a focus on cognitive and behavioral digital biomarkers, such as voice, gait, sleep, and interaction. This is seeing the development of new opportunities in the field of digital biomarkers outside the realm of physiological monitoring. At the same time, there is a shift in the therapeutic areas where there is a focus on the use of digital biomarkers. This is seeing a shift in the way in which there is a focus on the management of various conditions in the real world. In this context, there is also a blurring of the lines between consumer and clinical-grade products. This is seeing the development of new opportunities in terms of scalability. This is seeing the development of new questions in terms of clinical validation. In this context, there is also a shift in the way in which perceptions of ownership and transparency of algorithms and devices are being developed.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Expansion of decentralized and remote clinical trials

-
Expansion of smartphones, wearables, and connected health ecosystems
Level
-
Data privacy and security concerns related to health data
-
Limited digital literacy and technology access among patients
Level
-
Increasing use of digital biomarkers in drug development and clinical trial endpoints
-
Growth of precision medicine and personalized healthcare
Level
-
Lack of standardized validation frameworks for digital biomarkers
-
Regulatory uncertainty around digital endpoints and biomarker validation
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Expansion of decentralized and remote clinical trials
The growth of decentralized and remote clinical trials is a major factor driving the expansion of the digital biomarkers market. This shift is moving clinical trials from a site-centric approach to a patient-centric approach. Digital biomarkers enable continuous and passive data collection at high frequencies using smartphones and connected devices, moving away from the traditional clinical trial settings that rely heavily on scheduled visits and subjective measures. Additionally, decentralized trials are positively impacting patient recruitment and retention by making these processes more diverse and easier. As more pharmaceutical companies and contract research organizations (CROs) adopt hybrid and fully virtual approaches for their clinical trials, digital biomarkers are playing a vital role in this transition.
Restraint: Data privacy and security concerns related to health data
The market for digital biomarkers is currently facing a significant challenge due to increasing concerns about privacy, security, and ownership of sensitive information. Continuous and passive monitoring technologies have the capacity to collect extensive amounts of detailed physiological, behavioral, and location-based data, which makes them susceptible to risks such as data breaches and cyberattacks. Furthermore, the difficulties in implementing international data protection regulations pose operational challenges for companies managing cross-border data flows. Additionally, the lack of transparency regarding the use of collected sensitive information can undermine patient trust.
Opportunity: Increasing use of digital biomarkers in drug development and clinical trial endpoints
The increasing adoption of digital biomarkers in drug development and trial endpoints presents a significant opportunity for this market. Pharmaceutical companies are increasingly utilizing digital biomarkers to collect continuous and objective real-world data about patients, which enhances their sensitivity to treatment outcomes. Additionally, these biomarkers can be employed to establish new digital trial endpoints, helping to minimize subjective assessments and improve trial efficiency. As regulatory bodies show a growing willingness to permit the use of digital trial endpoints, the application of digital biomarkers is expected to expand further. This advancement can lead to a more effective decision-making process in drug development.
Challenge: Lack of standardized validation frameworks for digital biomarkers
One of the main challenges facing the digital biomarker market is the absence of standardized validation frameworks. There is considerable uncertainty regarding the accuracy, reliability, and relevance of endpoints derived from digital methods. Unlike traditional biomarkers that depend on uniform data sources, digital biomarkers utilize varied sources such as smartphones, wearable devices, and algorithmic analytics. This heterogeneity complicates the establishment of consistent standards for data collection, processing, and interpretation. Additionally, there is a significant lack of clear regulatory guidelines for validating and qualifying these digitally derived endpoints. These challenges place increased pressure on companies to generate robust clinical evidence necessary for drug development. The absence of well-defined guidelines hinders the integration of digital biomarkers into drug development pipelines and slows their overall adoption in this field.
DIGITAL BIOMARKERS MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
AI-driven imaging biomarkers for neurological clinical trials (Alzheimer’s, Parkinson’s) | High-precision endpoints | Reduced trial variability | Improved CNS drug decisions |
 |
Real-world digital biomarker data capture and analytics platforms | Scalable insights | Continuous monitoring | Improved research outcomes |
 |
Wearable-based physiological monitoring for neurological and mental health conditions | Continuous tracking | Early event detection | High patient compliance |
 |
Smartphone-based ECG and AI rhythm analysis for arrhythmia detection | Remote cardiac monitoring | Early diagnosis | Reduced hospital visits |
 |
Integrated digital health platform for remote monitoring and patient-reported data | Better patient engagement | Personalized care | Improved disease management |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The digital biomarkers market ecosystem consists of various key players, including technology and platform suppliers such as Empatica Inc., AliveCor, Inc., Koneksa Health, and IXICO PLC. Additionally, emerging companies like Ametris, LLC and CONNEQT Health are focusing on developing AI-driven digital biomarkers and analytics platforms. The ecosystem is further strengthened by device and data capture enablers, including wearable technologies and smartphone applications. Key suppliers of data analytics and cloud infrastructure, such as Amazon Web Services, Google Cloud, and Microsoft, also contribute to the market. The pharmaceutical and biotechnology industries, along with contract research organizations (CROs), play a crucial role in adopting digital biomarkers in clinical trials and drug development processes. Moreover, regulatory bodies like the FDA and EMA are involved in establishing guidelines for digital endpoints and their validation processes. The healthcare sector also plays a significant role in developing the digital biomarkers market ecosystem.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Digital Biomarkers Market, By Type
The physiological biomarkers segment is the leading category in the digital biomarkers market, primarily due to the growing use of wearable technology. These devices constantly monitor vital health information, including heart rate, physical activity, sleep patterns, and respiratory rate, among others. Pharmaceutical companies and healthcare organizations prefer these biomarkers because they are highly validated clinically, easy to use, and capable of providing continuous information related to health conditions and clinical trials. Additionally, their seamless integration with commonly used devices like smartwatches and fitness trackers enhances their effectiveness in healthcare. This makes physiological biomarkers a dominant segment in the overall digital biomarkers market compared to other types of emerging biomarkers.
Digital Biomarkers Market, By Therapeutic Area
The cardiovascular segment holds the largest market share in the healthcare industry due to the high prevalence of cardiovascular conditions worldwide and the need for ongoing patient monitoring. Digital biomarkers derived from wearable technologies—such as heart rate, heart rate variability, electrocardiograms (ECG), and physical activity—are increasingly utilized to track patients with these conditions. Pharmaceutical companies are adopting these digital biomarkers for clinical trials, as they offer objective endpoints that enhance the reliability of the studies. Cardiovascular conditions are often chronic and may not present symptoms, emphasizing the importance of continuous monitoring to improve patient outcomes and reduce hospitalization risks. Additionally, the rising focus on preventive cardiology further solidifies the cardiovascular segment's dominance in the market.
Digital Biomarkers Market, By Application
The clinical research applications segment holds a significant market share in the digital biomarkers market. This is primarily due to the growing adoption of digital health technologies in clinical trials and drug development processes. The use of digital biomarkers—collected through wearable devices, mobile technologies, and sensors—allows for continuous, objective, and real-time data collection. This capability is crucial for enhancing endpoint measurement, patient monitoring, and evaluating drug treatment outcomes more efficiently. Pharmaceutical and biotech companies are increasingly utilizing digital biomarkers in clinical trials because they can improve trial processes, reduce costs, and support decentralized and hybrid trial designs. Additionally, the ability of digital biomarkers to generate high-frequency data in real-world settings is vital for assessing drug efficacy and safety, especially in late-stage clinical trials. The rising emphasis on incorporating real-world evidence in research, along with the need to enhance clinical development processes, contributes to this segment's dominant position in the digital biomarker market.
Digital Biomarkers Market, By End User
The pharmaceutical and biotechnology companies segment holds the largest market share in the digital biomarkers market among end users. This is primarily due to the growing emphasis on utilizing digital biomarkers in drug development and clinical trials. In this segment, pharmaceutical and biotechnology companies use wearable devices and mobile applications to continuously monitor patients' health and collect high-frequency data. This approach allows them to obtain objective endpoints that facilitate the efficient and precise evaluation of both the therapeutic efficacy and safety of drugs. Additionally, the increasing focus on personalized medicine and data-driven decision-making within these industries is further contributing to the market share of this segment in the digital biomarkers market.
REGION
Asia Pacific to register highest CAGR in digital biomarkers market during forecast period
The Asia Pacific market is expected to experience the fastest growth in the digital biomarkers sector during the forecast period. This growth is fueled by an increase in healthcare spending, a rising prevalence of chronic diseases, and a higher adoption of digital health technologies. Favorable government initiatives, expanded telemedicine infrastructure, and an increase in decentralized clinical trials are also contributing to market growth in the Asia Pacific region. Countries such as China and India, which have large patient populations, are driving the demand for continuous monitoring and the collection of real-world data through digital biomarkers. Additionally, a growing awareness of preventive healthcare, personalized medicine, and telemedicine services is further propelling the market for digital biomarkers in this region.

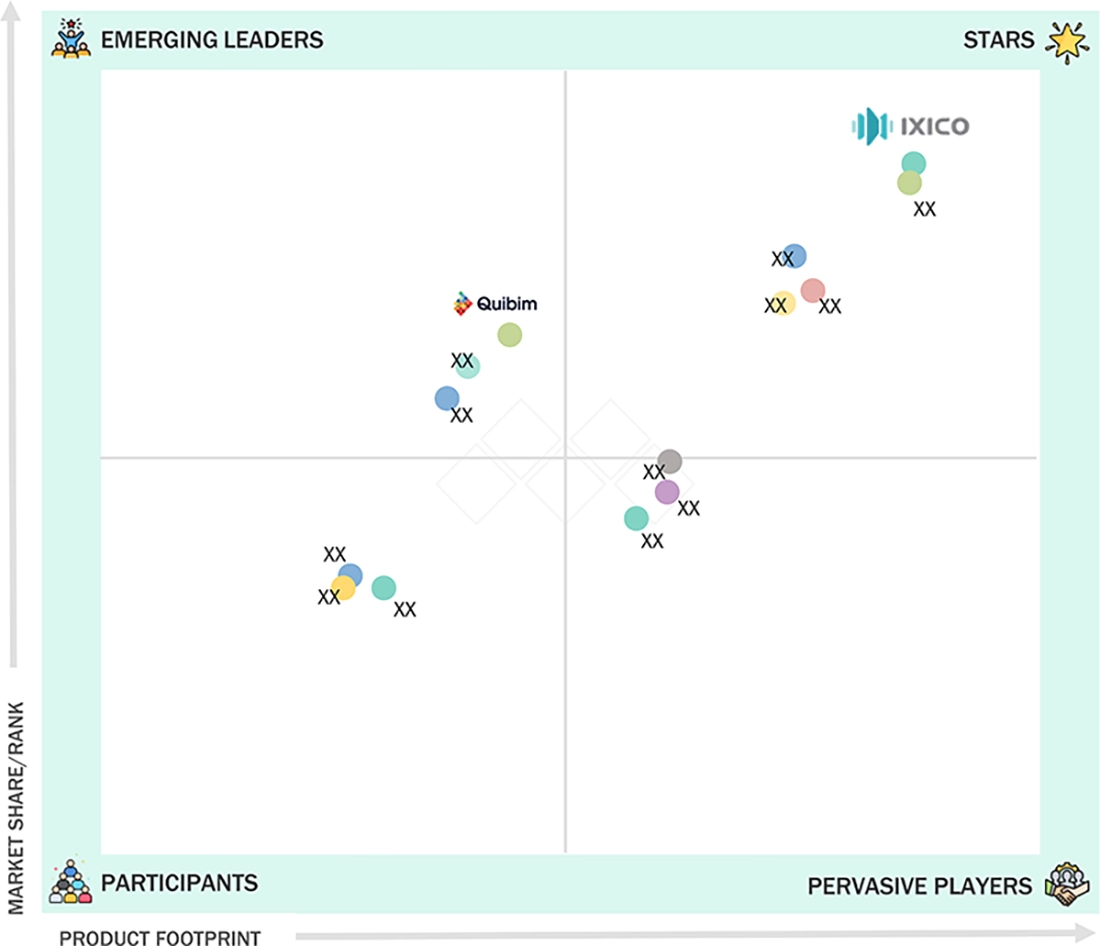
DIGITAL BIOMARKERS MARKET: COMPANY EVALUATION MATRIX
IXICO plc (Star Player) is a leading contributor to the digital biomarker field, thanks to its expertise in neuroimaging, data analysis, and the development of digital endpoints for clinical trials. The company is dedicated to providing its clients with reliable and high-quality digital biomarker information for disease monitoring and drug development, setting a benchmark for innovation and quality in the industry. On the other hand, Quibim (Emerging Leader) is an up-and-coming company utilizing advanced imaging analysis and artificial intelligence to create digital biomarkers for clinical research and patient monitoring. Quibim aims to expand its presence in this field by offering innovative, technology-driven solutions that enable precise data analysis and collection. While IXICO plc has a well-established portfolio and significant experience in trials, it is increasingly facing competition from emerging innovators like Quibim, who are making substantial advancements in the field.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- IXICO PLC (UK)
- Ametris, LLC (US)
- Empatica Inc. (US)
- AliveCor, Inc. (US)
- CONNEQT Health (US)
- VivoSense (US)
- BioSensics (US)
- Lunit Inc. (South Korea)
- Akili, Inc. (US)
- Quibim (Spain)
- Clario (US)
- Proscia Inc. (US)
- Koneksa Health (US)
- Linus Health (US)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size in 2025 (Value) (Base Year) | USD 6.29 Billion |
| Market Size in 2026 (Value) (Estimated Year) | USD 7.41 Billion |
| Market Forecast in 2031 (Value) (Forecast Year) | USD 17.73 Billion |
| CAGR | 19.1% |
| Years Considered | 2024–2031 |
| Base Year | 2025 |
| Forecast Period | 2026–2031 |
| Units Considered | USD Billion |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Regions Covered | North America, Asia Pacific, Europe, Middle East & Africa, Latin America |
| Related Segment & Geographic Reports |
Biomarkers Market North America Biomarkers Market Europe Biomarkers Market Asia Pacific Biomarkers Market Genomic Biomarkers Market Predictive Clinical Biomarkers Market Neurological Biomarkers Market EPO Biomarkers Market |
WHAT IS IN IT FOR YOU: DIGITAL BIOMARKERS MARKET REPORT CONTENT GUIDE
DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Competitive Landscape Mapping | In-depth information on the various digital biomarker companies and their products and market share in the market for physiological, behavioral, vocal, imaging, and cognitive biomarkers and various AI-based analytics solutions and software offered by them |
|
| Market Entry & Growth Strategy | Regional market assessment of the digital biomarkers market in terms of adoption rate, disease prevalence, and regulatory acceptance in the pharmaceutical and biotech industries and research institutions |
|
| Regulatory & Operational Risk Analysis | Assessment of the digital biomarkers market in terms of compliance with various regulations, such as FDA, EMA, CE, ISO, HIPAA, GDPR, and other local regulations in the field of medical device and data privacy regulations, HL7, FHIR, data privacy, security, and ethical issues in RPM and clinical research |
|
| Technology Adoption Trends | Information on the adoption of various AI and ML-based analytics solutions in the field of digital biomarkers in the areas of EHR, clinical trial research, population health management, and precision medicine initiatives and the current trends in multi-modal biomarker fusion and real-time monitoring |
|
RECENT DEVELOPMENTS
- February 2026 : AliveCor partnered with Ametris to integrate its medical-grade ECG monitoring technologies into the Ametris Digital Health Technology platform used for clinical trials. The partnership enabled remote collection of ECG data and QT interval measurements within decentralized study environments. By embedding AliveCor’s cardiac monitoring capabilities into the platform, the collaboration supported digital cardiac endpoints, improved patient accessibility, and reduced operational burden for clinical research sites and biopharmaceutical sponsors.
- September 2025 : Proscia expanded collaboration with AWS to integrate Concentriq with AWS HealthImaging, enabling scalable pathology image management and AI-driven analysis to accelerate digital biomarker development and precision medicine research.
- May 2024 : Lunit completed the acquisition of Volpara Health Technologies to expand its AI-driven cancer diagnostics portfolio. The integration combined Volpara’s breast imaging analytics with Lunit’s AI diagnostic technologies to create a comprehensive cancer screening and risk assessment platform, strengthening capabilities in population-scale breast cancer detection, workflow optimization, and personalized screening strategies.
Table of Contents

Methodology
The study involved five major activities to estimate the current size of the digital biomarkers market. Exhaustive secondary research was done to collect information on the market and its different subsegments. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. Thereafter, market breakdown and data triangulation procedures were used to estimate the market size of the segments and subsegments.
Secondary Research
In the secondary research process, various secondary sources such as annual reports, press releases & investor presentations of companies, white papers, certified publications, articles by recognized authors, gold-standard & silver-standard websites, regulatory bodies, and databases (D&B Hoovers, Bloomberg Business, and Factiva) were referred to identify and collect information for the study of the digital biomarkers market. It was also used to obtain important information about the top players, market classification, and segmentation according to industry trends to the bottom-most level, geographic markets, and key developments related to the market. A database of the key industry leaders was also prepared using secondary research.
Primary Research
Extensive primary research was conducted after acquiring basic knowledge about the global digital biomarkers market scenario through secondary research. Several primary interviews were conducted with market experts from the demand side (Hospital directors, Hospital Vice Presidents, Department heads, and Critical Care Specialists) and supply side (C-level and D-level Executives, Technology Experts, Product Managers, Marketing and Sales Managers, among others) across North America, Europe, the Asia Pacific, Latin America, Middle East, and Africa. This primary data was collected through questionnaires, e-mails, online surveys, personal interviews, and telephonic interviews.
The following is a breakdown of the primary respondents:
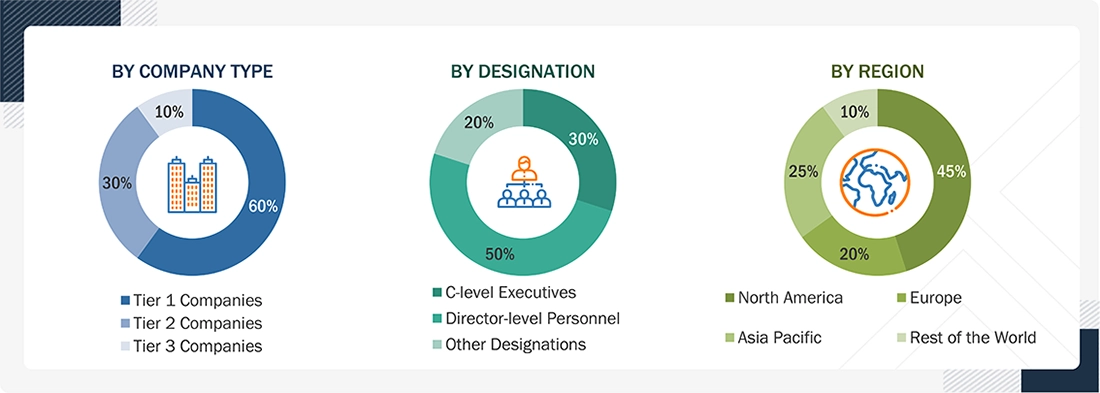
Notes:
- Others include Sales Managers, Marketing Managers, and Product Managers.
- Tiers of companies are defined on the basis of their total revenues in 2025.
- Tier 1 = > USD 1 billion, Tier 2 = USD 500 million to USD 1 billion, and Tier 3 = < USD 500 million
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
Both top-down and bottom-up approaches were used to estimate and validate the total size of the digital biomarkers market. These methods were also used extensively to estimate the size of various subsegments in the market.
Digital Biomarkers Market : Top-Down and Bottom-Up Approach

Data Triangulation
After arriving at the overall market size using the market size estimation processes, the market was split into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics of each market segment and sub-segment, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides in the digital biomarkers market.
Market Definition
The digital biomarkers market comprises software solutions and analytics platforms that collect and analyze data from connected devices to generate objective, quantifiable health indicators. These platforms use AI/ML and cloud technologies to enable disease monitoring, clinical decision-making, and digital endpoints in clinical trials.
Key Stakeholders
- Digital Health & Software Companies (AI analytics, Platforms, Mobile Apps)
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Healthcare Providers (Hospitals, Clinics, Telehealth Providers)
- Payers & Insurance Companies
- Regulatory Bodies (FDA, EMA, etc.)
- Academic & Research Institutions
- Cloud & Data Infrastructure Providers
- Patients & End Users
- Payers & Insurers
- Clinical Trial Sponsors & Investigators
- Data Analytics & AI Solution Providers
- Telehealth & Remote Patient Monitoring Providers
- EHR/Health IT Vendors
- Digital Therapeutics (DTx) Companies
- Government & Public Health Agencies
- Biomarker Validation & Certification Bodies
Report Objectives
- To define, describe, and forecast the global digital biomarkers market by type, therapeutic area, application, end user, and region
- To provide detailed information regarding the major factors (such as drivers, restraints, opportunities, and challenges) influencing market growth
- To strategically analyze micro-markets with respect to individual growth trends, prospects, and contributions to the overall digital biomarkers market
- To analyze opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders
- To strategically analyze the market structure profile of the key players of the digital biomarkers market and comprehensively analyze their core competencies
- To forecast the size of the market segments with respect to five regions: North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa
- To track and analyze competitive developments, such as product launches and enhancements, investments, partnerships, collaborations, acquisitions, expansions, product approval, and alliances in the digital biomarkers market during the forecast period
Available customizations:
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for the report:
Product Analysis
-
Product matrix, which gives a detailed comparison of the product portfolios of each company.
Regional Analysis
-
Further breakdown of Latin America, Europe, and the Middle East & Africa digital biomarkers into specific countries
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Digital Biomarkers Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free CustomisationTESTIMONIALS





















Growth opportunities and latent adjacency in Digital Biomarkers Market